Abstract
Background
Aims
Methods
Results
Conclusion
Introduction
Blood alpha-fetoprotein (AFP) was initially reported as a cancer-related oncofetal antigen (fetal albumin) in several rare tumors (1, 2), but it has then become a useful tool in the management of patients with hepatocellular carcinoma (HCC). AFP is widely used to evaluate prognosis, as well as failure or response to therapy (3, 4), and is also used in several classification schemes, such as CLIP and bm-JIS (5). Although AFP is commonly used together with ultrasound in the screening of patients at risk for developing HCC, there is no general agreement on its usefulness as a surveillance tool (6-7-8). AFP is not very effective as a screening tool for small size HCCs (9), as it is negative in a significant proportion of these tumors. However, large HCCs can also associate with low AFP values. We therefore examined the patient and tumor characteristics of subjects with HCC having either high or low blood AFP levels, as to try to understand the possible differences in their biology.
Methods
Data collection. We retrospectively analyzed prospectively-collected data in the Italian Liver Cancer (ITA.LI.CA) study group database of 2,440 HCC patients accrued until 2008 at 11 centers (9). Patients had full baseline parameter data, including the following: CT scan information on maximum tumor diameter (MTD); number of tumor nodules and presence of portal vein thrombosis (PVT); demographics (gender, age, alcohol history, presence of hepatitis B or C); blood counts (hemoglobin, white cells, platelets, prothrombin time); blood AFP and routine liver function tests (total bilirubin, AST, ALKP, GGTP, albumin); and survival information. The ITA.LI.CA database management conforms to the Italian legislation on privacy and this study conforms to the ethical guidelines of the Declaration of Helsinki. Approval for the study on de-identified patients was obtained by the Institutional Review Board of the participating centers.
Statistical Methodology
As indices of centrality and dispersion of the distributions we used means and standard deviations (M±SD) for continuous variables, and relative frequencies for categorical variables. To test the associations between groups we used the chi-square test for categorical variables, and the Kruskal-Wallis rank test and Wilcoxon rank-sum test (Mann-Whitney test) for continuous variables.
The Z test for proportions was used for comparison between 2 categorical variables.
The Pearson's correlation was estimated for selected tumor parameters in the total cohort. A multiple logistic regression model was used to evaluate the association between MTD and selected parameters. For the GGTP scatter plots and the AFP and GGTP dichotomization studies, the median value of the parameter for the total cohort was used in each case. All Cox proportional hazard models were corrected for gender, age, and alcohol history. When testing the hypothesis of a significant association, for all the analyses we considered the two-tailed p-value <0.05 as being statistically significant. All statistical analyses were performed using STATA 10.0 Statistical Software (StataCorp, College Station, TX, USA).
Results
AFP Groups in the Total Cohort
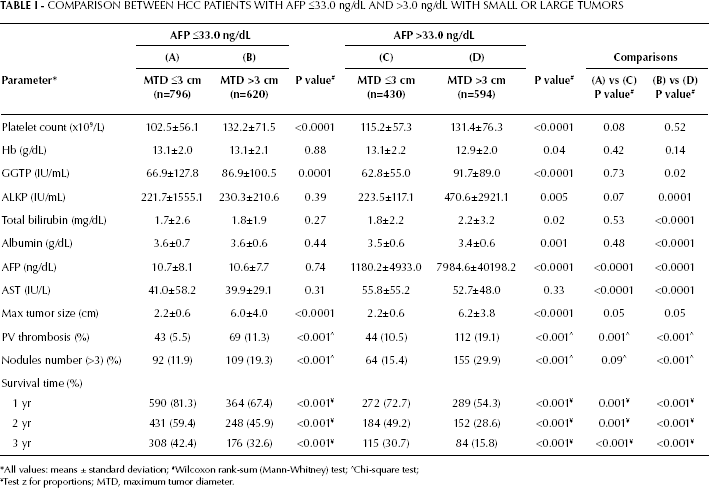
The characteristics of the whole cohort of HCC patients is shown in Table I, in which the patients are divided according to the median blood AFP value of 33 ng/mL. Patients of each AFP group were then subdivided according to smaller or larger HCCs (MTD <3 cm or >3 cm in diameter). More patients had low AFP levels: 1,416 patients (796+620), 58% of the total cohort, while 1,024 patients had high AFP levels (430+594), 42% of the total cohort. There were nearly twice as many patients with small HCCs that had low AFP levels than with high AFP levels (790 vs 430; 64% vs 35%). By contrast, for large tumors, the ratio of low-to-high AFP patients was 1:1 (620:594).
