Abstract
Background
The aim of this study was to investigate the impact of number of circulating tumor cells (CTCs) on the treatment outcome for metastatic gastric cancer (GC) following palliative chemotherapy.
Methods
CTCs were isolated from 7.5 mL of whole blood from 100 patients with metastatic GC by anti-EpCAM antibody coated magnetic particles using the CTC-Profiler (Veridex). Correlations between CTC counts and clinicopathological variables, progression-free survival and overall survival were examined.
Results
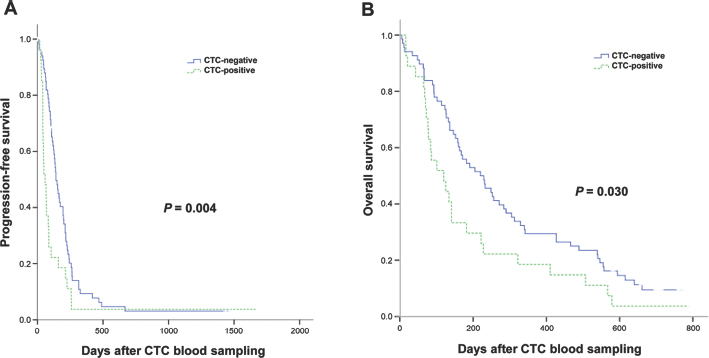
Between January 2010 and August 2010, 100 metastatic GC patients were enlisted. Among 100 patients, 5 or more CTCs (CTC-positive) were detected in 27 of 95 patients (28%). Even though the clinical characteristics of the CTC-positive and CTC-negative groups were not significantly different, the treatment response to cytotoxic chemotherapy in the CTC-positive group was significantly poorer (progressive disease: 23.4% vs. 60.0% in CTC-negative vs. CTC-positive group, respectively; p = 0.004). The median progression-free survival of the CTC-positive group was substantially shorter than that of the CTC-negative group (59 days vs. 141 days; p = 0.004). For overall survival, CTC-positive group had significantly shorter survival than CTC-negative group (median OS, 120 days vs. 220 days; p = 0.030). A multivariate Cox proportional hazards regression model showed that CTC positivity was an independent adverse factor for progression-free survival and overall survival.
Conclusions
This study suggests CTCs are associated with poor response to chemotherapy in metastatic GC patients.
Introduction
Gastric cancer (GC) is a major global health problem as it is the fourth most common type of cancer and the second most common cause of cancer-related death worldwide (1). In an inoperable setting, 5-year survival is around 5%-20%, with median overall survival being less than 1 year (2, 3). Although the incidence of GC is declining, the prognosis for the disease remains poor.
Recently, several studies have focused on circulating tumor cells (CTCs) in patients with various types of cancers (4-8). CTC has been suggested as an early biomarker of tumor recurrence after curative surgery, and CTC quantitation predicted poor prognosis in a number of epithelial-derived cancers (9, 10). The most important clinical implication of CTC is as a real-time liquid biopsy (11). CellSearch system (Veridex LLC, Warren, NJ, USA) was designed to detect tumor cells in blood. Using the CellSearch system, it became possible to obtain highly reproducible quantitative results from different laboratories (12). Clinical significance of CTCs assessed by the CellSearch system was first reported in metastatic breast cancer, which showed CTCs as poor prognostic marker (13, 14).
Allard et al (12) reported that CTCs were detected in patients with various types of carcinomas, including gastrointestinal cancer. In addition, it has been suggested that the presence of CTCs is an independent adverse factor for recurrence in GC patients following surgical resection (15). Another Japanese study reported on a survey of CTCs in 130 GI cancer patients (GC, n = 27), which demonstrated that CTCs were correlated with advanced tumor stage, peritoneal seeding and shorter survival (16).
In this study, we evaluated the detection rate of CTCs by the CellSearch system in 100 metastatic GC patients and analyzed the clinical implications of the presence of CTCs in terms of response to chemotherapy and survival.
Materials and Methods
Patients
Eligibility criteria were as follows: pathologically confirmed of metastatic gastric adenocarcinoma, age >18 years, Eastern Cooperative Oncology Group (ECOG) performance status 0-2 and written informed consent. This study was approved by the institutional review board of the Samsung Medical Center, Seoul, South Korea. All study participants provided a written informed consent form. This protocol was registered with clinicaltrials.gov (NCT#01299688).
CTC measurement
For CTC evaluation, 7.5 mL of whole blood was taken from the patients, collected in evacuated ethylenediaminetetraacetic acid (EDTA) tubes and processed for CTC isolation within 24-48 hours after collection. Peripheral blood samples were enriched with the CellSearch system (Veridex) which used ferrofluids conjugated to antibody against epithelial cell adhesion molecule (EpCAM). The enriched EpCAM+ population samples were stained with phycoerythrin-conjugated antibodies against CK8, CK18 and CK19 with allophycocyanin-conjugated antibodies specific for leukocytes (anti-CD45 antibody) and with the nuclear dye 4’,6-diamidino-2-phenylindole (DAPI) for nucleic acid staining. The CK+/DAPI+/CD45- cells were counted as CTCs using a CellTracks Analyzer II (Veridex), a 4-color semiautomated fluorescent microscope.
