Abstract
Background
Although recent evidence has implicated viruses in the regulation of epithelial-to-mesenchymal transition and tumor progression, little is known regarding viral infections in thyroid malignancies. Thus the aim of this study was to detect sequences of 3 potentially oncogenic viruses – BK virus (BKV), Epstein-Barr virus (EBV) and human papillomavirus (HPV) – in a series of postoperative thyroid gland specimens.
Methods
Thirty patients with thyroid nodules who underwent surgery for thyroid disease within a 3-year period were enrolled. Both nodular and adjacent normal thyroid tissue was surgically excised from each patient. Viral gene sequences of BKV (VP1), EBV (LMP1, EBNA2 and EBER1) and HPV were amplified by PCR. The PCR results were confirmed by direct sequencing analysis.
Results
VP1 gene sequences were detected in 60% (18/30) of thyroid cancer or multinodular hyperplasia lesions compared with in 43.3% (13/30) of adjacent normal thyroid tissue specimens. Fifteen of thirty (50%) of thyroid cancer or multinodular hyperplasia samples revealed LMP1 sequences compared with 46.7% (14/30) of corresponding normal thyroid tissues. EBNA2 gene sequences were detected in 90% (27/30) of thyroid cancer or multinodular hyperplasia samples, compared with 90% (27/30) of adjacent normal thyroid tissue specimens. All samples were negative for EBER1 sequences, while HPV DNA was not detected in either nodular or normal thyroid tissue.
Conclusions
This study suggests that BKV and EBV “infection” is an early event, occurring within normal tissue. Our findings do not show a clear role for the viruses examined, instead they suggest an “endemicity” pattern rather than a causal effect.
Introduction
Thyroid cancer represents the most common endocrine malignancy (1, 2). Most cases arise from thyroid follicular epithelial cells and include well-differentiated papillary (80%) and follicular (15%) carcinoma, the latter further divided into the conventional and oncocytic (Hürthle cell) types, poorly differentiated carcinoma and anaplastic carcinoma (2–3–4). Poorly differentiated, as well as anaplastic carcinoma may either arise de novo, or from papillary and follicular thyroid carcinoma (2–3–4). Patients with a diagnosis of early-stage well-differentiated papillary or follicular carcinoma, usually have an excellent prognosis, while those with either aggressive tumors or distant metastases have a 5-year survival rate of 40% (5).
Major risk factors known to predispose to thyroid cancer acquisition include radiation exposure, either through medical treatment or radioactive fallout from nuclear accidents during childhood (6), and history of thyroid nodules or goiter, as well as a family history of thyroid cancer (6). Thyroid cancer incidence rates significantly increased for men and women of all ages, but particularly for women between 55 to 64 years of age, during the period of 1999 through 2008 (7). Increased rates referred to tumors of all stages, though these were mostly noted in localized disease (7). The reasons for this increase remain largely unknown (7). Despite the fact that enhanced medical scrutiny – through ultrasound imaging and fine needle aspiration cytological confirmation – of small lesions that may have otherwise gone undiagnosed, has been suggested to account for the rising incidence rates (8, 9), the increased incidence of both small and large tumors across sexes and multiple racial/ethnic groups suggests that it may not be solely this enhanced medical scrutiny that has driven this trend (7, 10–11–12).
Over recent decades, knowledge of the genetic alterations involved in thyroid cancer has drastically expanded (1, 2). More than 70% of papillary carcinomas are known to contain BRAF, a serine-threonine kinase of the RAF protein family, and RAS, an intracellular G protein that propagates signals from receptor tyrosine kinases and G coupled receptor-point mutations, as well as RET/PTC, the fusion between the 3′ portion of the cell membrane receptor tyrosine kinase encoded by the RET protooncogene and the 5′ portion of other unrelated genes and TRK rearrangements (1, 2, 13–14–15–16). All of the above mutually exclusive mutations may activate the mitogen-activated protein kinase (MAPK) pathway (1).
