Abstract
Aim
Colorectal cancer (CRC) is one of the most prevalent malignancies in Argentina with 11,043 new cases and 6,596 deaths estimated to have occurred in 2008. The present study was developed to clarify the differential expression of MUC1, MUC2, sLex, and sLea in colorectal cancer patients and their relationship with survival and clinical and histological features.
Methods
Ninety primary tumor samples and 43 metastatic lymph nodes from CRC patients were studied; follow-up was documented. Twenty-six adenoma and 68 histological normal mucosa specimens were analyzed. An immunohistochemical approach was applied and statistical analysis was performed.
Results
In tumor samples, MUC1, sLea, and sLex were highly expressed (94%, 67%, and 91%, respectively); also, we found a significantly increased expression of the 3 antigens in primary tumors and metastatic lymph nodes compared with normal mucosa and adenomas. MUC2 was expressed in 52% of both normal mucosa and CRC samples; this reactivity significantly decreased in metastatic lymph nodes (p<0.05). A multiple comparison analysis showed that MUC1 and sLex discriminated among 3 groups: normal, adenoma, and CRC tissues. The increase of sLex expression showed an association with recurrence, and survival analysis showed that a high sLex staining was significantly associated with a poor survival. By multivariate analysis MUC1 inmunoreactivity correlated positively and significantly with tumor size, while MUC2 expression showed the opposite correlation.
Conclusions
The correlation of sLex overexpression in primary tumors and metastatic lymph nodes, the discrimination among the normal, adenoma, and CRC groups based on sLex expression, as well as its association with recurrence and survival, all suggest a prognostic role of sLex in Argentinian CRC patients.
Introduction
Colorectal cancer (CRC) is one of the most prevalent malignancies in Argentina with 11,043 new cases and 6,596 deaths estimated to have occurred in 2008 (1), for a total population of about 40-million people.
Although early detection, deeper knowledge, and ameliorated therapies have improved the management of this disease, distant metastatic dissemination is still a serious event that makes CRC a lethal disease. Changes in mucin expression are usually found at the site of tumor localization. Specifically, mucin MUC1 expression is increased in colon cancers and correlates with a worse prognosis, while the expression of the MUC2 secreted gel-forming mucin is generally reduced in colorectal adenocarcinoma (2–8). Furthermore, alterations in mucin expression and glycosylation status of the membrane of tumor cells may play an important role in tumor progression, as it may affect tumoral migration and, consequently, tumor dissemination (9–11). Carbohydrate determinants containing sialylated structures, such as sLex and sLea, have proved to serve as ligands for selectins; sLex was first shown to bind directly to E-selectin, which also binds sLea; they play a role in the extravasation of cancer cells from the bloodstream and promote spreading to other tissues (10–12). Furthermore, the overexpression of sLea and sLex is associated with a worse prognosis in different gastrointestinal tumors (13, 14); in relation to this, Matsumoto et al (15) reported that the survival of CRC patients was directly related to the expression levels of sLex and sLea on their tumors.
The present study was developed to clarify the differential expression of MUC1, MUC2, sLex, and sLea in Argentinian CRC patients, and the relationship of these molecules with survival as well as with clinical and histological features. Although differences in the tissue distribution and prognostic value of MUC1, MUC2, and carbohydrate antigens have been documented (3, 4, 15, 16), an exhaustive analysis in relation to survival in Latin Americans has never been performed.
Materials and Methods
Patients and samples
The study included 90 consecutively patients with primary CRC who were diagnosed and underwent surgical resection at the Dr. José Penna Hospital (Bahía Blanca, province of Buenos Aires, Argentina) between 1997 and 2003; follow-up was documented for all patients.
The American Joint Committee on Cancer criteria (AJCC Cancer Staging Manual 6th edition, 2002) was used for patients’ staging. Seventy-one patients underwent surgical resection with curative intent and 19 with palliative surgery care. The mean number ± standard deviation of resected lymph nodes (LN) was 10±8 (range 0–47).
Formalin-fixed paraffin-embedded tissues were obtained from 90 primary CRC, while a metastatic LN was obtained from each of the 43 patients with lymph-disseminated disease. Twenty-five adenomas and 64 histological non-tumoural mucosa specimens were included as controls.
All procedures followed the World Medical Association Declaration of Helsinki (Finland, 1964) and its further modifications. Informed consent was obtained from all patients included in this study. This research was approved by the Regional Ethical Committee of the province of Buenos Aires, Argentina.
