Abstract
Abstract
Introduction
Lung cancer is responsible for more deaths than any other malignancy and causes over 1 million deaths worldwide each year (1). A major factor implicated in the high mortality rates of lung cancer patients is the presence of metastatic tumors in approximately two thirds of patients at the time of diagnosis. Thus, efforts aimed at early identification and interventions in lung cancer are of primary importance. Fiberoptic bronchoscopy is one of the most important modalities used in the diagnosis of suspected lung cancer, and it has been recognized as the best technique for obtaining specimens to diagnose endobronchial lung carcinoma (2, 3). When an endoscopically visible tumor is identified during bronchoscopy, most bronchoscopists use forceps biopsy, brushing, and bronchial washing to obtain specimens for either histologic or cylotogic examination. However, in peripheral tumors that are beyond the visual segmental bronchi, the diagnostic sensitivity of the bronchoscopic methods is usually lower, depending on tumor size and location (4, 5). Although some studies already showed the diagnostic complementary effect of the cytologic analysis of bronchoalveolar lavage (BAL) fluids for peripheral lung cancer (6-7-8), the diagnostic yield of this analysis alone rages between 4% and 65% (9). One promising approach is the identification of lung cancer-specific biomarkers and molecular methods for the detection of these biomarkers at an early stage (10).
Molecular techniques have proven to be suitable for the identification of oncogenes' mutations in cytologically-negative specimens obtained from patients before the diagnosis of lung cancer. These tests showed an impressive sensitivity and diagnostic accuracy in the detection of tumor cells exfoliated in human fluids, including sputum, pleural effusion, and BAL fluid (11-12-13-14-15). Among the various potential markers, the accurate detection of KRAS and p53 gene mutations could be clinically meaningful since these 2 genes play an important role in the pathogenesis of lung cancer. Moreover, it has been reported that KRAS and p53 mutations are frequent and early events in the development of lung cancer (16-17-18). Several studies have assessed whether KRAS mutations could be used to detect tumor cells in BAL fluid samples from patients with suspected lung cancer. These studies reported variable frequencies of success, with detection rates ranging from 33% to 75%, in samples in which the primary tumor was positive for KRAS mutations (19-20-21-22). However, these studies were restricted to a small group of patients. On the other hand, in the study by Oshita et al, the authors detected KRAS mutations in BAL fluid not only from patients with lung cancer, but also from some patients with non-malignant lung disease (20). Thus, applying this method for clinical diagnosis can increase the probability of false positive diagnoses and lead to difficulties in patients' care. Thus, for the clinical interpretation of the KRAS mutations' analysis it may be important to include the results of the p53 mutations' analysis as well as the clinical evidence.
In the present study, we examined the mutations of the KRAS and p53 genes in matched samples of BAL fluid, tumor, and normal lung tissues from patients with peripheral non-small cell lung cancer (NSCLC), as well as in BAL fluid samples from patients with a benign lung disease, as to evaluate whether these 2 genes' mutations in BAL fluids can be used for the early diagnosis of peripheral NSCLC.
Materials and Methods
Patients
Forty-eight patients with peripheral NSCLC were consecutively enrolled into our prospective study that was conducted from October 2010 to May 2012. In this study, peripheral NSCLC was defined as a lung tumor with no bronchoscopically visible airway abnormality; at chest computed tomography (CT) all patients had a peripheral lung nodule or a mass that was operable, and had not received chemotherapy or radiation therapy. Preoperative evaluation included chest and upper abdominal CT, fiberoptic bronchoscopy with BAL and transbronchial lung biopsy (TBLB), brain magnetic resonance imaging (MRI) or CT, radionuclide bone scan or positive emission tomography (PET)/CT, and pulmonary function. Patients with extrathoracic malignancy, or those who had radiographic evidence of possible metastasis, were excluded from this study. Twenty-six patients with benign lung disease (BLD) who had an abnormal shadow on chest CT were included as controls. Bronchoscopy was performed in all patients, including those with benign lung disease and those with NSCLC. After bronchoscopy, all patients with peripheral NSCLC underwent surgery. Written informed consent for participation in the study was obtained. The study protocol was approved by the ethics committee of the Affiliated Hospital of Jiangsu University, in China.
