Abstract
Background
8-Hydroxydeoxyguanosine (8-OHdG) is a commonly used marker of DNA oxidative stress in epidemiological studies. The aim of this study was to establish whether the urinary concentration of 8-OHdG varies during the first part of the day, when clinical tests are usually performed, and whether it can therefore be measured without bias in spot urine samples.
Material and methods
Spot urine samples were collected using a convenience sample. A linear mixed-effects model for repeated measurements was used to analyze 8-OHdG levels.
Results
A significant increasing trend in time in the 8-OHdG concentration was found among smokers, but not in the case of nonsmokers.
Conclusions
In epidemiological studies on oxidative stress, all participants should collect their early morning urine specimens – before their first cigarette if they are smokers – to gather information on individual background oxidation levels.
Keywords
Introduction
The measurement of oxidative stress is assuming increasing importance. In fact, oxidative stress is linked to many pathologies: an increase in DNA oxidation has been seen in some cancers, in some hematological disorders, in diabetics, in diabetic nephropathy patients and in subjects at risk of coronary heart diseases (1). One of the most used biomarkers to measure oxidative stress is 8-hydroxydeoxyguanosine (8-OHdG) (2), an oxidized nucleoside of DNA also known as 8-oxo-7, 8-dihydro-2-deoxyguanosine (8-oxodG), which upon DNA repair is excreted in the urine.
Some respiratory diseases, such as chronic obstructive pulmonary disease, have been correlated to a high level of 8-OHdG (3). 8-OHdG levels are also high in some stressful situations including smoking, aging, lack of or extreme exercise and occupational exposure to chemicals (4, 5). Individual 8-OHdG levels are usually measured in leukocytic DNA or in the urine. The measurement of urinary 8-OHdG offers some advantages as it is noninvasive, its excretion is likely to reflect the oxidative DNA damage and repair from all cells in the organism, and 8-OHdG shows high stability in urine (2). Although the current gold standard assessment of 8-OHdG is carried out by chromatographic methods using high-performance liquid chromatography (HPLC) or liquid chromatography–electrospray tandem mass spectrometry (LC-MS/MS) (2), in epidemiological surveys commercial 8-OHdG ELISA kits are often used, which are less costly and time consuming than chromatography. Furthermore, they have been shown to correlate with the gold standard, when conducted in particular conditions (6).
It is usually recommended to collect 24-hour urine (2), but in large epidemiological studies, that is impractical, so some studies have explored the possibility of using spot morning urine samples, concluding that it is feasible to analyze spot morning urine samples instead of those collected during 24 hours, due to the consistent correlation between the 2 measurements (2).
Over the course of the day, the composition and concentration of urine changes continuously. The first voided morning specimen (upon waking) is particularly valuable because it provides a time average for biomarker concentrations that may occur during the hours of sleep (approximately 8 hours). An (early) morning specimen is also relatively free of dietary influences. A late morning (or early afternoon) specimen may reflect dietary, physical and environmental (i.e., tobacco smoke, pollution) exposures.
Very few studies have investigated the influence of urine sampling time on 8-OHdG concentration levels (7). For this reason, we performed a pilot study aimed at testing whether the 8-OHdG concentration varies according to the time the spot urine sample is collected. During the day, 3 specimens were collected: the first voided morning specimen, a specimen in the morning before lunch and a specimen after lunch in the early afternoon. The reason for 3 urine collection times in this pilot study was to imitate an epidemiological survey where a subject may be asked to self-collect urine after waking up, or to provide a urine sample during a clinical checkup in the morning or in the early afternoon.
The results of this pilot study can be applied to epidemiological studies where biological markers are planned to be measured.
