Abstract
Keywords
Introduction
Postradiation osteosarcoma/chondrosarcoma is a rare delayed malignant tumor caused by radiotherapy of other tumors, and is usually found at the end of long bones of teenagers or young adults. As indicated by Murray et al (1) in 1999, the criteria for diagnosing postradiation chondrosarcoma include the following: (i) radiation must have been given previously, and the sarcoma that subsequently developed must have arisen in the area included within the 5% isodose line; (ii) there is no evidence that the sarcoma was likely to have been present before the onset of irradiation; (iii) all sarcomas must be proven histologically and must clearly be of a different pathology than that of the primary condition. The average latency period of postradiation chondrosarcoma is generally 8 to 15 years (2-3-4-5). For a majority of postradiation osteosarcoma/chondrosarcoma cases that develop in the maxillofacial region, the latency period is normally 10 years later than those of the mandible or cranial bones (6). To our knowledge, there are a few relevant reports that have indicated that maxillofacial chondrosarcoma may develop after radiotherapy for basal cell carcinoma (BCC) with a short latency of 6 months.
In this case report, we present a case of chondrosarcoma arising in the craniofacial bone, which occurred in the irradiation field with a short latency period of 6 months. At the time, the patient was a 35-year-old man who had received radiotherapy at a dose of 70 Gy for 8 weeks. Following radiotherapy, a rapidly growing mass at the upper right alveolar was found, where the radiation had been given, and this was diagnosed as chondrosarcoma. Informed consent was obtained from the patient, explicitly giving permission for use of clinical photographs and clinical information.
Clinical Summary
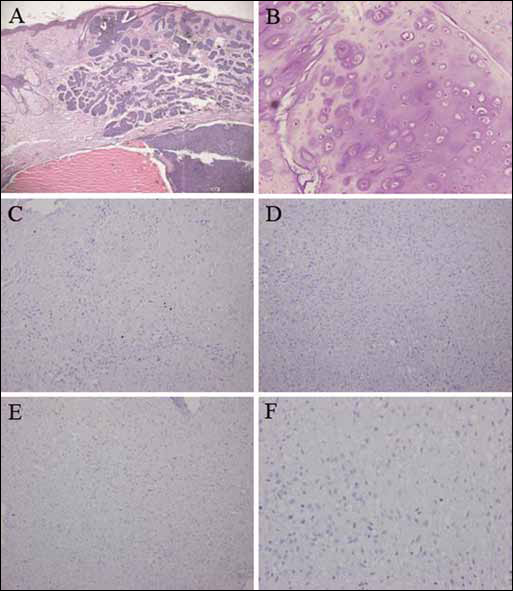
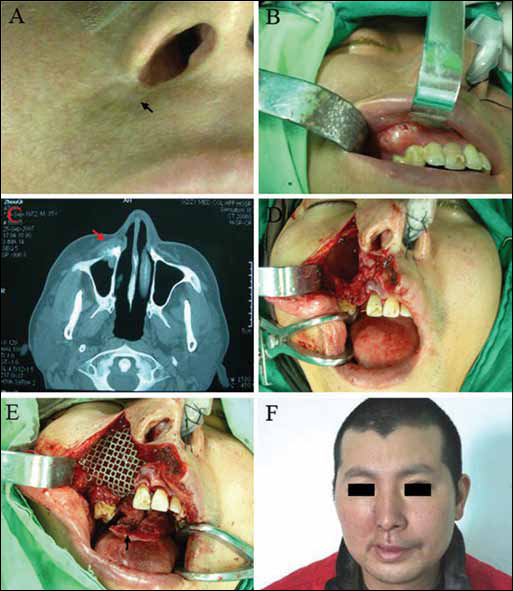
A 35-year-old man was found to have repeated skin ulceration without healing, which was located next to the right nasal wing, in September 2006. In June 2007, the skin ulcer was diagnosed as BCC of the lateral right nasal wing via pathology assessment in West China College of Stomatology (Fig. 1A). As the patient refused surgery, he was sent back to the local hospital for radiotherapy. After treatment with radiotherapy at a dose of 70 Gy for 8 weeks, the patient achieved a clinical cure. Six months later, however, the patient discovered a hard, rapidly growing mass in the upper right alveolar ridge of the gums, where radiation had been given. In March 2008, the mass was confirmed to be chondrosarcoma, by biopsy (Fig. 1B-F). Scarring tissue measuring 1.5×1.5 cm was located at the right nasal wing as shown in Figure 2A. The bone measured to 2.0×1.5 cm (Fig. 2B). The mass adhered tightly with the deep tissue of the radiotherapy scar, but did not affect the function of the right nasal airway. Computed tomography (CT) examinations showed that the bone destruction involved the right maxillary bone, but mainly the anterior wall of right maxillary sinus (Fig. 2C). According to the localization of the lesion, surgery of right subtotal maxillectomy combined with postradiation scar removal was planned for the patient (Fig. 2D). Intraoperative pathological examination confirmed the absence of tumor cells in all surgical margins. Maxillary reconstruction was reconstructed by prefabricated titanium mesh, and intraoral soft tissue reconstruction was reconstructed by the transposition of palate flap (Fig. 2E). A postsurgery image is show in Figure 2F. Following surgery, the patient received postoperative radiotherapy (35 Gy) and chemotherapy (180 mg of cisplatin, and 70 mg of doxorubicin). Follow-up at 20 months ruled out recurrence and metastasis.
