Abstract
Background
Serum albumin levels have been shown to have prognostic significance in hepatocellular carcinoma (HCC), as part of an inflammatory index. The aim of this study was to examine the possible relationship of serum albumin levels to parameters of HCC aggressiveness.
Methods
A large HCC patient cohort was retrospectively examined, and the possible relationships of albumin levels to tumor diameter, multifocality, portal vein thrombosis (PVT) and α-fetoprotein levels were examined.
Results
HCC patients with lower serum albumin levels had significantly larger maximum tumor diameters, greater prevalence of PVT, increased tumor multifocality and higher α-fetoprotein levels, than HCC patients with higher albumin levels. A correlation was found between levels of these tumor parameters and serum albumin levels.
Conclusions
These results indicate that low serum albumin levels correlate with increased parameter measures of HCC aggressiveness, in addition to their role as a monitor of systemic inflammation. Decreased serum albumin might have a role in HCC aggressiveness.
Introduction
Clinical prognosticators in patients with hepatocellular carcinoma (HCC) are generally considered in 2 groupings. First to be considered are tumor factors, such as tumor size, number of tumor nodules, presence or absence of portal vein thrombosis (PVT) and α-fetoprotein (AFP) levels. Second, are demographic factors (age, sex, associated diseases and their severity in the liver) and liver or tumor microenvironmental factors, such as bilirubin, degree of cirrhosis and liver damage or other factors that are reflected in various liver gene signatures (1-8).
Indices of systemic inflammation have also been found to be independent cancer and HCC prognosticators (9-16). A widely used inflammation index is the Glasgow Prognostic Score (15, 16), which consists of 2 factors - namely, serum albumin and C-reactive protein (CRP) levels. A worse prognosis in this score is indicated by a combination of higher CRP levels and lower serum albumin levels. This has recently been applied to HCC therapy outcomes (17-20).
Furthermore, albumin values have been used in the 2-parameter albumin-bilirubin (ALBI) quantitative grading system for liver function in HCC, which has in turn been incorporated into several staging systems (21-25). There is thus much recent interest in serum albumin levels in prognostication, mainly as a reflection of liver function in HCC. However, there is also some direct experimental evidence that increased albumin levels can actually suppress HCC cell growth (26, 27). Viewed this way, albumin levels may influence prognosis as much by their actions on HCC biology as by their reflection of impaired liver function.
In this study, we examined the clinical correlations of serum albumin levels in a large HCC patient cohort with parameters of HCC behavior, and found an inverse relationship between serum albumin levels and HCC aggressiveness.
Methods
Patient data
We retrospectively analyzed a prospectively collected database of 4,139 HCC patients, as previously reported (28, 29), who had full baseline tumor parameter data, including computed axial tomography (CT) scan information on HCC size, number of tumor nodules and presence of PVT and plasma AFP levels; complete blood counts; routine blood liver function tests (total bilirubin, γ-glutamyl transpeptidase [GGT] and albumin); and patient demographics. Database management conformed to legislation on privacy, and this study conformed to the ethical guidelines of the Declaration of Helsinki. Approval for this retrospective study of deidentified HCC patients was obtained from the institutional review board.
Statistical analysis
Means and standard deviation (SD) for continuous variables and relative frequency for categorical variables were used as indices of centrality and dispersion of the distribution. For categorical variables, the chi-square and z test for proportions were used. The chi-square test for linear trend was also used to evaluate any linear trend against the null hypothesis of no trend among variables compared; a significant result suggested that the slope of the trend line was non-zero.
Since the distributions were nonnormal, the Spearman rank correlation was used to measure the association between 2 continuous variables, and the Wilcoxon rank sum (Mann-Whitney) test was used to test any difference between categories.
When testing the null hypothesis of no association, the probability level of α error, 2-tailed, was 0.05. All of the statistical computations were made using Stata version 10.0 statistical software (StataCorp., College Station, TX, USA).