The metastatic potential of thyroid cancer has been associated with the induction of an epithelial-to-mesenchymal transition (EMT), which involves the invasive ability of epithelial cells to enter into the surrounding tissues (17). Although oncogene mutations have been linked to a contribution to the EMT, secondary factors are considered necessary (18). Recent evidence has implicated viruses in direct EMT regulation and the development of metastases (18). Epstein-Barr virus (EBV) proteins may potentially lead to the development of metastases through the regulation of the metastasis suppressor Nm23 (19) and control the expression of Twist (20). Metastatic potential is also enhanced by viral infection through the induction of angiogenesis and affects on the tumor microenvironment (21).
BK virus (BKV) has been implicated in tumorigenesis due to its behavior both in vitro and in vivo in animal models (22). The expression of BKV sequences may transform rodent cells in culture and immortalize human cells, while its inoculation in animal models may cause the development of different tumor types (22). BKV DNA sequences have been mostly reported in urinary tract malignancies, particularly prostate and bladder cancers (22). EBV has been associated with malignancies of the lymphoid and epithelial origin, including Hodgkin's lymphoma, NK/T lymphoma, posttransplantation lymphoproliferative disorders (PTLD) and nasopharyngeal carcinoma (23). Human papillomavirus (HPV) replicates in the stratified layers of the skin and mucosa, giving rise to either benign lesions such as warts, or lesions that progress to intraepithelial neoplasia and cancer, depending on the specific HPV type (24). Low-risk HPV types, such as HPV-6 and HPV-11, are associated with benign lesions, while high-risk types such as HPV-16 are associated with cancer (24).
The aim of this study was to detect sequences of 3 potentially oncogenic viruses, BKV, EBV and HPV, in the same postoperative thyroid gland specimens. BKV and EBV, but not HPV DNA was detected both in pathological and normal thyroid tissues, suggesting an “endemic” pattern of infection.
Materials and Methods
Study design and subjects
In this study, 30 patients with thyroid nodules who underwent surgery for thyroid disease within a 3-year period (from June 2003 to May 2006), at the First Department of Surgery, 251 Hellenic Air Force Hospital, Athens, Greece, were included. Both nodular and phenotypically adjacent normal thyroid tissue was obtained from each patient. All samples were immediately frozen after surgical excision and stored at -80oC until use.
Thirty patients (24 women and 6 men) were enrolled in this study. The mean age (±SD) of the patients was 48.8±13.7 years). Fourteen out of 30 patients (46.7%) had cancer lesions, while 53.3% (16/30) of the patients had multinodular hyperplasia. With regard to sex differences, the mean age of the women was 48.9±12.5 years. Half of the female patients (12/24) had thyroid cancer, while the remainder had multinodular hyperplasia. The mean age of the men was 48.0±19.3 years. Two out of 6 male patients had cancer lesions, while 4 out of 6 had multinodular hyperplasia.
The study was approved by the ethics committees of the University of Crete and the 251 Hellenic Air Force Hospital, and written informed consent was obtained from all patients.
DNA extraction
DNA extraction was performed using the Genomic DNA Purification Kit (Genomed, Lohne, Germany), according to the manufacturer's protocols. Dissected material was resuspended in 10 μL of cell lysis buffer with 30 μg of proteinase K and incubated at 56oC for 2-3 days, with the daily addition of 30 μg of proteinase K. After proteinase K inactivation and protein removal, genomic DNA was precipitated with isopropanol and washed with ethanol. DNA was subsequently dried and rehydrated with hydration buffer, and stored at -20oC until use.