Discussion
The incidence of postradiation osteosarcoma/chondrosarcoma is approximately 3.4%-5.5% of all osteosarcoma/chondrosarcomas (4). It is a rare complication of radiotherapy that occurs within a previously irradiated field after several years of latency. For this patient, it occurred without evidence that the sarcoma was likely to have been present before the onset of irradiation. The results of the biopsy revealed a different pathology than that of the primary BCC. These points suggest the presence of postradiation osteosarcoma/chondrosarcoma according to the criteria of Murray et al (1). There are few if any reports to indicate that maxillofacial chondrosarcoma is caused by radiation treatment of BCC. The incidence of metastasis in BCC is around 0.03% (7), and BCC-related lesions are normally induced by distant organ metastases such as from the brain or the lungs (7-8-9). Generally, the latency of postradiation chondrosarcoma is around 8 to 15 years. Matsuyama et al (10) report a case of postradiation osteosarcoma with a short latency period of 3 years. In the case of this patient, chondrosarcoma was detected with a latency period of 6 months after radiotherapy.
We hypothesize that the short period of latency of chondrosarcoma of higher malignancy that developed in the primary radiotherapy region is due to the short-term high-dose radiotherapy for the treatment of BCC. The high dosage of radiotherapy given to the patient may play a key role in this case of postradiation chondrosarcoma. This hypothesis is supported by the fact that in Hedgehog (Hh) signaling pathway, Gli family of transcription factors 2 (Gli2) expression could promote the BCC formation while inactivation of Gli2 could delay BCC tumor cell in growing and being cleared by apoptosis. However, some tumor cells were living in a Gil2-inactivated condition, and there were remaining stem cell characteristics of self-renewal and differentiation (11, 12). Gil2 expression in the Hh signaling pathway might be silent because of the radioactivity during treatment. Therefore, a large number of tumor cells might stop growing and lead to apoptosis. At the same time, tumor stem cells with multidirectional differentiation may transform to more highly malignant tumor cells, which have a stronger ability of invasion and migration. Affected by radiotherapy at a high dose, these stem cells could transform to mesenchymal tumor cells in the role of different factors (13-14-15). It has been well accepted that nutrition support (NS), either as parenteral nutrition (PN) or enteral nutrition (EN) stimulates tumor growth and causes cancer progression. Tumor cells have a requirement for a persistent supply of new blood vessels to nourish their growth and to facilitate metastasis (16). Deficiency of blood supply would result in inhibition of tumor cell proliferation or induce tumor cell necrosis (2). However, a small fraction of cells within a tumor possess a self-renewal capacity and can survive without sufficient blood supply (14). It is possible that some stem-like cells survived in the postradiation region, which consists of scar contractures instead of blood vessels. These cells would have the ability to promote an epithelial-to-mesenchymal transition, and the tumors could acquire a greater ability to invade and migrate (17). Additionally, the tumor can penetrate from the basement membrane of epithelium, and transfer to other adjacent areas under the chemotaxis of relevant factors (18). This indicates that although BCC is a low-grade malignant skin tumor, it could develop into other high-grade malignant tumors, if treated incorrectly. Although it is widely recognized that BCC can be treated with radiotherapy, it is unknown whether radiotherapy can completely eradicate BCC. Because BCC is a slow-growing and invasive cancer, surgery is still the best option for treatment of BCC. For some patients with a large cancerous lesion(s), a treatment plan may include surgery and radiotherapy. We did not find any other BCC patients who had received radiotherapy and also had neogenesis tumors, until the end of our data collection. We hypothesize that dosage of radiation and time of radiation therapy may have controlled the opening/closing of the Hh-signaling pathway in this case. This gives a valuable clinical basis for future fundamental research.