Results
Serum albumin levels in relation to HCC parameters
Liver parameters and tumor characteristics (maximum tumor diameter [MTD]), number of tumor nodules, presence or absence of PVT and blood AFP levels were examined in small and large tumors, after dichotomization according to serum albumin levels (with 3.5 g/100 mL being the lower limit of the normal range). We found that patients with small tumors (<5 cm)who had low serum albumin levels had worse liver function (higher bilirubin levels) and significantly higher AFP levels and tumor multifocality, than patients with small tumors and normal albumin levels (Tab. I; left-side columns). Patients with larger tumors (≥5 cm diameter) and low serum albumin levels had significantly larger MTD and a significantly higher prevalence of PVT, as well as higher AFP and percentage tumor multifocality, compared with patients with larger tumors and normal serum albumin levels (Tab. I, right-side columns). This analysis was repeated in patients who had bilirubin levels <2.0 mg/mL, and similar differences were found between patients having normal or low serum albumin levels (Tab. II). Thus, serum bilirubin alone was insufficient to explain the tumor differences in patients with high or low albumin levels.
Comparisons between HCC patients, with serum albumin dichotomized into <3.5 vs. ≥3.5 g/dL
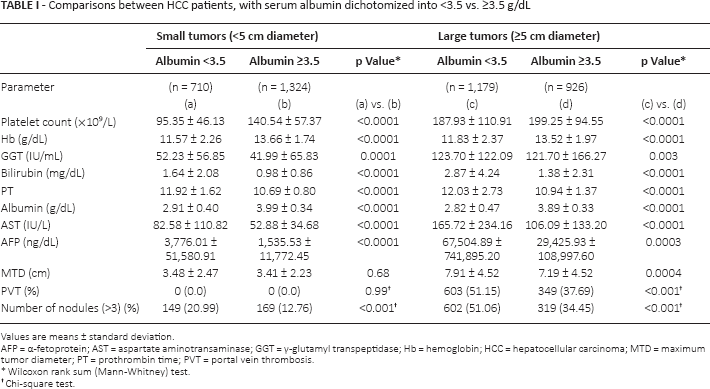
Values are means ± standard deviation.
AFP = α-fetoprotein; AST = aspartate aminotransaminase; GGT = γ-glutamyl transpeptidase; Hb = hemoglobin; HCC = hepatocellular carcinoma; MTD = maximum tumor diameter; PT = prothrombin time; PVT = portal vein thrombosis
Wilcoxon rank sum (Mann-Whitney) test.
Chi-square test.
Comparisons between HCC patients, with serum albumin dichotomized into <3.5 vs. ≥3.5 g/dL, and bilirubin <2.0 mg/dL
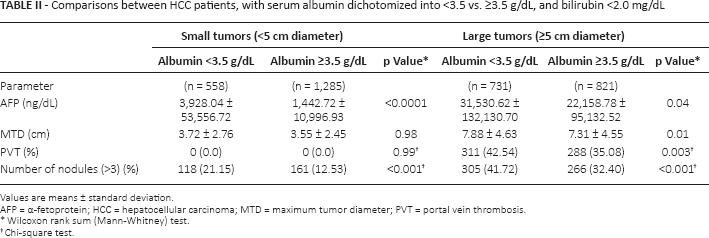
Values are means ± standard deviation.
AFP = α-fetoprotein; HCC = hepatocellular carcinoma; MTD = maximum tumor diameter; PVT = portal vein thrombosis
Wilcoxon rank sum (Mann-Whitney) test.
Chi-square test.
AFP and albumin subgroups
Elevated serum AFP levels have been considered to be a poor prognosis parameter in HCC patients (30, 31). We therefore examined a possible connection of normal or low serum albumin levels with AFP status. Patients were dichotomized into high or low AFP level cohorts (≤ or >100 ng/mL). Each AFP cohort was then further dichotomized into normal or low serum albumin levels, and only patients with serum bilirubin <2.0 mg/dL were included (Tab. III). Regardless of AFP cohort, patients had significantly larger tumors, more tumor multifocality and increased prevalence of PVT in the low-albumin groups.
