Abstract
Objective
Material and methods
Results
Conclusion
Introduction
Hepatocellular carcinoma (HCC) is the sixth most common cancer worldwide, and the third most frequent cause of cancer death (1, 2). HCC is often associated with underlying liver cirrhosis, chronic hepatitis B virus (HBV) or hepatitis C virus (HCV) infection, autoimmune hepatitis, alcoholic liver disease, nonalcoholic fatty liver disease, diabetes, obesity and food intake of aflatoxin contamination (1-2-3). Despite the fact that the short-term survival rate of patients with HCC has improved with advances in surgical techniques and perioperative management, the mortality rate of HCC is still high because of delayed diagnosis, lack of effective therapy and frequent recurrence and metastasis. Early detection is of utmost importance (1). Tumor marker detection is an important facet of cancer diagnosis. In particular, α-fetoprotein (AFP) is a critical marker for the diagnosis and development of HCC. However, 30%-40% of HCC patients are negative for serum AFP. Therefore, the search for a more specific and sensitive tumor marker is an important aspect of current research on HCC (4).
Dickkopf-1 (DKK1) is a 266–amino acid (35 kDa) secreted glycoprotein that has been shown to play a crucial role in limb and head development in Xenopus embryos, and is known as a secretory antagonist of the canonical Wnt signaling pathway (5, 6). DKK1 is hardly expressed at all in normal human adult tissues, and overexpression of DKK1 has been described in hepatoblastoma, multiple myeloma, prostate cancer, Wilms’ tumor and kidney, lung and esophageal cancers (6-7-8). DKK1 can be secreted from the tumor into blood, and some recent reports have indicated that serum DKK1 may be a useful cancer biomarker, and that it has been found to be increased in various cancers including HCC, multiple myeloma, osteosarcoma breast, and lung, ovarian and prostate cancers (8-9-10-11). In 2012, a large multicenter study by Shen et al indicated serum DKK1 could complement measurement of AFP in the diagnosis of HCC and improve identification of patients with AFP-negative HCC (11).
Because there have been no previous such meta-analyses, the aim of the current study was to perform a meta-analysis on eligible clinical studies to assess the diagnostic accuracy of serum DKK1 for diagnosing HCC.
Methods
Data Sources and Searches
We included cohort studies addressing the diagnostic accuracy of serum DKK1 for HCC. We developed a search strategy using the following text words: (i) Dickkopf-1 or DKK1, (ii) hepatocellular carcinoma or HCC or liver cancer or hepatoma or hepatocarcinoma. We searched the following databases: Embase, PubMed, Wanfang, Weipu Periodical Database and the Chinese National Knowledge Infrastructure (CNKI). The “References” sections of the studies included were also searched manually to identify additional eligible studies, but unpublished data were difficult to obtain. The articles were restricted to those on human subjects, written in either English or Chinese, and published online before July 10, 2013.
Inclusion and Exclusion Criteria
Inclusion criteria for this meta-analysis were (i) full-text publications; (ii) studies about the diagnostic accuracy of serum DKK1 for HCC; (iii) studies with raw data such that true-positive, false-positive, false-negative and true-negative results of the diagnostic tests for the detection of HCC were reported or could be calculated; (iv) studies with reference standards for the diagnosis of HCC. HCC was defined on the basis of ultrasound, radiological examination (CT/MRI) and biochemistry (AFP serology and liver function enzymes), with postresection histological confirmation (12). Tumors with Barcelona Clinic Liver Cancer (BCLC) stage 0+A were defined as early-stage HCC (13); and (v) studies with more than 20 patients.
Exclusion criteria were as follows: (i) duplicate publication; (ii) studies that were editorials, letters, case series or case reports, or those that were published in articles that failed to meet checklist criteria.
