Abstract
Background
HER family receptors play a key role in tumor progression in several malignancies, such as colorectal, lung or breast cancer. The aims of this study were to investigate expression of HER-1, HER-2 and HER-3 in pancreatic cancer (PC) samples and evaluate the association between HER-family receptor expression and patients’ clinical outcomes.
Methods
Tissue samples from 91 PC patients were subjected to immunohistochemical staining to assess the expression of HER-1, HER-2 and HER-3. Semiquantitative scores of zero (no staining or staining in less than 10% of cancer cells), 1+, 2+ or 3+ were assigned to each sample based on the intensity of staining for HER receptors. Scores of 2+ or 3+ were defined as positive staining.
Results
HER-1 overexpression was observed in 41 out of 91 samples (45.1%), while HER-2 was not overexpressed in any of the analyzed samples. HER-3 was overexpressed in 37 samples (40.7%) and was found to be associated with advanced TNM stage. In particular, HER-3 was overexpressed in 12 out of 16 stage IV patients (75%) compared with only 33.3% of stage I-III patients (p = 0.02). Among 79 patients with available survival data, the 6 patients with strong HER-3 expression (score 3+) had a shorter survival compared with remaining patients (median overall survival 6.9 months vs. 12.3 months, respectively).
Conclusions
HER-1 and HER-3 were found to be expressed in a significant proportion of PC patients. Strong HER-3 expression represents an indicator of poor prognosis in PC patients, being associated with advanced stage and shorter survival.
Introduction
Pancreatic cancer (PC) still represents one of the most lethal human malignancies, the fourth leading cause of cancer-related death worldwide, with an almost identical annual incidence and mortality rate. In this scenario, new effective treatments are urgently needed to improve patients’ prognosis. Agents directed against human epidermal growth factor receptors (HER or ErbB) have yielded promising results in several types of cancer, including gastrointestinal malignancies (1, 2). The ErbB family includes 4 members, the epidermal growth factor receptor (EGFR; HER-1 or ErbB1), HER-2 (ErbB2), HER-3 (ErbB3) and HER-4 (ErbB4) (3). The abnormal activation of these receptors has been linked with different pathological processes including malignant transformation (4), and overexpression of these proteins in cancer has been associated with worse prognosis (5).
In pancreatic cancer, EGFR overexpression has been reported in 30%-68% of tumors (6, 7), and it has been correlated with advanced stage (8). However, anti-EGFR treatment has not resulted in any improvement in pancreatic cancer. In particular, the addition of cetuximab to gemcitabine failed to show any benefit in clinical outcomes (9, 10), while erlotinib demonstrated only a modest improvement in overall survival (11).
Data about HER-2 overexpression in pancreatic cancer are largely variable, ranging from 0% to 82%. This is mainly due to differences in the definition of overexpression among the studies, as well as the fact that its effects on prognosis are inconsistent (12).
HER-3 overexpression has been reported in various tumor types, including breast, gastric and colorectal cancer, and it has been shown to identify a subtype of tumors with worse outcome in several studies (13, 14). Only a few studies found overexpression of HER-3 in pancreatic cancer (15, 16), and it has been demonstrated as an indicator of poor prognosis in 1 study (17).
Formation of dimers between different members of the ErbB family, such as EGFR and HER-3 or HER-2 and HER-3, seems to be crucial for the activation of the HER-driven signaling pathways in different cancers (18). A better knowledge of HER family receptor expression in pancreatic cancer may be useful to find new targets for therapy or predictive markers for response to anti-EGFR treatment.
The aims of this study were therefore to investigate immunohistochemical expression of HER-1, HER-2 ad HER-3 in pancreatic cancer samples and evaluate the association between HER-family receptor expression, clinicopathological features and patients’ clinical outcome.
Patients and Methods
Tumor samples from patients who were treated for pancreatic cancer between January 2001 and February 2011 were collected. Clinical and histopathological characteristics of patients, including age, sex, surgery, TNM stage and grading, were collected and analyzed. Data were retrospectively collected from medical chart reviews and electronic records. All study procedures were carried out in accordance with the Declaration of Helsinki on human research.
Immunohistochemistry
The tissue block with the highest tumor percentage based on the hematoxylin and eosin (H&E) stained sections was selected by the pathologist for further analysis.