COMPARISON BETWEEN HCC PATIENTS WITH AFP ≤33.0 ng/dL AND >3.0 ng/dL WITH SMALL OR LARGE TUMORS
All values: means ± standard deviation;
Wilcoxon rank-sum (Mann-Whitney) test;
Chi-square test;
Test z for proportions; MTD, maximum tumor diameter.
The patients with low AFP were then examined separately (Tab. I, columns A and B). Platelet counts were significantly lower for smaller than for large tumors, as previously described (11). Although the bilirubin values were similar in the 2 subgroups, the GGTP levels were significantly higher in patients with large tumors than in those with small tumors. The presence of PVT and tumor multifocality were more frequent in patients with large tumors, as also noted by others, and survival was significantly worse for patients with large tumors.
Similar results were found within the group of patients with high AFP when comparing the 2 subgroups (Tab. I, columns C and D), with the exception that, in addition to GGTP and AFP, also alkaline phosphatase (ALKP) was significantly higher in patients with large tumors; similarly, the subgroup with a large tumor size also had significantly higher bilirubin levels, possibly due to parenchymal destruction by the enlarging tumor. Also in this case, patients with large tumors had a significantly worse survival.
We then compared patients with small tumors with either high or low AFP levels (Tab. I, columns A vs C). Most blood parameters’ comparisons were no longer significantly different, except for the significant difference in AST levels (and AFP, by definition). Bilirubin levels were notably similar between the 2 subgroups with small size tumors. However, there was a significantly higher incidence of PVT in patients with small tumors who had high AFP levels than in those with low AFP levels. Survival for patients with small tumors who had high AFP levels was significantly worse than those who had low AFP levels (72.7% vs 81.3% at 1 year, and 30.7% vs 42.4% at 3 years).
Similar findings were obtained among HCC patients with large tumors from the comparison between those with high vs low AFP levels (Tab. I, column B vs D). Unlike the results obtained among patients with small tumors, GGTP, ALKP, AST, and bilirubin levels were all significantly different among patients with large tumors, being higher in the high-AFP subgroup. As in the 2 subgroups with small tumors, survival was significantly different between the subgroups of patients with large-size tumors, being worse for high-AFP patients (54.3% vs 67.4% at 1 year and 15.8% vs 32.6% at 3 years).
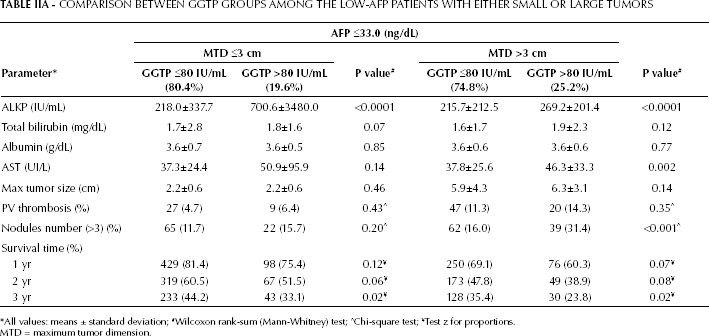
GGTP Subgroups among Low-AFP Patients
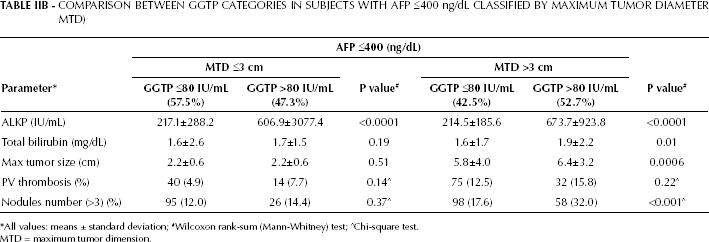