Data Abstraction
Data from the studies were independently abstracted by 2 reviewers (J. Z. and Y. Z.) using a standardized protocol. Any disagreements were resolved through consensus, and consultation with the senior investigator (Q. Y.). The characteristics of the articles were abstracted as follows: (i) basic characteristics of studies including the first author, year of the publication, country or region of origin, mean/median age of patients; (ii) the methods used in the studies, including study design, diagnosis criteria for HCC, source of control and methods of DKK1 detection; and (iii) outcomes, including the number of patients, true- or false-positive and true- or false-negative results.
Quality Assessment
We assessed the quality of the studies included, using the recommended the modified Quality Assessment of Diagnostic Accuracy Studies (QUADAS-2) (14) and the Standards for Reporting of Diagnostic Accuracy (STARD) checklists (15). Each item was scored as yes, no or unclear if there was not sufficient information to make an accurate judgment. Some articles that failed to meet checklist criteria were excluded from the analysis (Fig. 1). Figure 1 presents the flow chart documenting how the reviewers abstracted the characteristics for study objectives.
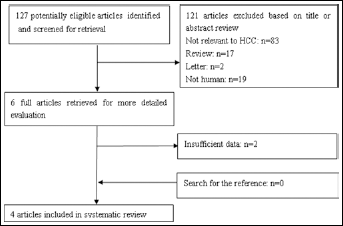
Flow chart of studies identified and evaluated during the selection process. HCC = hepatocellular carcinoma.
Data Synthesis and Analysis
Two of the authors (J. Z and Y. Z.) extracted the data from the studies included. The variables extracted for data analysis were entered on an electronic data sheet. We used a bivariate regression approach to calculate the pooled sensitivity (SEN) and specificity (SPE) and pooled positive and negative likelihood ratios (pooled PLR and pooled NLR, respectively), and we constructed summary a receiver operating characteristic (sROC) curve to summarize the main study results (16). We also calculated the area under the sROC curve and Q point value (where sensitivity = specificity) on the sROC curve. The values of pooled PLR greater than 10 and values of pooled NLR less than 0.1 were noted as providing convincing diagnostic evidence, while those values greater than 5 and less than 0.2, respectively, provided strong diagnostic evidence (17, 18). As a single indicator measure of the accuracy of a diagnostic test that comprises a combination of SEN and SPE (19), the diagnostic odds ratio (DOR) describes the odds of positive test results in patients with the disease compared with the odds of positive results in those without disease. The between-studies heterogeneity was evaluated by the Q test and I-square (I2) test for the pooled DOR (20). The DerSimonian-Laird method was used for pooled analyses if the value for heterogeneity was more than 50% (21, 22). Furthermore, to explore the sources of between-study heterogeneity, subgroup analyses were performed. Publication bias was inspected by Deek's funnel plot and associated regression test of asymmetry (23). A non-zero slope coefficient suggested a significant small study bias (p value <0.10) (23). All analyses were conducted with STATA software version 12.0 (Stata Corp, College Station, TX, USA) with the MIDAS modules and Meta-Disc 1.4 (XI Cochrane Colloquium, Barcelona, Spain). All statistical tests were 2-tailed with p values <0.05 taken as significant.
Results
Basic Characteristics of Studies
Figure 1 presents the flow chart to describe the process of selecting the studies for our meta-analysis. The database searches elicited 127 abstracts; from these, 121 articles were excluded based on title or abstract review, 2 articles were excluded based on full-text screening. According to the inclusion and exclusion criteria, 4 articles (6 cohort studies) (11, 24-25-26) were ultimately included in this meta-analysis, which included a total of 2,678 participants. The main characteristics of the studies included in the meta-analysis are shown in Tables I and II. The included cohort studies were performed in China and Hong Kong. The cutoff values for DKK1 positivity varied from 1.209 to 4.2 ng/mL.