The clinicopathological characteristics of the patients included in the study are shown in Table I. We included 27 men and 21 women, and the median age was 58 years (range 42- 75 years). Thirty-two patients were diagnosed with an adenocarcinoma, 14 patients had a squamous cell carcinoma, and 2 patients had a large cell carcinoma. The distribution into pathologic stages was as follows: stage I for 19 patients (9 stage IA and 10 stage IB), stage II for 25 patients (16 stage IIA and 9 stage IIB), and stage IIIA for 4 patients. Twenty-five patients had a cigarette smoking history, and 23 patients denied having a smoking history. In the group of patients with BLD, 17 were men and 9 were women, and the median age was 52 years (range 28-70 years). The causes of BLD included 7 cases of interstitial lung disease, 6 of chronic obstructive pulmonary disease, 6 of pulmonary tuberculosis, 4 of pneumonia, 2 of hamartoma and 1 case of inflammatory pseudotumor. Sixteen patients were smokers, and 10 patients were non-smokers.
ASSOCIATION BETWEEN KRAS AND P53 MUTATIONS IN THE PRIMARY TUMOR AND PATIENT CLINICAL CHARACTERISTICS
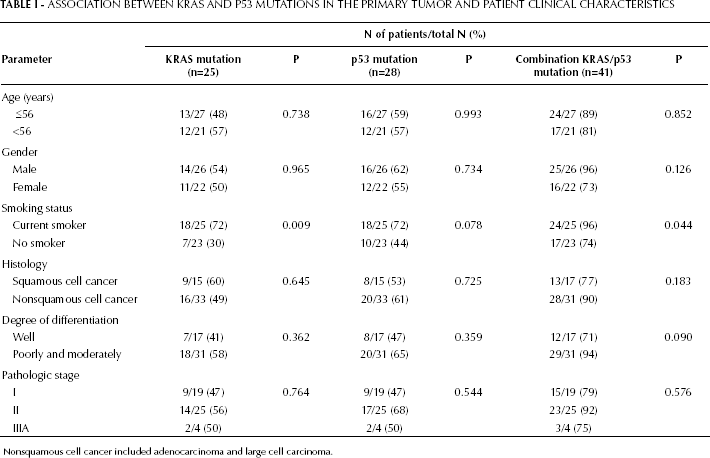
Nonsquamous cell cancer included adenocarcinoma and large cell carcinoma.
Specimen Collection
Bronchoscopy was performed with the usual method under local anesthesia using 2% xylocaine. In the majority of the cases, the bronchoscope was introduced transnasally. After routine inspection of the tracheobronchial tree, BAL was performed by wedging the bronchoscope in the segment or subsegment bronchus at the site showing abnormal shadow on chest CT. In patients with diffuse pulmonary disease, the bronchoscope was wedged into the middle lobe or lingual. Then, 3 aliquots of 40-50 mL of sterile saline solution were instilled and immediately aspirated by medical suction. The total amount of saline solution used varied between 120 mL and 150 mL. The recovered fluid was divided into 2 fractions: 1 was transferred to the Pathology Department for cytologic examination, and the other was immediately transported on ice to the laboratory for molecular analysis. Here, the fluid was centrifuged at 2,000g for 10 minutes at 4°C and cell pellets were washed twice with 3 mL of 1× phosphate-buffered saline; an aliquot of this material was stored at -80°C until processing. After BAL, TBLB and brushing were also performed for patients with suspected lung disease. During bronchoscopy, none of the patients had severe side effects other than some bleeding at the biopsy site.
Tumor tissues and nearby normal lung tissue (from macroscopically uninvolved areas 5-6 cm away from the tumor site) were obtained by surgical resection from NSCLC patients and immediately frozen in liquid nitrogen and then stored at -80°C until the time of molecular analysis.
Peripheral blood mononuclear cells from healthy donors were isolated by density gradient centrifugation and used as control normal DNA.