Polymerase chain reaction
Gene sequences of BKV (VP1), EBV (LMP1, EBNA2 and EBER1) and HPV (GP5/6) were amplified using the primer pairs listed in Table I. The quality and integrity of the DNA in each sample was confirmed after amplification of the β-actin gene by PCR. Each PCR was set up in a total volume of 20 μL, containing 2 μL of ×10 reaction buffer, 2.5 μL of 1.5 mM MgCl2, 2.5 μL dNTPs (0.2 mM), 2 μL each of forward and reverse primers (0.5 μM), 0.2 μL of 0.625 U Taq DNA Polymerase (5 U/μL) (MBI Fermentas) and 100 ng DNA. The reactions were carried out in a MJ Research Inc. thermocycler according to the reaction programs as follows: after initial denaturation at 94ºC for 5 min, the samples were subjected to 40 cycles of amplification, comprised of denaturation at 94ºC for 30 sec, annealing (for Tm of each gene see Tab. I) for 30 sec and elongation at 72ºC for 30 sec, followed by a final step at 72oC for 5 min. Ten microliters of the PCR product was electrophoresed on a 2% agarose gel and visualized under a UV transilluminator after staining with ethidium bromide.
Primer Sequences Used For The Detection Of Bkv, Ebv, Hpv And β-Actin In Thyroid Samples

S = sense primer; A = antisense primer; BKV = BK virus; EBV = Epstein-Barr virus; HPV = human papillomavirus.
DNA sequencing
DNA sequencing was performed according to the Macrogen Advancing Through Genomics protocol, after purifying the PCR products using the Qiagen Gel Extraction kit. The primer concentration was 5 pmol/μL, while the PCR product had a concentration of 50 ng/μL and an A260/280 ratio of 1.8 according to a NanoDrop Spectrophotometer. A total volume of 20 μL of PCR product was achieved. The correct size of the PCR products prior to sequencing analysis was confirmed using a 2% agarose gel.
Statistical analysis
Data were analyzed using the SPSS 16.0 statistical package (SPSS Inc., Chicago, IL, USA). Variables were presented as counts and proportions. Fisher's exact chi-square test was performed for comparisons among different subgroups, and odd ratios (OR) with 95% confidence interval (95% CI) were presented. A p value <0.05 was considered as statistically significant.
Results
All specimens were tested by PCR for the presence of BKV, EBV and HPV DNA. Representative examples of PCR-positive samples are shown in Figure 1.
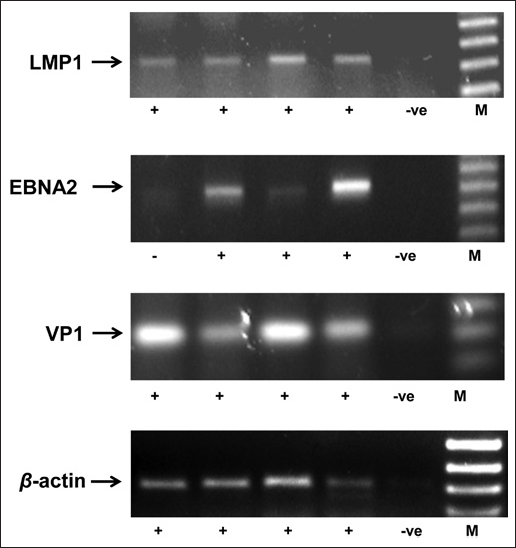
Representative examples of PCR-positive specimens for the viral sequences LMP1, EBNA2 and VP1. –ve = negative control; M = molecular weight marker.
Eighteen of the 30 (60.0%) thyroid cancer or multinodular hyperplasia lesions were positive for the VP1 sequences, compared with 43.3% (13/30) of the corresponding adjacent normal thyroid tissue specimens. Among the 14 malignant specimens, 8 specimens were positive (57.1%), compared with 42.8% (6/14) of the adjacent normal thyroid tissues. In the multinodular hyperplasia lesions alone, VP1 sequences were detected in 62.5% (10/16), compared with 43.7% (7/16) of the corresponding normal tissue.