CHARACTERISTICS OF STUDIES INCLUDED IN THE META-ANALYSIS
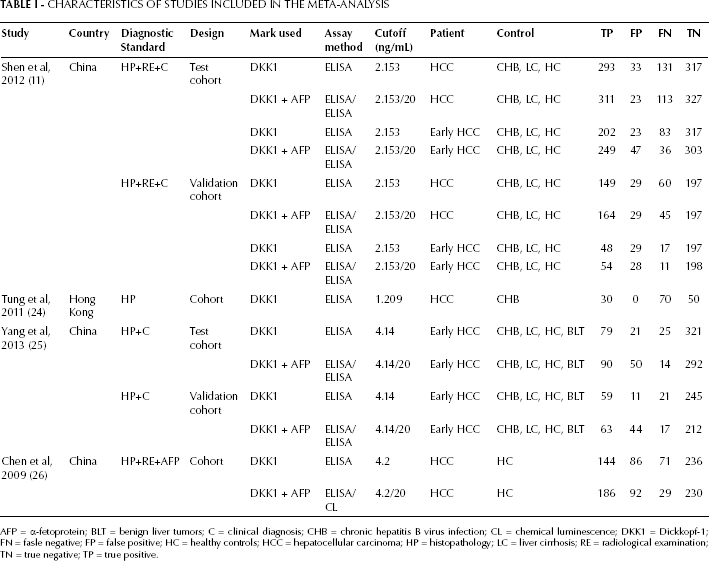
AFP = α-fetoprotein; BLT = benign liver tumors; C = clinical diagnosis; CHB = chronic hepatitis B virus infection; CL = chemical luminescence; DKK1 = Dickkopf-1; FN = fasle negative; FP = false positive; HC = healthy controls; HCC = hepatocellular carcinoma; HP = histopathology; LC = liver cirrhosis; RE = radiological examination; TN = true negative; TP = true positive.
SUMMARY OF THE CLINICOPATHOLOGICAL CHARACTERISTICS OF THE HCC PATIENTS
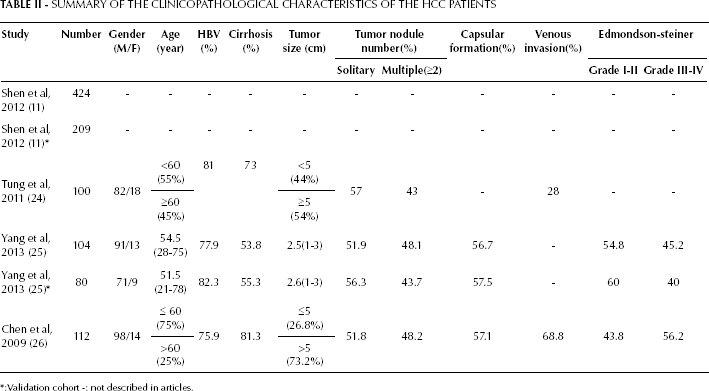
:Validation cohort -: not described in articles.
Assessment of Study Quality
The STARD scores of each study were 17, 14, 20 and 18. Quality assessment was shown with a bar graph according to the QUADAS-2 tool (Fig. 2).
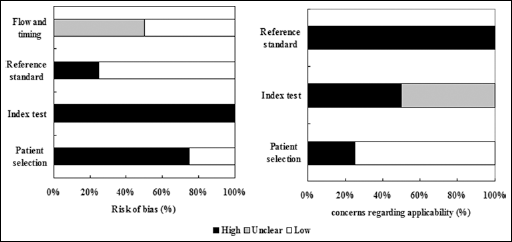
Overall quality assessment of studies included (Quality Assessment of Diagnostic Accuracy Studies [QUADAS-2] tool).
Diagnostic Accuracy of Serum DKK1 Detection
Four articles (6 studies) (11, 24-25-26) provided DKK1 diagnostic data. The pooled SEN and SPE of DKK1 for the diagnosis of HCC were 0.65 (95% confidence interval 95% CI], 0.52-0.76) and 0.94 (95% CI, 0.82-0.98), respectively (Fig. 3; Tab. III), with significant heterogeneity (p<0.01, Q=71.06, I2=92.96%; and p<0.01, Q=99.57, I2=94.98%) found among the studies. Additionally, the pooled PLR was 10.1 (95% CI, 3.68-27.74), the NLR was 0.38 (95% CI, 0.28-0.51), and the DOR was 26.90 (95% CI, 10.45-69.19) (Tab. III). The sROC curve was for an overall summary of the studies included, which illustrates the relationship between SEN and SPE. As shown in Figure 4, the area under the sROC curve (AUC) was 0.84 (95% CI, 0.81-0.87), indicating a high diagnostic accuracy.