The expression of HER-1 (EGFR pharmDX; Dako), HER-2 (HercepTest; Dako) and HER-3 (Monoclonal Mouse Anti-Human HER-3, Clone DAK-H3-IC; Dako) was evaluated with an immunohistochemistry technique on 5-μm-thick tissue sections obtained from paraffin-embedded specimens fixed in 10% (v/v) neutral buffered formalin. The sections were deparaffinized and hydrated by passing through xylene and a graded series of ethanol, followed by washing in distilled water. For HER-1 and HER-2 immunostaining was performed according to the manufacturer's instructions. For HER-3, peroxide blocking was performed with 3% H2O2 at room temperature for 10 minutes. The monoclonal mouse antibody anti-human HER-3 (DAK-H3-IC) was used (1:20 dilution; Dako), and samples were incubated overnight at 4°C. Incubation with the secondary antibody (EnVision System, Dako HRP; Dako) was performed for 30 minutes, followed by application of diaminobenzidine chromogen for 5 minutes. Subsequently, the slides were counterstained with Meyer's hematoxylin for 1 minute, dehydrated in a graded series of alcohol, treated with xylene, and coverslipped. The negative control for the validation of stainings was sections incubated with secondary alone without primary antibody. A positive control was included in each immunohistochemistry run.
The slides were scored by a pathologist blinded to the clinicopathological data. HER-1 and HER-2 membrane staining was scored according to the Dako scoring system. HER-3 expression was detected as homogeneous finely granular cytoplasmic staining of neoplastic cells and, rarely, membrane staining. Stained slices were evaluated semiquantitatively as negative (no staining or staining in less than 10% of cancer cells) or 1+ (11%-25%), 2+ (26%-50%) or 3+ (>50%). Overexpression was defined as 2+ or 3+ expression.
Statistical Analysis
The chi-square test was used to compare frequency distributions. Overall survival (OS) was defined from the time from the diagnosis to death or censoring. Survival analysis was conducted via Kaplan-Meier product-limit method, and the Mantel-Haenszel log-rank test was employed to compare survival among groups. A Cox regression model was applied to the data with a univariate and multivariate approach. Variables included in the univariate analysis were sex, age, stage, grading, surgical radicality (R0, R1, R2), HER-1, HER-2 and HER-3. Variables that were not significantly correlated with survival at univariate analysis were excluded from the multivariate model. Non-multicollinearity of the grouped covariates was checked. The significance level in the univariate model for inclusion in the multivariate final model was more liberally set at a 0.2 level.
Results
Characteristics of the Patients
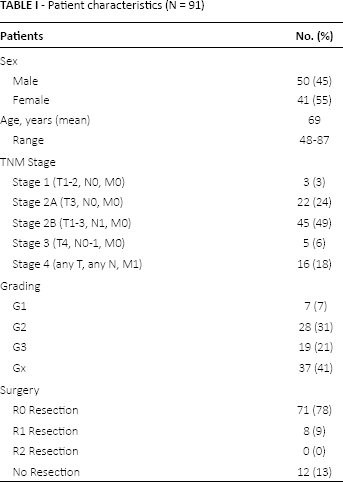
Patients characteristics are summarized in Table I. Pancreatic cancer samples were collected from 91 patients, 50 men and 41 women. Median age was 69 years (range 48-87). Twelve patients presented with stage IV disease at diagnosis and did not undergo surgery, while the remaining 79 patients underwent pancreatic cancer resection. Among those with the surgically removed tumors, 71 patients received a R0 resection, while in 8 patients, margins were microscopically involved (R1). In 4 of the 79 resected patients, distant metastases were found during surgery; in 2 cases, liver metastases were diagnosed, while in the other 2 cases, peritoneal involvement was found. Tumor grading showed 7 well-differentiated tumors (G1), 28 moderately differentiated (G2) and 19 poorly differentiated tumors (G3). Most of the patients presented with stage II disease – specifically, 22 with stage IIA and 45 with stage IIB disease – while 3 patients had stage I and 5 patients stage III disease. Sixteen patients had distant metastases (stage IV).
Patient characteristics (N = 91)
HER Expression
HER-1 staining was negative in 30 samples (33%), 1+ in 20 (22%), 2+ in 32 (35.2%) and 3+ in 9 (9.9%) tumors (Fig. 1). Overall, 41 samples (45.1%) showed HER-1 overexpression. HER-2 overexpression was not demonstrated in the samples analyzed, and only in 1 tumor sample was 1+ staining found (1.1%) in at least 10% of cells (Fig. 2). HER-3 staining was negative in 35 tumors (38.5%) and 1+ in 19 (20.9%). HER-3 overexpression was demonstrated in 37 tumors (40.7%): specifically, 28 samples had 2+ staining (30.8%) and 9 samples 3+ staining (9.9%). Fourteen samples showed concomitant overexpression of HER-1 and HER-3 (15.4%), while 27 tumors were negative for both HER-1 and HER-3 (29.7%).