GGTP levels were significantly different between patients with small and large tumors (Tab. I), in case of both low and high AFP levels. Patients within the low-AFP cohort were then analyzed according to the median GGTP levels (80 IU/mL was the GGTP median value for the total cohort) (Tab. IIA). A total of 20% of patients with small tumors and 25% of patients with large tumors had elevated GGTP levels. Both the small and large-size tumor groups had significantly higher ALKP levels within the high-GGTP subgroups, but no significant differences were recorded for MTD or PVT. However, for patients with large tumors, higher GGTP values were significantly associated with increased multifocality. Bilirubin values were higher in the high-GGTP than in the low-GGTP subgroups, without statistical significance. However, survival at 3 years was significantly shorter for high-GGTP patients with either small or large tumors, compared with low-GGTP patients. In this analysis we used the median AFP value of the total cohort for dichotomization; AFP has been found to be elevated in patients with hepatitis and several groups use an AFP cutoff level of 300 ng/mL or 400 ng/mL to distinguish HCC for non-HCC conditions. Table IIB shows a GGTP dichotomization using an AFP cutoff level of 400 ng/mL; the results are similar to those reported in Tab. IIA. However, patients with large tumors had higher total bilirubin levels than patients with small tumors (Tables I, IIA, and IIB).
COMPARISON BETWEEN GGTP GROUPS AMONG THE LOW-AFP PATIENTS WITH EITHER SMALL OR LARGE TUMORS
All values: means ± standard deviation;
Wilcoxon rank-sum (Mann-Whitney) test;
Chi-square test;
Test z for proportions.
MTD = maximum tumor dimension.
COMPARISON BETWEEN GGTP CATEGORIES IN SUBJECTS WITH AFP ≤400 ng/dL CLASSIFIED BY MAXIMUM TUMOR DIAMETER MTD)
All values: means ± standard deviation;
Wilcoxon rank-sum (Mann-Whitney) test;
Chi-square test.
MTD = maximum tumor dimension.
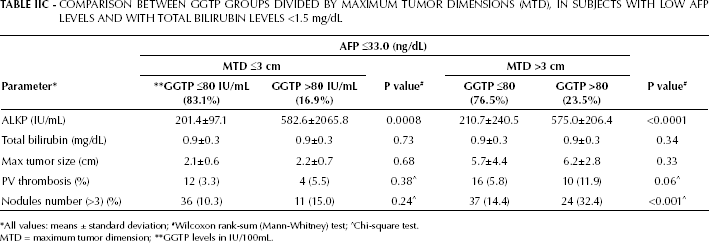
Both bilirubin and GGTP levels are a measure of liver function tests, and elevated bilirubin levels are often associated with elevated GGTP levels. We therefore reanalyzed the data reported in Table IIA after excluding all patients with elevated bilirubin levels. We then found that in these patients, with either small or large tumors, the normal bilirubin levels and elevated GGTP levels were significantly associated with elevated ALKP levels (Tab. IIC). For large-tumor patients, the elevated GGTP levels were associated with a significantly higher incidence of PVT and tumor multifocality.
COMPARISON BETWEEN GGTP GROUPS DIVIDED BY MAXIMUM TUMOR DIMENSIONS (MTD), IN SUBJECTS WITH LOW AFP LEVELS AND WITH TOTAL BILIRUBIN LEVELS <1.5 mg/dL
All values: means ± standard deviation;
Wilcoxon rank-sum (Mann-Whitney) test;
Chi-square test.
MTD = maximum tumor dimension;
GGTP levels in IU/100mL.
Given the significantly higher GGTP levels associated with large HCCs (Tab. I), we performed a Pearson correlation analysis between the GGTP levels and MTD (scatter plots in Fig. 1). The correlation was significant in the total cohort, as well as for the low-AFP patients. However, the correlation was stronger for the total cohort. A multiple logistic regression analysis of MTD >3.0 cm with platelet counts or GGTP levels (significant parameters for both low and high-AFP patients; Tab. I) showed an increased Odds Ratio (OR) for both high platelets and elevated GGTP levels in both low and high-AFP groups; however, the highest OR was recorded in the elevated GGTP group with high AFP levels (Tab. III).