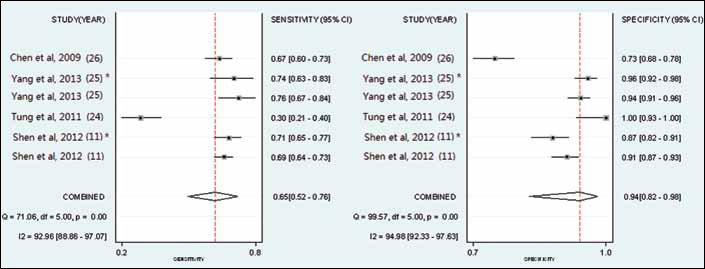
A forest plot showing study-specific (right-axis) and mean sensitivity and specificity for diagnostic accuracy of DKK1 for hepatocellular carcinoma (HCC). * : validation cohort.
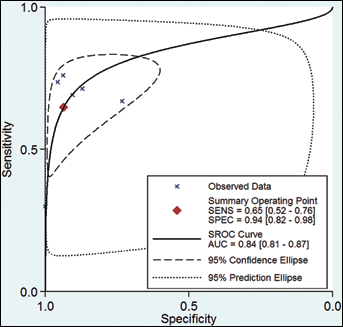
Summary receiver operating characteristic (sROC) curves for individual studies for the diagnostic accuracy of DKK1 for hepatocellular carcinoma (HCC).
POOLED RESULTS OF THE META-ANALYSIS OF DKK1 AND DDK1 PLUS AFP TESTS FOR HCC DIAGNOSIS
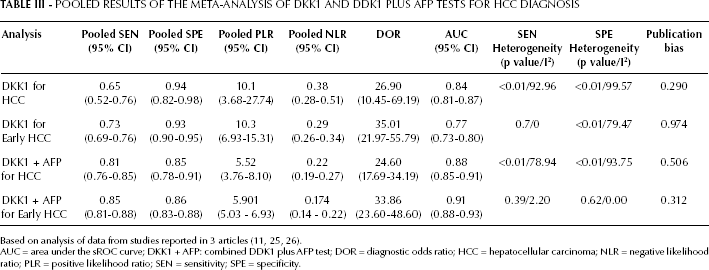
AUC = area under the sROC curve; DKK1 + AFP: combined DDK1 plus AFP test; DOR = diagnostic odds ratio; HCC = hepatocellular carcinoma; NLR = negative likelihood ratio; PLR = positive likelihood ratio; SEN = sensitivity; SPE = specificity.
The results of a threshold effect analysis showed that the Spearman correlation coefficient was -0.029, and the p value was 0.957. These values indicated that there was no threshold effect. To explore other potential heterogeneities, a subgroup meta-analysis was performed. As shown in a Fagan's nomogram, with a pretest probability of HCC of 20% in this meta-analysis, the results indicated that the DKK1 for detection HCC increased the posttest probability to 72% when the results of DKK1 were positive, and reduced the posttest probability to 9% when the results were negative. Deek's funnel plot asymmetry test showed the p value was 0.29 for the slope coefficient, and there was no publication bias.
Data from the subgroup analysis of the diagnostic accuracy of serum DKK1 for patients with early HCC are showed in Table III. Two articles (4 studies) (11, 25) provided early DKK1 diagnostic data. The pooled SEN and SPE of DKK1 for the diagnosis of early HCC were 0.73 (95% CI, 0.69-0.76) and 0.93 (95% CI, 0.90-0.95), respectively, with SEN showing no significant heterogeneity (p=0.77, Q=1.13, I2=0.00%), and SPE significant heterogeneity (p<0.01, Q=14.61, I2=79.47%).