Comparisons between HCC patients, dichotomized by AFP (≥100 vs. >100 ng/dL), with serum albumin (<3.5 vs. ≥3.5 g/dL) and normal bilirubin
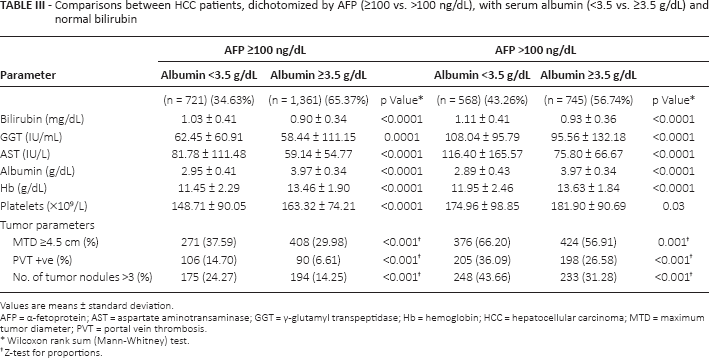
Values are means ± standard deviation.
AFP = α-fetoprotein; AST = aspartate aminotransaminase; GGT = γ-glutamyl transpeptidase; Hb = hemoglobin; HCC = hepatocellular carcinoma; MTD = maximum tumor diameter; PVT = portal vein thrombosis
Wilcoxon rank sum (Mann-Whitney) test.
Z-test for proportions
Trends in relationships of tumor parameters and serum albumin levels
We next examined the possibility of a trend existing between serum albumin levels and the 4 tumor parameters studied – namely, MTD, percentage of patients with PVT, tumor nodule multifocality and serum AFP levels (Fig. 1) – using 3 different albumin cutoffs of <2.5, 2.5-3.5 and >3.5 g/dL. We found a significant trend between each of the 3 albumin levels and measures of all 4 tumor parameters in patients with tumors >5 cm, but not in small tumors, as reflected in Tables I and II.
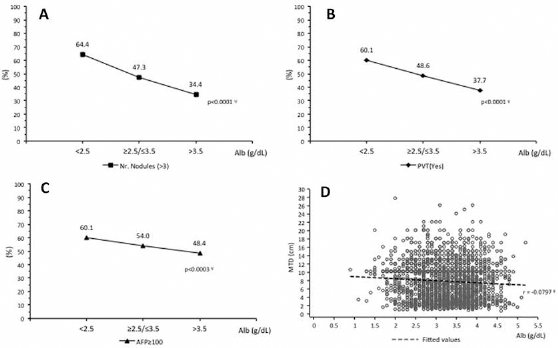
Comparisons between percentages of patients in high and low albumin (g/dL) groups for (A) number of tumor nodules (>3) (chi-square test, p<0.001; chi-square test for trend, p<0.0001); (B) PVT (yes) (chi-square test, p<0.001; chi-square test for trend, p<0.0001); (C) α-fetoprotein (AFP; ≥100 ng/mL) (chi-square test, p = 0.001; chi-square test for trend, p<0.0003); (D) MTD (cm) and albumin (g/dL) correlation (Spearman rank correlation, p = 0.0003, r=-0.0797). All patients had tumors >5 cm MTD. Alb = albumin; MTD = maximum tumor diameter; PVT = portal vein thrombosis. *Chi-square test; ψchi-square test for trend; ρSpearman rank correlation.
Multiple linear regression analysis
Albumin is a liver function test and is reflective of synthetic function. We were concerned that it was only one representative of a relationship between liver function in general and tumor characteristics. We therefore performed a multiple linear regression analysis of MTD on serum total bilirubin, aspartate aminotransferase (AST) and albumin (Tab. IV), and found that whereas albumin had a negative relationship to tumor diameter (protective), AST and bilirubin beta correlated positively with MTD. Thus, the liver parameters had differing relationships to MTD.