PCR-SSCP Analysis and DNA Sequencing
DNA was extracted from the samples of primary tumor tissue, normal lung tissue, and BAL fluid by the standard phenol-choroform extraction method (23), and stored at -80°C until use. Mutations within the KRAS (exon 1 and 2) and p53 (exon 5-8) genes were detected by PCR-single strand conformation polymorphism (PCR-SSCP) analysis following DNA sequencing. The primers were designed by Shanghai Invitrogen Biotechnology Co. Ltd., in China. The primer sequences and expected products' size are listed in Table II. Genomic DNA was amplified in a 25 μL total volume reaction mixture containing 100 ng of DNA, 100 ng of each primer, 100 μM of each dNTP, 1.5 mM MgCL2, 10× Taq buffer and 0.5 U Taq DNA polymerase (Aqqlied Biosystems, CA, USA). All the PCR programs included an initial denaturation for 5 minutes at 94°C, followed by 35 cycles of 45 seconds at 94°C, 45 seconds at the annealing temperature (56°C-60°C) of each PCR primer pair, and extension at 72°C for 45 seconds with a final extension at 72°C for 10 minutes. The PCR products were electrophoresed through a 2% agarose gel with ethidium bromide to confirm the quality of the amplification.
PRIMER SEQUENCES OF KRAS AND P53 EXONS AND ANNEALING TEMPERATURE

The PCR products mixed in denaturing buffer (95% formamide, 10 mM NaOH, 0.05% xylene-cyanol FF, and 0.05 bromophenol blue) in a 1:1 ratio were heat-denatured at 98°C for 10 minutes, and then immediately cooled on ice for 15 minutes; 5 μL of the solution were loaded on a 15% polyacrylamide gel and electrophoresed in 0.5x tris-borate EDTA buffer at 150 V for 5-6 hours at room temperature. The bands were visualized with a DNA silver staining kit (Bio-Rad, Richmond, CA, USA). Normal control samples were run simultaneously with specimens of tumor tissues and BAL fluids to identify the wide-type SSCP banding pattern. Samples with mutations show additional bands with different electrophoretic mobilities. Each sample with an SSCP mutation band was sequenced to verify the point mutation. The mutation sites were re-amplified with the respective untagged primers, purified, and sequenced using an ABI Prim 377 DNA Sequecing system (Perkin Elmer, Foster City, CA, US).
Detection of KRAS and p53 mutations were performed with the investigators being blinded to the information of the final diagnosis.
Statistical Analysis
Qualitative data are presented as frequencies and/or percentages and compared using the chi-square test or Fisher's exact test. Associations between mutations of the 2 genes and patients clinical characteristics were analyzed using the Fisher's exact test for categorical variables. The diagnostic accuracy of KRAS and p53 mutations in BAL fluid was evaluated by calculating sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV). All p values shown are two sided, and a p value of <0.05 was considered statistically significant. Statistical analyses were conducted using the statistical Analysis System Software version SPSS 13.0 (SPSS; Chicago, IL).
Sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV) were defined as follows: sensitivity=TP/(TP+FN), specificity=TN/(TN+FP), PPV=TP/(TP+FP), and NPV=TN/(TN+FN); where TP is true positive, FN is false negative, TN is true negative, and FP is false positive.
Results
Primary Tumor and Normal Lung Tissue
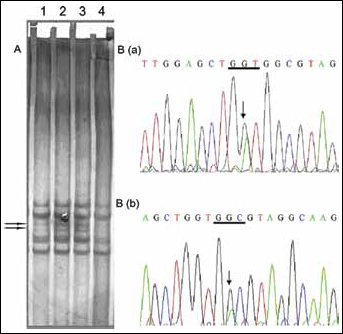
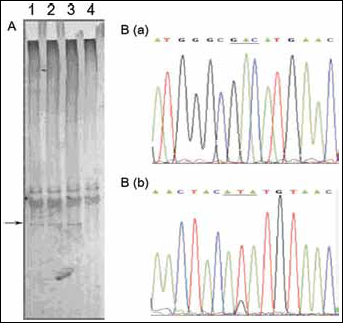
Molecular analysis of the primary tumor was performed for all 48 patients with peripheral NSCLC. Figure 1 shows some examples of the samples examined for KRAS gene mutation. Using PCR-SSCP analysis and DNA sequencing, KRAS mutations were found in 25 (52%) of the 48 primary tumors. Twenty-nine KRAS mutations were identified in 25 of the 48 NSCLC tumors screened. Of the 29 mutations, 59% (17/29) were found in codon 12, 34% (10/29) in codon 13, and 7% (2/29) in codon 61; 21/25 tumors presented single mutations, 2/29 presented a double mutation in codon 12, 2/29 presented a mutation in codon 12, and 1 in codon 13. Figure 2 shows some examples of the samples examined for p53 gene mutation. Mutation analysis of exon 5 to 8 of the p53 gene showed mutations in 28 (58%) of the 48 primary tumors. In particular, 36 p53 mutations were identified in 28 of the 48 primary tumors screened. Of the 36 mutations, 19% (7/36) were in exon 5, 33% (12/36) in exon 6, 39% (14/36) in exon 7, and 11% (4/36) in exon 8. Six tumor samples presented 2 (either 2 in same exon or 2 in two different exons) or 3 (of which 2 in two different exons) different p53 mutations. KRAS and p53 mutations coincided in 10 primary tumors (23%). The incidence rates for KRAS mutations alone and p53 mutations alone in primary tumors were, respectively, 29% (14/48) and 35% (17/48). At least one of the 2 genes' mutations was found in the primary tumor of 41 NSCLC patients (85%).