Diagnostic Accuracy of Combined Serum DKK1 Plus AFP
Three articles (5 studies) (11, 25, 26) provided combined DKK1 plus AFP diagnostic data. The pooled SEN and SPE of combined DKK1 plus AFP tests for the diagnosis of HCC were 0.81 (95% CI, 0.76-0.85) and 0.85 (95% CI, 0.78-0.91), respectively (Fig. 5; Tab. III), with significant heterogeneity (p<0.01, Q=19, I2=78.94%; and p<0.01, Q=63.98, I2=93.75%) among the studies. Additionally, the pooled PLR was 5.52 (95% CI, 3.76-8.10), the NLR was 0.22 (95% CI, 0.19-0.27), and the DOR was 24.60 (95% CI, 17.69-34.19) (Tab. III). The AUC was 0.88 (95% CI, 0.85-0.91), indicating a high diagnostic accuracy.
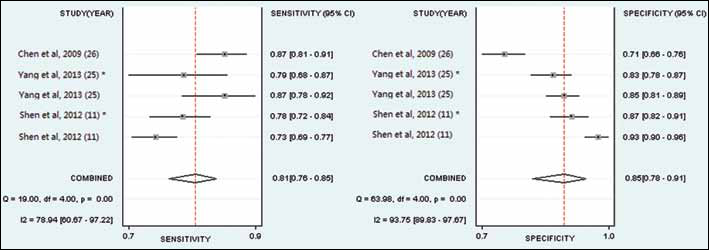
A forest plot showing study-specific (right-axis) and mean sensitivity and specificity for diagnostic accuracy of the combined DKK1 plus AFP test for hepatocellular carcinoma (HCC). * : validation cohort.
The results of the threshold effect analysis showed that the Spearman correlation coefficient was 0.7, and the p value was 0.188, indicating no threshold effect. To explore other potential heterogeneities, a subgroup meta-analysis was performed. The results of a Fagan's nomogram of DKK1 plus AFP test indicated that the pretest probability of HCC was 20%. The combination of DKK1 plus AFP tests for the detection of HCC increased the posttest probability to 58% when the results for DKK1 were positive, and decreased the posttest probability to 5% when the results were negative. There was no publication bias (p=0.506).
Subgroup analysis of the diagnostic accuracy of combined serum DKK1 plus AFP tests for patients with early HCC is shown in Table III. Two articles (4 studies) (11, 25) provided early DKK1 plus AFP diagnostic data. The pooled SEN and SPE of DKK1 for the diagnosis of early HCC were 0.85 (95% CI, 0.81-0.88) and 0.86 (95% CI, 0.83-0.88), respectively, with SEN showing no significant heterogeneity (p=0.39, Q=4.09, I2=2.20%) and SPE no significant heterogeneity (p=0.62, Q=2.64, I2=0.00%).
Discussion
Both AFP measurements and abdominal ultrasound have been widely used for HCC diagnosis and surveillance (4, 7). However, AFP levels may be normal in patients with HCC, and the accuracy of ultrasound can be limited by the ability of the operator to differentiate HCC from non-neoplastic lesions (7). Their poor sensitivity and specificity have prevented widespread implementation of HCC surveillance.
Tao et al (27) have shown that elevated DKK1 levels in human HCC tissues were significantly correlated with the presence of tumor nodules, Edmondson-Steiner grade and the presence of vascular invasion, especially in HCC with vascular invasion, and they were associated with a high metastatic potential of tumors (28). The clinicopathological characteristics of the HCC patients are shown in Table II. Tung et al (24) found that serum DKK1 was significantly correlated with the presence of venous invasion, and Chen et al (26) found that serum DKK1 was significantly correlated with the presence of tumor nodules, Edmondson-Steiner grade and the presence of vascular invasion. The clinicopathological characteristics of the patients included in the studies analyzed (Tab. II) also indicated that serum DKK1 positivity was significantly correlated with venous invasion and suggests its involvement in the invasion and metastasis of HCC.