Statistical Analysis
For continuous variables, data were expressed as means ± standard deviation and compared by the Mann-Whitney test. Association between categorical variables was analyzed using a chi-square test. Progression-free survival (PFS) was calculated as the period from the date of blood sampling to the date of progression or death from any cause. Overall survival (OS) was defined as the period from the date of blood sampling to the date of death from any cause or the last follow-up. Survival analysis was done using the Kaplan-Meier method and log-rank test. Cox's proportional hazard model was used for multivariate analyses. Findings were considered significant when the p value was <0.05. All tests were performed using SPSS, version 18.0 (SPSS Inc., Chicago, IL, USA). For evaluating tumor response, a computed tomography (CT) scan was performed every 2 cycles of chemotherapy and RECIST v.1.1 was used as response criteria (17).
Results
Detection of CTCs
From January 2010 to August 2010, a total of 100 patients with metastatic GC were prospectively enrolled in this study (Fig. 1). Using the CellSearch system, at least 1 CTC was detected in 43 of 95 samples (45.3%) tested. The mean number of detected CTCs was 54 (range 0-2,500 cells) in all patients. We considered ≥5 CTCs per 7.5 mL of blood as a cutoff value for positivity as previously described (13, 18). There were 27 GC patients (28.4%) with ≥5 CTCs in our patient cohort. The baseline characteristics of these 2 groups (<5 CTCs vs. ≥5 CTCs) are shown in Table I. All patients were metastatic, with peritoneal seeding being the most common site of metastasis. At the time of CTC collection, approximately half of the patients were on first-line chemotherapy, one third on second-line chemotherapy and others on third-line or more of chemotherapy. Most patients had CTCs collected at the start of each line of therapy. There were no significant differences in clinicopathological features between CTC-positive vs CTC-negative groups.
Baseline characteristics of the patients (N = 95)
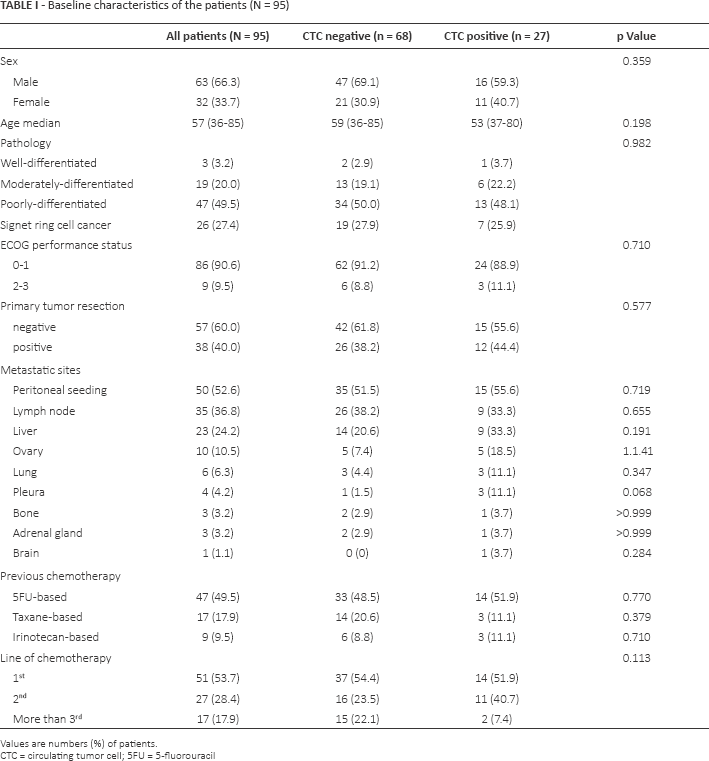
Values are numbers (%) of patients.
CTC = circulating tumor cell; 5FU = 5-fluorouracil
CONSORT flow chart of this study. CTC = circulating tumor cell.
CTCs on Response to Chemotherapy and PFS
Next, we analyzed the correlation between CTC positivity and response to chemotherapy, PFS and OS. In patients with CTC-positive results, more progressive disease cases were observed than in CTC-negative patients (60.0% vs. 23.4%, p = 0.004) (Tab. II). Likewise, CTC-positive patients had significantly worse median PFS (59 days) when compared with that of CTC-negative patients (141 days) (p<0.004) (Fig. 2A). In terms of OS, CTC-positive GC patients had significantly shorter median survival (120 days) when compared with CTC-negative GC patients (220 days) (p = 0.030) (Fig. 2B).