The occurrence of both KRAS and p53 mutations in primary tumor was not correlated with age, gender, histology type, tumor differentiation, or pathologic stage. There was a correlation between the occurrence of KRAS mutations and smoking status (p=0.009). A borderline significant association between the incidence of p53 mutations and cigarette smoking was observed (p=0.078) (Tab. I).
On the other hand, KRAS mutations and p53 mutations were observed respectively in 3 (6%) and 2 (4%) normal lung tissues. The type and locations of these mutations of the KRAS and p53 genes in normal lung tissue were identical to those present in the corresponding primary tumor.
BAL Fluid
KRAS mutations in BAL fluids were found in 18 (72%) of the 25 patients who had KRAS mutations in their primary tumor. p53 mutations in BAL fluids accompanied mutations in the primary tumor in 21/28 patients (75%). Simultaneous mutations of KRAS and p53 genes were observed in 7 BAL fluid samples. The type and locations of all KRAS and p53 mutations detected in BAL fluids were the same as those found in the corresponding primary tumors of the NSCLC patients. For the 48 patients with peripheral NSCLC included in this study, the detection rates of KRAS and p53 mutations in BAL fluids were, respectively, 38% (18/48) and 44% (21/48). Among them, 7 BAL fluid samples had coincident mutations of the KRAS and p53 genes. Thus, at least 1 of the 2 gene mutations was found in 32 BAL fluid samples (66%). In addition, KRAS and p53 mutations in BAL fluids were detected respectively in 2 (8%) and 1 (4%) of the 26 patients with BLD. The non-malignant diseases with KRAS of p53 mutations were 2 cases of tuberculosis and one of pneumonia. The detection rates of KRAS and p53 mutations were significantly higher in BAL fluids from NSCLC patients than those from BLD patients (for both genes, p<0.001). Among NSCLC patients, no significant difference was found in the detection rates of KRAS and p53 mutations between patients with mass tumors (≥3 cm) and those with nodular tumors (<3 cm). The rates of KRAS and p53 mutations in BAL fluids were lower than those in primary tumors; these results were due to the fact that tumor DNA in BAL fluids was diluted by normal DNA, and thus a mutation may have fallen below the sensitivity threshold.
Bronchoscopy
The diagnosis of malignancy was confirmed in 18 patients with peripheral NSCLC (38%) by the first diagnostic bronchoscopic procedure. Of these 18 patients, 12 (25%) were positive for cytology of brushing and/or BAL fluid, and 13 (27%) were positive for histology of TBLB (Tab. III). In the remaining 30 patients, the diagnosis of lung cancer was made by surgery.
DIAGNOSTIC PERFORMANCE OF CYTOLOGY, HISTOLOGY, AND KRAS AND P53 MUTATION IN BAL FLUIDS
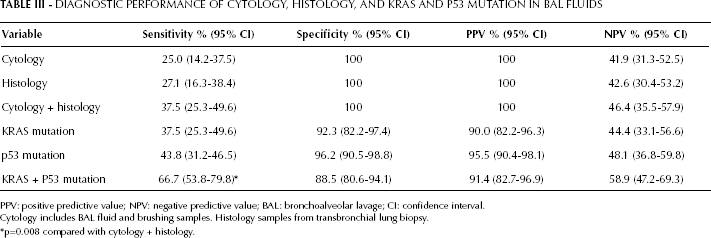
PPV: positive predictive value; NPV: negative predictive value; BAL: bronchoalveolar lavage; CI: confidence interval.
Cytology includes BAL fluid and brushing samples. Histology samples from transbronchial lung biopsy.
p=0.008 compared with cytology + histology.