Materials and Methods
Sample Collection and Questionnaire
Spot urine samples were collected using a convenience sampling of 55 volunteers (our colleagues), 3 times a day: early morning after waking up, at about 7.00
Urine samples were stored at −80°C until they were analyzed. Urine sample aliquots were used in an ELISA competitive assay kit (Cosmo Bio Co., Tokyo Japan) for the quantification of 8-OHdG (ng/mL). The urinary creatinine concentration (mg/mL) was quantified using another ELISA assay kit (Cayman Chemical, Ann Arbor, MI, USA), and it was used to relate data to a homogeneous reference parameter. The resulting creatinine-corrected 8-OHdG concentrations (ng/mg) were used for the analyses.
All of the volunteers filled in a questionnaire on height, weight, sex, age, smoking habits, fruit and vegetable consumption and physical activity. The volunteers were classified as “smokers” if they were “current smokers” (1 or more cigarettes a day for at least 1 year). Questions about smoking habits and physical activity were taken from the ECRHS II Questionnaire (www.ecrhs.org/quests.htm) (8, 9), and the questions about consumption of fruit and vegetables were based on the scientific literature.
Statistical Analysis
Usual descriptive statistics were used to summarize the sample characteristics. To normalize the distribution of the 8-OHdG concentration, the Box-Cox transformation was used. Univariate associations between participants’ characteristics (age, sex, body mass index [BMI], smoking habits [smoker/nonsmoker], physical activity, fruit and vegetable consumption, urine collection time as a continuous variable) and 8-OHdG concentration levels were evaluated with a linear mixed-effects regression model for repeated measurements.
A multivariable linear mixed-effects regression model was used to analyze whether 8-OHdG levels varied with times of urine collection, adjusting for BMI, age, smoking habits and sex. Independent variables were retained in the model according to their statistical significance at the univariate analysis and/or according to biological plausibility. Interactions between sampling time and participants’ characteristics were studied when appropriate by including interaction terms in the regression model.
A sensitivity analysis was performed excluding a potential outlier (8-OHdG concentration equal to 40.13 ng/mg). All of the analyses were performed with Stata version 9 (http://www.stata.com) and R version 2.6.2 (R Development Core Team 2008; http://www.r-project.org).
Results
The sample consisted of 22 men (40%) and 33 women (60%), aged 24-61 years; 30.9% of the participants were smokers, and they dedicated a median of 2 hours per week to physical activities (Tab. I). The median of 8-OHdG concentration varied from a minimum of 3.76 ng/mg (observed in men in the early morning) to a maximum of 6.46 ng/mg (observed in men in the early afternoon).
Participants’ characteristics and 8-OHdG/creatinine concentrations stratified by sex
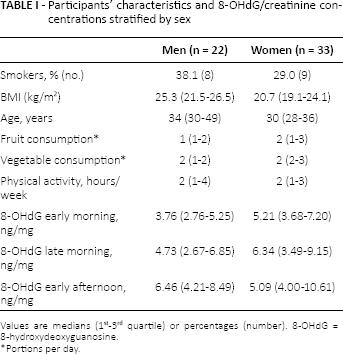
Values are medians (1st-3rd quartile) or percentages (number). 8-OHdG = 8-hydroxydeoxyguanosine.
Portions per day.
On univariate analysis (Tab. II), level of normalized urinary 8-OHdG/creatinine (ng/mg) decreased with increasing BMI (p = 0.049) and increased with urine collection time (p = 0.024). Specifically, the concentration of 8-OHdG slightly increased from early morning to early afternoon (early morning, median: 4.41 ng/mg, interquartile range [IQR] = 3.27 ng/mg; before lunch, median: 6.03 ng/mg, IQR = 5.03 ng/mg; and early afternoon, median 7.32 ng/mg, IQR = 6.48 ng/mg).
No other significant associations were found for the remaining variables considered (Tab. II). After excluding the outlier, all of these results were confirmed. In the multivariable analysis (Tab. III), a statistically significant interaction between smoking habits and diurnal variation of urinary 8-OHdG was found (p = 0.011). The concentration of 8-OHdG did not vary in the urine collection times among nonsmokers, while it showed a significantly increasing trend in smokers from the morning to the afternoon (Fig. 1). This result was confirmed after excluding the outlier (p = 0.007).