Response to chemotherapy according to CTC positivity
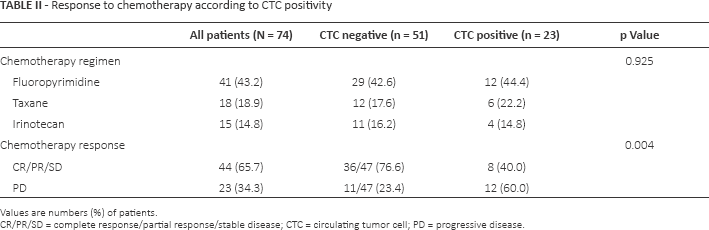
Values are numbers (%) of patients.
CR/PR/SD = complete response/partial response/stable disease; CTC = circulating tumor cell; PD = progressive disease.
In the univariate analysis, CTC positivity, lines of chemotherapy (1 or >1) and ECOG performance status were significantly associated with both PFS and OS. At multivariate analysis, CTC positivity retained its statistical significance (p = 0.004) with a hazard ratio of 2.062 (95% confidence interval [95% CI], 1.259-3.375) (Tab. III), even after factoring out the lines of chemotherapy. Hence, regardless of time points when CTC was collected, CTC positivity was an independent adverse factor for poor PFS or OS.
Multivariate analysis for progression-free survival and overall survival
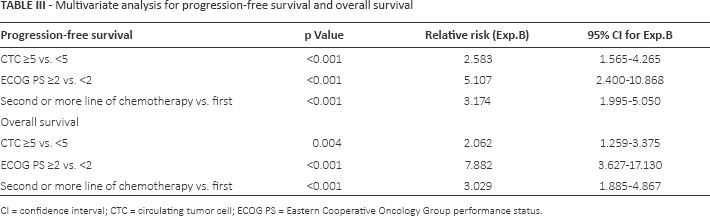
CI = confidence interval; CTC = circulating tumor cell; ECOG PS = Eastern Cooperative Oncology Group performance status.
Discussion
In this study, we surveyed the prevalence of CTCs using the CellSearch system in metastatic GC patients and investigated the correlation between CTCs and response to chemotherapy. This study represents one of the largest studies of metastatic GC. We have demonstrated that 28% of metastatic GC patients do have CTCs of more than 5 in number. The study cohort consisted of patients with metastatic disease, with half of them being on second or later lines of chemotherapy at the time of CTC collection.
Recently, the clinical implications of CTCs were analyzed in 251 GC patients, and it was reported that patients with CTCs demonstrated poorer survival. The incidence of detected CTCs (defined as at least 1 CTC) was similar between the Japan study (10.8%-62.0%) and our study (45.3%). The mean number of enumerated CTCs was 54 cells in our study (range 0-2,500), while the mean count was 109.3 cells in the unresected GC group in the Japanese study (19). In line with their study, which reported poor survival of the CTC-positive group, our CTC-positive patients who were undergoing palliative chemotherapy demonstrated significantly poorer responses to chemotherapy and shorter PFS and OS when compared with CTC-negative patients. Another Japanese study quantified CTCs in 52 GC patients who were treated with TS-1 or paclitaxel (20). They used a cutoff value of ≥4 CTCs and analyzed CTCs at baseline, 2 and 4 weeks. Similar to our results, they showed that patients with ≥4 CTCs at 2 weeks demonstrated shorter survival (3.5 months) than those with <4 CTCs at 2 weeks from chemotherapy (20).
One of the weaknesses of our study was that we included all metastatic GC patients on first-line, second-line and third-line chemotherapy. Nevertheless, we showed that the CTC ≥5 group remained statistically significantly different after factoring out performance status and lines of chemotherapy at multivariate analysis (Tab. III). Hence, although definitive conclusions cannot be drawn from 95 GC patients, patients with CTC ≥5 at any time during treatment may represent a group with poor prognosis with palliative chemotherapy.
One of the plausible reasons for our observation is that CTCs may potentially reflect tumor burden of the patient. However, in order to define the role of CTCs in clinical practice, prospective trials incorporating CTCs at the time of evaluation need to be conducted.
In conclusion, our data suggest that identifying CTCs may be predictive of poor response to chemotherapy regardless of number of lines of chemotherapy. Furthermore, patients with a high CTC number were prone to progressive disease and to die earlier than those with low or no CTCs during palliative chemotherapy. Further molecular characterization of CTCs is needed to investigate whether CTCs truly represent the metastatic disease with concordant molecular features such as HER2 amplification.
Footnotes
Acknowledgement
We would like to thank Dr. Phillip Kim for his scientific advice.
Financial Support: This work was supported by a grant from the Korean Health Technology R&D Project, Ministry of Health & Welfare, Republic of Korea (HI14C2188) and by grants from the 20 by 20 project of Samsung Medical Center (GF01140111). The study was also supported by the Samsung Biomedical Research Institute (#SMX1131871).
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