Diagnostic Performance of the 2 Genes' Mutation in BAL Fluid
Table III describes the sensitivity, specificity, PPV, and NPV of cytology, histology, and KRAS and p53 mutations' detection in BAL fluid. The combination of KRAS and p53 mutation analysis reached a significantly higher sensitivity than that of cytology plus histology (p=0.008). It is worth noting that 14 patients with a negative cytology and histology at initial bronchoscopy had positive findings of KRAS and/or p53 mutations in BAL fluid. Therefore, the combination of KRAS and p53 mutation detection brought additional diagnostic information. Furthermore, all patients with positive bronchoscopic cytology and histology also had KRAS and/or p53 mutations in BAL fluid.
Discussion
This study shows that the occurrence of KRAS and p53 mutations in NSCLC is a relatively frequent and early event, and that it appears not to be related to the tumor pathologic stage and histologic type. These observations are in agreement with previous findings, suggesting that the detection of the 2 genes' mutations have a potential role as biomarkers for the early identification of NSCLC (19-20-21-22, 24, 25). However, our data should be interpreted with caution due to the relatively small size of our patients' group. The KRAS gene is one of the activating oncogenes most frequently found in human carcinomas. KRAS mutations (generally located in codons 12 and 13 in exon 2, and codon 61 in exon 3) have been found in several tumor types (26, 27). The majority of the mutations, especially those in NSCLC, are located in codon 12 and result in a constitutive activation of the k-ras protein (27). Mutations in the p53 tumor suppressor gene are also among the genetic abnormalities most frequently found in human cancers (28). In fact, mutations of the p53 gene have been observed in more than 50% of NSCLC patients (29). Since p53 mutations frequently occur in the so-called “hot-spot” region of exon 5-8, most studies only examined this region to evaluate the frequency of p53 mutation. It is possible that if the entire p53 gene had been sequenced, p53 mutations may also have been detected in other exons, which might have determined higher p53 mutation rates in the specimens considered. It has been reported that KRAS or p53 mutations in patients with NSCLC were associated not only with the genesis and progression of lung cancer, but also with shortened survival, thus indicating their role as predictors of poor prognosis (29, 30); however, this notion remains controversial.
In the present study, the positive rates of KRAS mutations were 52% in NSCLC tissues, 38% in BAL fluid samples from patients with peripheral NSCLC, 6% in normal lung tissues, and 8% in BAL fluid samples from patients with BLD. The incidence of KRAS mutations in the primary tumor was somewhat higher than the incidence reported in the majority of the other series (16, 18, 27). The finding of KRAS mutations in BLD is in keeping with those of a previous study, which reported that KRAS mutations were detected in BAL fluid of patients with pneumonia and focal fibrosis (20). KRAS mutation was considered an early event in lung carcinogenesis and was detected in precancerous lesions (18, 31). Therefore, KRAS mutation can be used not only as an early detection marker for lung cancer, but also as a prevention marker for lung carcinogenesis. In this study, benign lung diseases with KRAS mutation were mainly comprised of pulmonary tuberculosis and pneumonia, but the severity of the local inflammation was not evaluated, implying that false positive cases can be detected among patients with lung inflammatory disease. Furthermore, the positive rate of KRAS mutation was significantly higher in BAL fluid of peripheral NSCLC patients than in BAL fluid of BLD patients. However, the mutation assay alone had low efficiency for the early detection of NSCLC because its sensitivity was not superior to that of cytology plus histology by bronchoscopy. To reflect the molecular biological changes and improve the sensitivity of gene analysis, it is necessary to create additional molecular biological tests associated with lung carcinogenesis. Therefore, we performed PCR-SSCP and DNA sequencing for the combined assessment of KRAS and p53 mutations.