Associations between levels of normalized urinary 8-OHdG/creatinine (ng/mg) and participants’ characteristics
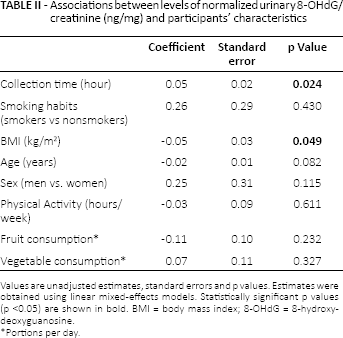
Values are unadjusted estimates, standard errors and p values. Estimates were obtained using linear mixed-effects models. Statistically significant p values (p <0.05) are shown in bold. BMI = body mass index; 8-OHdG = 8-hydroxydeoxyguanosine.
Portions per day.
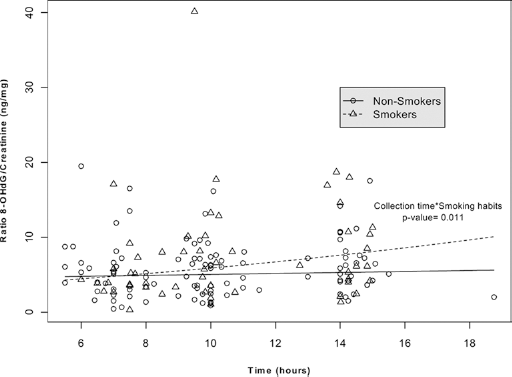
Fitted regression lines for trends in time for 8-hydroxydeoxyguanosine (8-OHdG) levels by smoking status: 8-OHdG values are reported for smokers (triangles, dashed line) and nonsmokers (circles, continuous line) during the first part of the day.
Further, another statistically significant interaction was observed between smoking habits and BMI (Tab. III): 8-OHdG levels significantly decreased as BMI increased, in smokers only (p = 0.040). This interaction only reached borderline significance (p = 0.065) when we excluded the outlier from the analysis.
Associations between level of normalized urinary 8-OHdG/creatinine (ng/mg) and participants’ characteristics
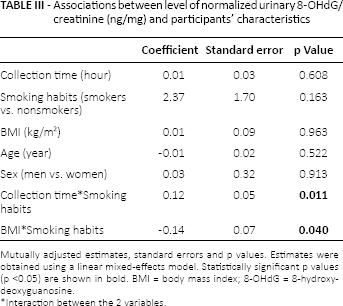
Mutually adjusted estimates, standard errors and p values. Estimates were obtained using a linear mixed-effects model. Statistically significant p values (p <0.05) are shown in bold. BMI = body mass index; 8-OHdG = 8-hydroxydeoxyguanosine.
Interaction between the 2 variables.
Discussion
This pilot study shows that the concentration of creatinine-corrected 8-OHdG does not vary by urine collection times among nonsmokers, in spite of a statistically significant increasing trend in smokers from the morning to the afternoon. In a study on the acute effect of smoke on oxidative stress (10), a 1.5-fold increase in leukocyte 8-OHdG was observed in 10 healthy men, 10 minutes after they had smoked 2 cigarettes. The acute effect of smoking was widely analyzed in a review by Van der Vaart et al (11): an increase of 8-OHdG in animals’ tissues 1 hour after smoking exposure was shown. There are still no scientific investigations on the effect of acute smoking in urine. However, our study suggests that the growing level of 8-OHdG in urine quickly after smoking can be caused by its short-term effect. These data imply that in epidemiological studies where the focus is not on the acute effects of smoking, but on having a measurement of the background effect (nonacute) of the exposure, smokers should collect their early morning urine specimens (before their first cigarette), while nonsmokers may collect their spot urine at any time during the first part of the day. For simplicity, it would be preferable to collect early morning urine from all participants.