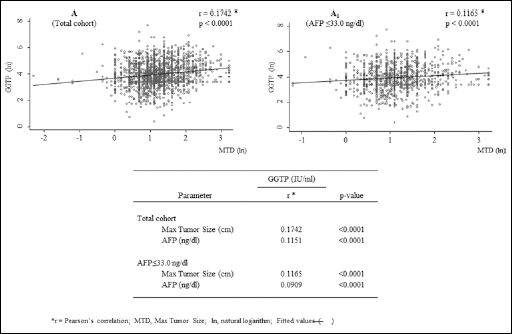
Scatterplots showing the maximum tumor diameter (MTD) and blood GGTP levels in (A) the total HCC patients’ cohort and (A1) patients with AFP levels <33.0 ng/mL. All data were transformed into natural logarithms. *Pearson's correlation.
MULTIPLE LOGISTIC REGRESSION ANALYSIS OF MTD >3.0 cm WITH PLATELET COUNT >105x109/L (MODEL A) AND GGTP >80 IU/mL (MODEL B), IN THE AFP ≤33.0 ng/dL or >33.0 ng/dL CATEGORIES. THE MODELS ARE CORRECTED FOR GENDER, AGE, AND ALCOHOL CONSUMPTION
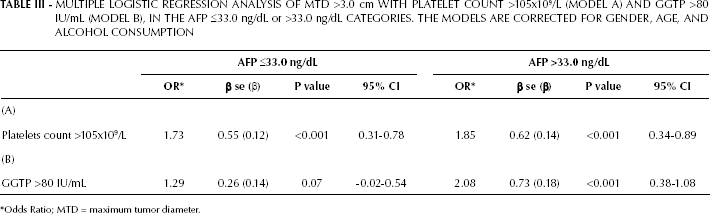
Odds Ratio; MTD = maximum tumor diameter.
Survival
We examined the effects of GGTP status, in both low and high-AFP groups, on survival for the total cohort: Figure 2 shows the Kaplan-Meier survival curves. We found a significant effect of elevated GGTP levels on survival in patients with both low and high AFP levels. The longest median survival time was found in the low-AFP and low-GGTP group, while the shortest median survival time was found in the high-AFP and high-GGTP group. Importantly, for the low-AFP group the GGTP levels had a significant impact on survival. When AFP and GGTP were separately introduced in the Cox model (models A and B, respectively), we found that patients with AFP >33.0 ng/dL had a greater risk (HR) compared with patients who had a GGTPP >80 IU/mL (Tab. IV). The same results were found for AFP >33.0 ng/dL and for GGTPP >80 IU/mL, when these parameters were introduced together into the model (model C). However, it is worth noting that when patients had both AFP >33.0 ng/dL and GGTP> 80 IU/mL, the HR increased more than two-fold.
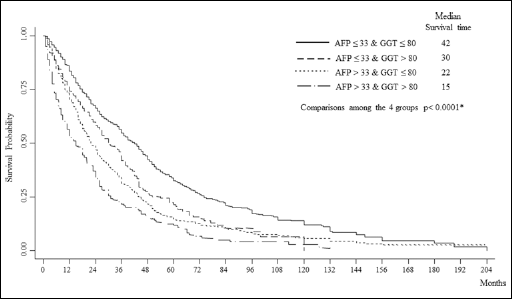
Kaplan-Meier survival plots for HCC patients with MTD >3 cm, according to 4 different combinations of blood AFP (≤33 ng/mL or >33 ng/mL) and GGTP levels (≤80 IU/mL or >80 IU/mL) in the total patients’ cohort. *Wilcoxon (Breslow) test.