Multiple linear regression model of MTD on all parameters, albumin, AST and total bilirubin, together in the model
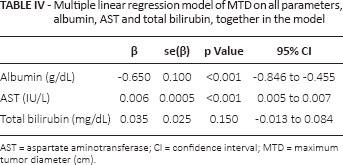
AST = aspartate aminotransferase; CI = confidence interval; MTD = maximum tumor diameter (cm).
Discussion
Albumin is 1 of the 2 components (together with CRP) of the Glasgow systemic inflammation index for cancer, an important and independent prognosticator for many cancers, including HCC (9-20). Additionally, albumin, as part of the ALBI grading system, has been shown to be a quantitative, useful and prognostic indicator of liver dysfunction in HCC patients (21-25). Albumin has previously been reported to have direct HCC growth inhibitory properties in vitro (26, 27), and there is also suggestive clinical evidence for an inhibitory effect on HCC in patients (30-32). Several studies have also shown a decrease in albumin levels in human HCC cells compared with the non-HCC liver (33-35).
In this study, we showed that in a large clinical HCC patient cohort, lower albumin levels were associated with more aggressive tumor parameters and higher AFP levels (Tab. I), especially in tumors >5 cm in diameter. We were concerned that lower albumin levels were associated with higher bilirubin levels and thus simply reflected liver parenchymal damage by tumor growth. However, when the data were reanalyzed with only patients having normal bilirubin levels (Tab. II), we still found an association between low albumin levels and more aggressive tumor parameters. In addition, a multiple linear regression model showed that whereas there was a positive relationship between AST or bilirubin and tumor size (MTD), there was a negative or protective effect for albumin (Tab. IV), thus dissociating the effects of albumin from AST and bilirubin. Furthermore, even in patients with high AFP levels, low albumin levels were associated with more aggressive HCC characteristics than in patients with high albumin levels (Tab. III). This represents another example of HCC biology in relationship to microenvironmental liver factors (2-8).
The mechanisms underlying this association between hypoalbuminemia and more aggressive HCC characteristics are currently unexplored. On the one hand, more aggressive HCC might cause liver parenchymal destruction, resulting from HCC growth in the liver. The association of low albumin with more aggressive HCC seen even in patients with normal bilirubin levels (Tab. II) renders this explanation less likely. However, there is recent evidence suggesting an association between HCC growth and poorer liver function, except for albumin, which seems to be protective (27, 30-32). The mechanisms by which albumin might protect against HCC growth or damage are not yet clear, although there is some experimental evidence that albumin might directly suppress HCC growth (26, 27), leading us to consider the possibility that in addition to its reflection of liver synthetic function and systemic inflammation, it might also have a direct tumor growth-modulating function in HCC patients. Furthermore, since albumin synthesis is a reflection of differentiated hepatocyte function, low albumin might even reflect poor HCC differentiation and more aggressive HCC biology, and conversely, normal albumin might reflect well-differentiated and slower growing HCCs, for which there is experimental support (6, 36). However, low albumin status is also a reflection of poor nutritional status and has been reported to have poor prognostic significance (37, 38). Whether this is cause or consequence is not quite clear. Albumin also has a variety of well-studied effects, in addition to regulation of plasma colloid osmotic pressure. These include transportation of hormones, fatty acids and drugs, and also antiinflammatory, antioxidant activity and protection against ischemic damage (34-42), and it may even be involved in TGFβ-mediated growth control (43). Low albumin is associated with enhanced inflammation, whereas albumin therapy in cirrhotic patients can suppress hepatic inflammation (44, 45). In NASH-associated HCC patients, there appears to be an inverse trend from oxidative markers including CRP, to antioxidative markers such as albumin (46). Thus, albumin has the potential for modulating various aspects of HCC biology, and might just possibly have potential uses in HCC therapy (47).
Footnotes
Abbreviations
Financial support: Funding was received in the form of a grant from the Italian Ministry of Health to the IRCCS de Bellis Medical Center.
Conflict of interest: The authors declare they have no conflict of interest.