In our univariate analysis, we found a tendency toward rising 8-OHdG concentrations (p = 0.024) from the morning until the early afternoon, in the data collected on the whole convenience sample. However, Andreoli et al (7) did not find any statistically significant trend in urinary levels for this biomarker during the day. But it should be mentioned that they observed nonsmoking volunteers only.
As already reported, we observed an increase in 8-OHdG levels in smokers, but not in nonsmokers, as the day went by. Tamae et al (12), Doruk et al (13) and Campos et al (14) detected greater oxidative DNA damage in smokers.
We found a significant decrease of 8-OHdG concentration at higher BMI in smokers, but not in nonsmokers. Van Zeeland et al (15) also found similar results: lower levels of leukocyte 8-OHdG in smokers compared with nonsmokers, and an inverse relationship between 8-OHdG and lifetime smoking. On the other hand, in a study of healthy Japanese, no difference was shown for either BMI or smoking habits (5), while Mizoue et al (16) observed a significant decrease in 8-OHdG levels as BMI increased.
With regard to other potential confounders considered in this pilot study, we found no association of 8-OHdG levels with age. The literature data on this topic are discordant: Miwa et al (17) showed a decreasing level at an older age, Barregard et al (2) found no association, but Sakano et al (5) pointed out that there was a higher level of urinary 8-OHdG in people aged over 40.
No sex difference was highlighted in our study, consistent with the study by Barregard et al (2), while Nakano et al (18) found a significantly higher concentration of 8-OHdG in men compared with premenopausal women, but in the case of postmenopausal women, this relation was reversed.
In our pilot study, no significant association was found between the consumption of fruit and vegetables, on the one hand, and 8-OHdG levels, on the other. Kim et al (19) did not observe a decrease of this biomarker in their study of people with diets rich in fruit and vegetables either. On the other hand, Tamae et al (12) found an inverse correlation between urinary 8-OHdG and fruit consumption.
While we did not find any association between DNA oxidative stress and physical activity, Powers et al (20) showed that extreme prolonged exercise causes oxidative DNA damage.
We used ELISA commercial kits in this study, instead of the chromatographic and mass spectrometry techniques, which are the elective methods for this analyte (2), as these techniques are expensive and time consuming. Even if ELISA kits show a good correlation with the results from chromatography, they overestimate 8-OHdG concentrations (2). This can be only a partial problem in epidemiological surveys, where finding any association with possible determinants is usually more important than detecting the true value of a substance, as in our case.
Interest in measuring oxidative stress in relation to specific pathological processes is increasing, since the scientific community is paying growing attention to this process which could be the potential cause of many chronic diseases. For this purpose, in many epidemiological studies, people from the general population are invited to clinical centers where they may be asked to undergo a clinical examination and/or physiological tests and to provide urine specimens for biomarker assessment. However, measuring oxidative stress in a large number of subjects, which is common practice in epidemiological studies, is complex. In particular, participants are more willing to have a urine test than a blood test, and they prefer to provide a spot rather than a 24-hour urine specimen. In fact, 24-hour urine collection can lead to a drastic reduction in participation in a survey. Nevertheless, some studies have found a correlation between early morning spot and 24-hour urine (2), which may greatly simplify the recruitment of subjects.
In this study, we wanted to assess whether the urine collection time was associated with a variation in urinary concentration of 8-OHdG and to establish whether it was possible to collect a spot urine sample at any time during the morning or early afternoon.
In conclusion, the main finding of this pilot study is that urine concentrations of creatinine-corrected 8-OHdG increase during the first part of the day in smokers, whereas they do not change in healthy nonsmokers. Because epidemiological studies on oxidative stress are usually addressed to gathering reliable measurements of individual background oxidation levels, participants should collect early morning urine specimens – before their first cigarette if they are smokers.
Footnotes
Acknowledgements
We thank Dr. Silvia Sembeni and Dr. Liliya Chamitava for their precious support.
Financial support: No grants or funding have been received for this study.
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