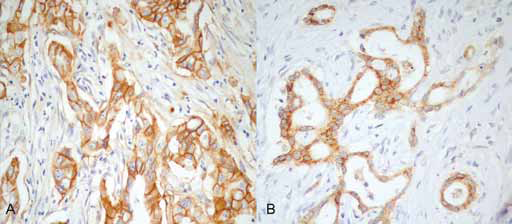
Immunohistochemistry examples.
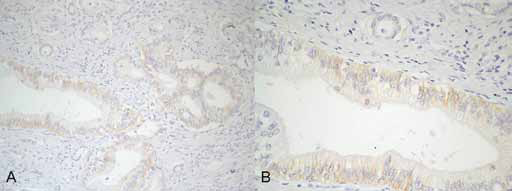
Immunohistochemistry examples.
Clinicopathological Correlations
HER-3 overexpression was significantly associated with advanced stage of disease. In particular, HER-3 was overexpressed in 12 out of 16 stage IV patients (75%), while in stage I-III patients, it was found to be overexpressed in only 33.3% of patients (p = 0.02). OS analysis of the 79 patients who underwent surgery showed a median OS of 12.1 months. HER-1 expression was not significantly associated with survival, with a median OS of 9.8 months for HER-1 positive patients versus 12.3 months for HER-1 negative patients (p = 0.45).
Then we compared the prognosis in terms of OS between patients with strong HER-1 expression (defined as 3+ staining) and patients negative for HER-1 or with low expression (0, 1+ or 2+) without finding any evidence of significant differences. In particular, we observed a median OS of 10.8 months and 12.1 months for patients with HER-1 overexpression and patients without HER-1 overexpression, respectively (p = 0.32) (Fig. 3).
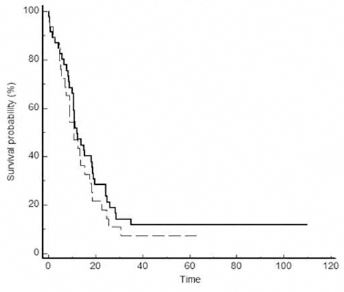
Overall survival (months) according to HER-1 expression. Dashed line represents HER-1: 3+; solid line indicates HER-1: 0, 1+ or 2+.
Also HER-3 expression was not significantly associated with survival. In particular, we observed a median OS of 12.1 months for patients without HER-3 expression (0 and 1+) compared with 8.9 months for patients with HER-3 expression (2+ and 3+) (p = 0.09). Since we found a trend for a shorter survival among patients with HER-3–positive tumors, we analyzed the outcomes for patients with strong HER-3 expression, defined as 3+ staining. In fact, the 6 patients with strong HER-3 expression (3+ staining) among the 79 resected patients, showed a significantly shorter survival compared with the other patients (median OS 6.7 months vs. 12.3 months, p = 0.035) (Fig. 4). In multivariate analysis, including stage, grading, surgery radicality and HER-3 overexpression (3+), only stage (p = 0.003) and HER-3 (p = 0.03) had independent prognostic value.
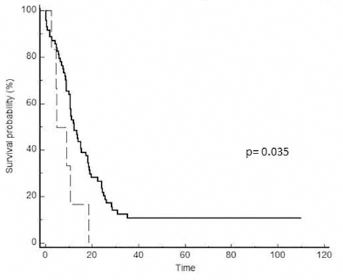
Overall survival (months) according to HER-3 expression. Dashed line represents HER-1: 3+; solid line indicates HER-1: 0, 1+ or 2+.
Discussion
Although HER family receptors have been shown to play a central role in tumor progression in different malignancies, their role in pancreatic cancer is not clearly defined.
The HER-1 overexpression rate observed in our study (45.1%) is comparable to the rates reported in the majority of published series, ranging from 30% to 68% (19, 20). The prognostic role of EGFR in pancreatic cancer has been evaluated in several studies with controversial results. A meta-analysis of 8 studies including 570 pancreatic cancer patients showed a significant prognostic value for EGFR with a hazard ratio of 1.225 (95% confidence interval [95% CI], 1.014-1.481; p = 0.035), indicating a negative impact of EGFR on patient survival (21). This negative prognostic impact was not confirmed in our study, where EGFR expression was not associated with survival.