In the current meta-analysis, the pooled SEN, SPE, PLR, NLR, DOR and AUC of serum DKK1 for diagnosing HCC (Tab. III) revealed that DKK1 had a good diagnostic accuracy for HCC. The DOR represented a single indicator of test accuracy (19), and the pooled DOR further confirmed serum DKK1 to be a useful marker for diagnosing HCC. Compared with combined DKK1 plus AFP tests, DKK1 had a lower pooled SEN, but a higher pooled SPE, and a similar DOR and AUC, which demonstrated that both DKK1 alone and combined DKK1 plus AFP tests were similar in terms of accuracy for diagnosing HCC.
The p and I2 values of the pooled SEN and SPE for DKK1 alone and for combined DKK1 plus AFP tests (Tab. III) indicated there were marked heterogeneities between the studies. The results of threshold effect analysis for DKK1 and combined DKK1 plus AFP tests had p values of 0.957 and 0.188, respectively, indicating no threshold effects. Therefore, we undertook a subgroup meta-analysis to explore the possible reasons for the heterogeneities. The subgroup analysis of DKK1 for early HCC (Tab. III) showed that the p value of the pooled SEN was 0.7, and that of the pooled SPE was <0.01. This indicated that the research object group (early HCC) was the reason for the heterogeneity of pooled SEN for DKK1. However, it had no effect on the pooled SPE. The other subgroup analysis of combined DKK1 plus AFP tests for early HCC (Tab. III) showed that the p values of the pooled SEN and SPE were all >0.05, and there were no heterogeneities between the subgroup studies. This indicated that the research object group (early HCC) was the reason for the heterogeneity of pooled SEN and SPE.
There were several limitations in the 4 articles (6 cohort studies) (11, 24-25-26) that we included in the current meta-analysis. Only in the study by Tung et al (24) was the diagnosis of HCC based completely on histopathology of liver biopsy samples. Only the study by Shen et al (11) was a large-scale study. Finally, 4 of the articles were limited to mainland and Hong Kong Chinese. Based on this information, we recommend that multicenter and multiethnic studies be conducted on the diagnostic accuracy of DKK1 for HCC in the future. In addition, there were several limitations in our current meta-analysis. First, the diagnosis of HCC obtained in our study was not completely based on histopathology of liver biopsy samples. Although histopathology remains the gold standard for HCC diagnosis and evaluation, it is difficult to conduct in large populations, so that ultrasonography and radiological examination remain the most common approach to diagnosing HCC in clinical practice (12). Second, the different research subjects and different cutoff values for DKK1 that were used in the studies included made it difficult to determine the optimized cutoff value. Third, the p values of the pooled SEN and SPE for DKK1 alone and for combined DKK1 plus AFP tests indicated there were marked heterogeneities between the studies. To investigate the sources of heterogeneity, subgroup meta-analyses were performed. However, the heterogeneity of pooled SPE (p<0.01) for DKK1 for diagnosing early HCC still existed. Fourth, we only searched studies published in English and Chinese. There may have been some publications in other languages which were not included in our meta-analysis, and for which unpublished data could not be obtained (27). Finally, nearly all of the patients and controls were Chinese, which may limit the generalization of the findings to other populations. Therefore, we recommend that multicenter and multiethnic studies on the diagnostic accuracy of DKK1 for HCC be conducted in the future.
In summary, despite the limitations mentioned above, our meta-analysis suggested that serum DKK1 positivity is a useful marker for diagnosing HCC. The pooled SEN and SPE of both DKK1 alone and of combined DKK1 plus AFP tests were all above 0.60. Compared with DKK1 alone, DKK1 plus AFP tests had a higher pooled SEN and slightly lower pooled SPE, indicating that both DKK1 and combined DKK1 plus AFP tests had high diagnostic SEN and SPE for diagnosing HCC.