In the present study, the positive rates of p53 mutations were 58% in NSCLC tissues, 44% in BAL fluid samples of patients with peripheral NSCLC, 4% in normal lung tissues, and 4% in BAL fluid samples from patients with BLD. The incidence of p53 mutations in the primary tumors was within the range reported in the literature. It is worth noting that in this study the samples were first screened for p53 mutations using the PCR-SSCP technique, and only positive samples were subsequently sequenced. This method may have resulted in a somewhat decreased rate of mutation detection compared with the method of direct sequencing. Several studies reported that p53 mutations are detectable in normal lung tissue of patients with NSCLC and precancerous lesions of bronchial tissue (17, 18, 32). Thus, just like KRAS mutations, also p53 mutations are not an exclusive feature of carcinomas, and may also be associated with chronic inflammatory processes, or tobacco smoke exposure and preneoplastic bronchial lesions; additionally, p53 mutations may be an important step in the progression of NSCLC (31-32-33). Our study demonstrates that in NSCLC patients cigarette smoking was significantly associated with mutations in the KRAS gene: KRAS mutations were present in 72% of the tumors from patients with a history of cigarette smoking, whereas only 30% of the non-smokers had tumors with a KRAS mutation (p=0.009). A borderline significant correlation between cigarette smoking and mutation of the p53 gene was also observed in NSCLC tumors (p=0.078). The results are in line with most of the previous reports, which demonstrated that the mutations of KRAS and p53 were associated with cigarette smoking (34, 35). Our study has not demonstrated a significant relationship between the histologic type of NSCLC and mutations of the KRAS and p53 genes. In contrast, other investigators reported a relatively high frequency of KRAS mutations in adenocarcinomas (27), and a significantly low incidence of p53 mutations in squamous cell carcinoma (34). The discrepancies between studies may be explained by the relatively small sample size of our study, which could result in a bias of the data. Another possible explanation for these discrepancies may be that the methods of gene mutation analysis in our study differed from other studies: while here we used SSCP and direct sequencing, in other studies the authors used PCR in combination with denaturing gradient gel electrophoresis for KRAS mutations, and sequencing using the GeneChip assay and oligonucleotide-specific hybridization for p53 mutations (27, 34).
Another aspect resulting from our study is the unexpectedly high frequency of KRAS and p53 mutations found in BAL fluid from patients with peripheral NSCLC compared with the frequencies indicated in previous reports (16-17-18, 22). Once again, these differences may be caused by the relatively small sample size of our study or the different methods used in the various studies. The sensitivity of the method detecting KRAS or p53 mutations alone in BAL fluids was similar to that of cytology plus histology by initial bronchoscopy (Tab. III). However, the combined detection of KRAS and p53 mutations increased the sensitivity to 66%, a percentage significantly higher than that of cytology plus histology (38%, p=0.008). We found that the combined detection of KRAS and p53 mutations detected 14 of the 30 peripheral NSCLC patients that were negative for cytology and histology by initial bronchoscopic procedure. Moreover, KRAS and p53 mutations occur in the tumor of NSCLC patients with an equal frequency among all pathologic stages and histologic types, suggesting that these genetic changes are early events in lung cancer development, are not specific to histologic type, and that detection of these genetic abnormalities may be used to diagnose early stage NSCLC patients and all histologic types of NSCLC, including peripheral tumors, such as adenocarcinomas. However, the sensitivity of both KRAS and p53 mutation for distinguishing NSCLC from BLD patients were not very high (66%); thus, assaying the 2 gene mutations does not seem to be highly valuable as a diagnostic tool. On the other hand, in peripheral NSCLC that are beyond the visual segmental bronchi, the diagnostic sensitivity of cytology and histology by bronchoscopy is usually lower. However, the detection of KRAS and p53 mutations achieved significantly higher sensitivity than cytology and histology, suggesting that the simultaneous analysis of the 2 gene mutations in BAL fluid obtained by a less-invasive approach may increase the diagnostic yield of the procedure. On the other hand, our results can be further improved by using more recent technological approaches, such as targeted deep sequencing and nested digital PCR (36, 37).
In addition, we found the same type of mutation and same location in both primary tumor samples and BAL fluid specimens for patients with the 2 gene mutations in BAL fluids. Therefore, we conclude that the majority of the mutations detected in BAL fluids by PCR-SSCP and DNA sequencing are, indeed, indicative of the original tumor lesion.
In conclusion, although still limited by their sensitivity, molecular approaches can detect tumor-specific mutations in BAL fluid specimens. Our results suggest that mutations of the KRAS and p53 genes in BAL fluids may be useful biomarkers for the detection of peripheral NSCLC. Just as other biomarkers' limitations, the detection of KRAS and p53 gene mutations would not be used to confirm the diagnosis of malignancy; additionally, there is no evidence that these biomarkers may replace cytology and histology assessment. Therefore, the assay of KRAS and p53 gene mutation could be a helpful adjunct to cytology and histology examination for the diagnosis of peripheral NSCLC.