Similar findings have also been reported for translational analysis of the prospective AIO-PK0104 trial, evaluating gemcitabine-erlotinib and capecitabine-erlotinib combinations in pancreatic cancer. In this study, a 49% rate of EGFR overexpression was detected (22). In spite of this common overexpression, the inhibition of EGFR showed disappointing results, and 2 randomized trials failed to show any benefit from the addition of cetuximab to chemotherapy in pancreatic cancer patients (9, 10). Moreover EGFR protein expression, as determined by immunohistochemistry, was not associated with tumor response, disease stability or any improvement in outcomes (9).
Studies of HER-2 expression in pancreatic cancer have yielded conflicting results. In our analysis, we did not observe overexpression of HER-2, similar to what was reported in a study of 33 pancreatic cancer samples by Potti et al (23). Interestingly, a study by Day et al in a series of 19 pancreatic cancer samples reported high levels of HER-2 expression in pancreatic carcinoma in situ with a decreasing expression in infiltrating adenocarcinomas (24). Nevertheless, in a recent study (25), HER-2 overexpression was demonstrated in 9 out of 87 samples (10.3%) examined, while other studies have shown HER-2 expression in up to 20%-45% of patients with pancreatic cancer (26, 27). This difference may be partly explained by the definition of overexpression applied in the studies. Indeed, in some older studies, the cutoff for overexpression was 1+ and not 2+ or 3+ (26, 28). Moreover, also differences in immunohistochemistry technique may explain these conflicting results. In a review of the literature, Saxby et al found that 12 different antibodies were used in 17 different immunohistochemical studies with variable results regarding the number of positive samples (29).
In a recent phase II study in 212 pancreatic cancer patients treated with capecitabine and trastuzumab, HER-2 overexpression was found in about 25% of samples. The study showed a lack of correlation between HER-2 immunohistochemistry overexpression and gene amplification, which was different from what was observed in other tumors such as breast or gastric cancer. This result seems to suggest that HER-2 overexpression in pancreatic cancer may be due to gene deregulation rather than gene amplification (30).
About 40% of pancreatic cancer patients included in our analysis presented HER-3 overexpression, and it was associated with advanced stage. Similar results were reported by Friess et al (31) in terms of overexpression rate (47%) as well as association with a more advanced stage. Interestingly, in our study, patients with resected pancreatic cancer and HER-3 strong expression (3+) showed a significantly shorter OS (median OS 6.7 months vs. 12.3 months, p = 0.035). This negative prognostic value for HER-3 in pancreatic cancer was also suggested by a previous study of 126 resected pancreatic cancer patients (32).
These data have been strengthened by a meta-analysis which assessed the prognostic role of HER-3 in different solid tumors, including pancreatic cancer (13). The median percentage of cancers with HER-3 overexpression was 42.2%, overexpression of HER-3 was associated with worse survival in all studies analyzed except for 2 studies of colorectal cancer and HER-3 was confirmed as a negative prognostic factor for OS at both 3 years (odd ratio [OR] = 2.24, 95% CI, 1.77-2.83, p<.001) and 5 years (OR = 2.20, 95% CI, 1.75-2.76, p<0.001). These results suggest that HER-3 is likely to represent an interesting target in pancreatic cancer. Indeed, strategies to inhibit HER-3 are currently in clinical development (33).
Moreover, our study showed a percentage of patients, about 15%, with concomitant overexpression of HER-1 and HER-3, suggesting a possible subgroup of tumors that may benefit from a combination of anti-EGFR and anti-HER-3 treatment. Data on HER-1 and HER-3 coexpression are also interesting, considering the possible role of HER-3 in mediating resistance to anti-EGFR treatment. Indeed, preclinical data have suggested that HER-3 biological activity as an EGFR lateral signaling partner may have a crucial function in mediating de novo or acquired resistance to anti-EGFR treatment in pancreatic cancer cell lines (34). Moreover, HER-3 overexpression has been proven to be a negative predictive factor for clinical outcomes in metastatic colorectal cancer patients treated with cetuximab in a retrospective analysis of 84 patients.
In pancreatic cancer, several clinical factors with prognostic significance, such as stage of disease or performance status or biochemical markers such as serum CA 19.9, have already been identified. In contrast, validated molecular prognostic markers or even predictive biomarkers for the treatment efficacy of biological agents (such as erlotinib or cetuximab) are still lacking. Our study has demonstrated that HER-3 is a negative prognostic factor in resected pancreatic cancer, indicating a role of this receptor in pancreatic cancer development and suggesting the evaluation of anti-HER-3 strategies in the treatment of this disease.
Footnotes
Financial support: No grants or funding has been received for this study.
Conflict of interest: The authors declare they have no conflict of interest regarding the publication of this paper.