We subsequently investigated the incidence of EBV in the same series of samples. Considering the LMP1 gene, 50.0% (15/30) of the thyroid cancer or multinodular hyperplasia samples revealed LMP1 sequences, compared with 46.7% (14/30) of the corresponding normal thyroid tissue. Eight of the 14 (57.1%) malignant specimens were found to be positive, compared with 35.7% (5/14) of the adjacent normal thyroid tissue. Multinodular hyperplasia lesions alone presented LMP1 sequences in 43.8% (7/16) of the specimens, compared with 56.2% (9/16) of the adjacent normal tissue. Of note, EBNA2 gene sequences were detected in 90.0% (27/30) of the thyroid cancer or multinodular hyperplasia samples, compared with 90.0% (27/30) of the adjacent normal thyroid tissue specimens. The vast majority of the malignant thyroid specimens (13/14, 92.9%) were EBA2-positive, compared with 85.7% (12/14) of the adjacent normal thyroid tissues. Fourteen of 16 (87.5%) of the multinodular hyperplasia lesions were PCR-positive for EBNA2 sequences, compared with 93.8% (15/16) of the corresponding normal tissue.
In the cancer or multinodular hyperplasia cases, the LMP1 sequence frequency (15/30 positive and 15/30 negative) presented a significant difference compared with the EBNA2 sequence frequency (27/30 positive and 3/30 negative; p=0.0015 by Fisher's exact test, OR=9.00, 95% CI, 2.24-36.2). In the normal thyroid tissue cases, the LMP1 sequence frequency (14/30 positive and 16/30 negative) presented a significant difference compared with the EBNA2 sequence frequency (27/30 positive and 3/30 negative; p=0.0006 by Fisher's exact test, OR=10.3, 95% CI, 2.55-41.4). The EBER1 sequence was not detected in any sample tested.
Finally, the prevalence of HPV was tested in the same series of samples using the general set of primers GP5+/GP6+; however, HPV DNA was not amplified in either nodular or normal thyroid tissues.
Discussion
In the present study, various viral sequences were detected in a series of paired nodular and adjacent normal thyroid tissues. The statistical analysis of our results showed that EBNA2 sequences were 9 times more likely to be found than LMP1 sequences in cancer or multinodular hyperplasias, while EBNA2 sequences were 10 times more likely to be found than LMP1 sequences in normal tissue.
Recently, viruses have been found to be involved in the development of thyroid disease (18, 25–26–27–28–29–30–31–32–33–34–35). Sequences of several viruses have been detected in human thyroid specimens, using molecular-based detection methods (Tab. II).
Viral Sequences Detected In Human Thyroid Samples

AITD = autoimmune disease; FA = follicular adenoma; FTC = follicular cancer; PTC = papillary cancer; FVPC = follicular variant of papillary thyroid cancer; ATC = anaplastic cancer; TMC = thyroid medullary cancer; MG = multinodular goiter; NTT = normal thyroid tissue; NTMG = nontoxic multinodular goiter; TL = thyroid lymphoma; CLTH = chronic lymphocytic thyroiditis; SCC = squamous cell cancer.
In the current study, VP1 sequences were detected in 60.0% of thyroid nodules (malignant or not), compared with 43.3% of adjacent normal thyroid tissue. More specifically, BKV was detected in almost 6 of 10 cases of malignant and multinodular hyperplasia lesions. The presence of BKV sequences in human thyroid specimens has never been studied thus far. Sequences of SV40, a monkey polyomavirus which was thought to be transmitted to humans via contaminated vaccines (36), has been detected in human thyroid tissue instead (30–31–32). In a previous study (32), both normal as well as nodular tissue was obtained from each of 99 patients with thyroid nodules who underwent thyroidectomy: papillary carcinoma was found in 8 cases and benign thyroid nodules in 91 cases of those presenting Hashimoto's thyroiditis. SV40 Tag sequences were found in 4 of 99 (4.04%) nodules, 2 of which were papillary thyroid carcinomas, while the remaining were benign nodules. No sequences were found in the 99 normal thyroid tissue or Hashimoto's thyroiditis specimens. In a separate study (30), SV40 sequences coding for the Large T-Antigen, VP1 and the regulatory region were detected in 3 cases (4.3%) of papillary carcinoma, 2 of which featured histological characteristics of Hashimoto's thyroiditis, while they were not detected in any of the 7 normal peritumoral thyroid tissue, 1 Hashimoto's thyroiditis, 5 toxic diffuse goiter, 3 medullary carcinoma and 9 breast carcinoma specimens. Furthermore, SV40 sequences were detected with a prevalence ranging from 66% in papillary carcinomas, to 100% in anaplastic carcinomas, while the corresponding frequency in adjacent normal thyroid tissue ranged from 60% to 100% (31). This prevalence differed significantly from the detection frequencies in patients with multinodular goiter and Graves’ disease, which were 10% and 20%, respectively, while SV40 sequences were detected in 25% of blood samples.