MULTIPLE COX PROPORTIONAL HAZARD MODEL FOR DEATH OUTCOME, CORRECTED FOR GENDER, AGE, AND ALCOHOL CONSUMPTION
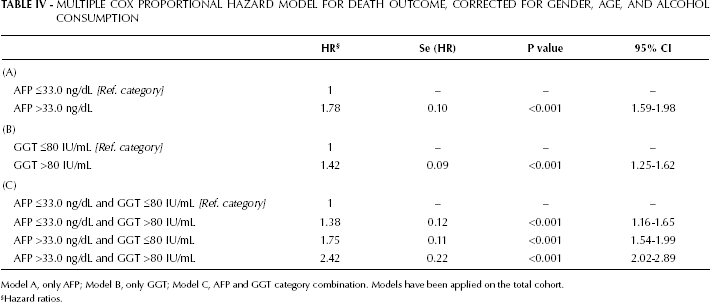
Model A, only AFP; Model B, only GGT; Model C, AFP and GGT category combination. Models have been applied on the total cohort.
Hazard ratios.
Discussion
Thrombocytopenia has been reported to be a cirrhosis surrogate, is regarded as a risk factor for HCC in cirrhotic patients, and is likely associated with small HCC size (10, 11). In this study we found similar results, which indicated that, for either low or high-AFP patients, platelet levels were higher in patients with large than small-size tumors. This was also true for GGTP, while for ALKP this was true only for the high-AFP patients. Survival was shorter among patients with larger tumors (in both AFP groups), as well as among high-AFP patients compared to low-AFP patients of the same tumor size group (Tab. I).
We then focused on the patients with low AFP, since they are more difficult to diagnose, but have better survival rates. GGTP status was examined more closely because of the findings reported in Table I. For each tumor size group, high GGTP levels were associated with shorter survival (Tab. IIA), higher ALKP, and slightly higher bilirubin levels. However, the tumor characteristics were similar, except for the increased tumor multifocality in the patients with high GGTP and large tumors. The presence of PVT or MTD were not significantly different. It is thus not clear how elevated GGTP levels are associated with worse prognosis, unless there is another tumor characteristic that was not measured. However, with respect to the worse survival in high-AFP patients, we previously noticed that neither the tumor size nor number was sufficient to explain their worse prognosis (12). Thus, there must be other parameters of HCC aggressiveness that we did not measure.
GGTP is part of the standard panel of routine blood liver enzymes that are measured in clinical practice as a quantification of liver damage; this parameter is also sensitive to smoking and alcohol consumption. However, GGTP also appears to have a rather specific relationship with HCC development in both experimental rodent hepatocarcinogenesis and human HCC (13, 14). To try distinguishing between the roles of GGTP as part of a standard liver damage test and its role as an HCC marker, especially since bilirubin levels were higher in larger tumors (Tab. I), we re-examined our data after excluding patients with elevated bilirubin levels (Tab. IIC). We found a non-significant association for patients with elevated GGTP levels with increased MTD and presence of PVT. However, in patients with large-size tumors, the incidence of PVT doubled and tumor multifocality was statistically increased by a 2.3-fold in the high compared to the low-GGTP group.
There have been many reports about the higher blood GGTP levels in HCC patients and its use as a possible tumor marker, as well as about its variant forms that may be HCC-specific (15-16-17-18-19-20-21-22-23-24-25-26-27-28). The significant effects of elevated GGTP levels on survival (Fig. 2 and Tab. IV), and the significant increase in the mean GGTP levels in both low and high-AFP patients that had large rather than small tumors (Tabs. I and III) all suggest that GGTP might be a potentially useful tumor marker, especially for those patients with low AFP levels, being more difficult to diagnose. ALKP levels were also significantly increased in both small and large-tumor groups, in association with the abovementioned increased GGTP levels; thus, ALKP has also been suggested to be an HCC tumor marker (29). Furthermore, the elevated GGTP but not AFP levels have been reported to be associated with a higher probability of death in HCC patients (30).
Conclusions
Most of the patients in our cohort had low AFP levels. Patients with elevated GGTP levels had a worse prognosis compared to those with low GGTP levels. GGTP may be a useful tumor and prognostic marker for HCC patients.