EBV sequences were detected in our study with a comparable incidence in both nodular and adjacent normal thyroid tissue. The same was also observed with LMP1 sequences which were found in 50% of thyroid nodules, while the corresponding percentage for the adjacent normal tissue was 46.7%. LMP1 sequences were positive in 57.1% of malignant specimens and 43.8% of multinodular lesion specimens. EBNA2 sequences were detected at a much higher percentage in both nodular and adjacent normal thyroid tissue. EBNA2 sequences were positive in 90% of malignant lesions and 87.5% of multinodular lesion specimens. These high percentages are in accord with the recently reported seroprevalence rate of 87% for EBV (37). The detection of EBV DNA sequences in thyroid specimens, has proven controversial (28, 33–34–35, 38–39–40). One study (34) investigated the potential role of EBV in the progression of differentiated thyroid carcinoma to the undifferentiated form, by testing 10 papillary carcinomas, 11 undifferentiated carcinomas and 1 squamous cell carcinoma with PCR, reverse-transcriptase PCR, mRNA in situ hybridization and indirect immunofluorescence staining. The examination included normal thyroid tissue adjacent to 4 papillary carcinoma specimens, while 2 specimens of Graves’ disease as well as 2 specimens of thyroid nodular hyperplasia were used as negative controls. EBV DNA in the region of Bam HIW was amplified. The expression of EBNA2 and LMP1 was more prominent in the undifferentiated carcinoma than in the papillary carcinoma samples. Another study (33) investigated the presence of the EBV genome in 30 specimens with thyroid lymphoma and 28 with chronic lymphocytic thyroiditis, using PCR. EBV DNA was detected in 2 and 1 case, respectively. Similarly (35), EBV mRNA and gene expression investigated in 23 cases of primary and 9 of secondary thyroid lymphoma, respectively, using in situ hybridization and immunohistochemistry, revealed positive results in 1 case of primary and 1 case of secondary thyroid lymphoma. In contrast to the above studies, negative results for either EBV genome presence (28, 39, 40) or gene expression (38, 39) in thyroid specimens have been reported. EBV DNA was not detected in any benign thyroid tumor specimen tested using PCR (28), while EBER-1 in situ hybridization was negative for 45 thyroid papillary carcinomas tested in another study (38). No positive sample for EBV using PCR and in situ hybridization was revealed in a Warthin-like thyroid tumor, a papillary carcinoma variant (39). Considering autoimmune thyroid disease, EBV DNA was undetectable in 50 specimens of all 50 cases of autoimmune thyroid disease, as well as in 50 cases of multinodular goiter used as controls (40).
The frequency of VP1, LMP1 and EBNA2 sequences is refereed to our study. Taking these results into consideration, we suggest that BKV and EBV “infection” is a very early event, occurring within normal tissue. As regards LMP1, we noted that malignant specimens more commonly harbor the above-mentioned sequences in comparison with normal ones. Our findings do not allow assumptions on the potential oncogenic role of the examined virus to be made, and perhaps suggest a coexistence or “endemicity” pattern, rather than a causal effect.
This study did not lack limitations, as the number of samples was limited and from one hospital source. Further studies are required to assess these observations in relation to locality features.