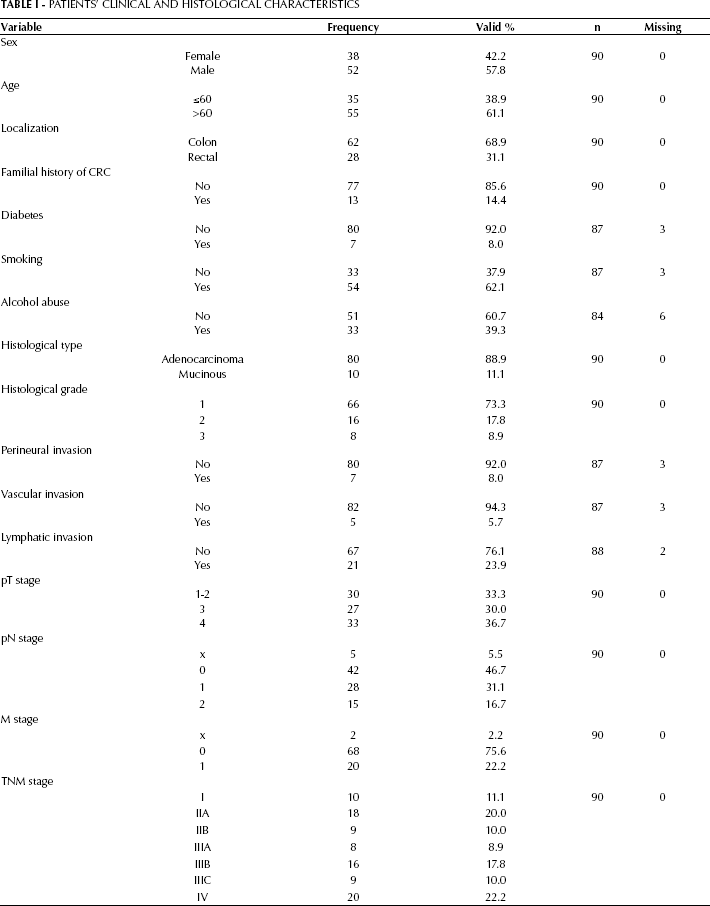
CRC patients’ clinical and histological information are summarized in Table I.
Patients’ Clinical and Histological Characteristics
Monoclonal antibodies (mAbs)
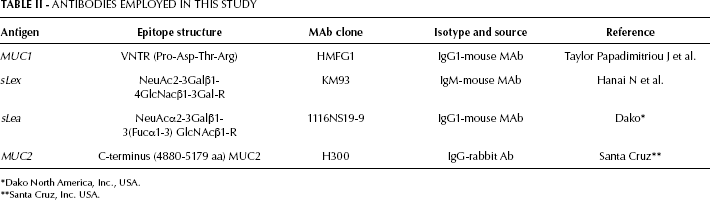
All the mAbs employed are summarized in Table II.
Antibodies Employed in this Study
Dako North America, Inc., USA.
Santa Cruz, Inc. USA.
Immunohistochemical analysis
The technique was performed following standard procedures. All specimens were fixed in formalin and embedded in paraffin. Sections were deparaffinized in xylene and hydrated in a graded ethanol series; dewaxed sections were placed in methanol with hydrogen peroxide (3%) for 15 minutes to block any endogenous peroxidase activity. After 3 washes with PBS, sections were blocked for nonspecific binding with 50 μL of normal horse serum diluted 1:10 in 1% bovine serum albumin/PBS for 15 minutes, and rinsed. Before samples were immunostained with mAbs, tissues were treated with 10 mM sodium citrate buffer at 100°C for 5 minutes for antigen retrieval. Afterwards, the sections were incubated with 50 μL mAbs at 4°C overnight in a moist chamber. The mAbs were diluted as follows: HMFG1 and KM93 1:1,000; 1116NS19-9 1:50; and H300 1:100. After 3 rinses with PBS, the sections were incubated with peroxidase-conjugated anti-mouse/rabbit Igs (LSAB+, Dako, CA, USA) for 15 minutes. After being washed, slides were counter-stained with hematoxylin, dehydrated in ethanol, washed with xylene and coverslipped with mounting media. Negative controls were performed by adding PBS instead of the primary antibody.
Evaluation of the staining
Specimens were examined with a light microscope by 2 independent observers (G.G. and A.O.Z.) who reached a final result by consensus. The antibody staining patterns were scored in a semi-quantitative manner. Staining intensity was graded as negative (–), low (+), moderate (++), and strong (+++). The number of low-power optical fields (x10) in a specimen with a positive reaction was expressed as a percentage of the total number of optical fields containing the tissue. The percentage of positive cells was graded as follows: 0, no positive cells; 1, positive in less than 5% of cells; 2, positive in 5%-30%; 3, positive in 30%-60%; 4, positive in more than 60% of cells.
A semi-quantitative scoring system was calculated as the product of intensity and percentage of positive cells; results were scored as rate 0 (absent/negative reaction), rate 1 to 6 (weak reaction), and rate 7 to 12 (strong reaction).
The pattern of reaction was classified as linear (membrane), cytoplasmatic, and mixed pattern (cytoplasmatic with plasma membrane staining); both apical and non-apical stainings were recorded. We also considered the positive reaction of the nucleus, as well as the staining of adjacent normal glands and the gland lumen content. The area of reaction (focal or diffuse) was also evaluated.
Statistical analysis
Correlations between mucins’ and carbohydrate antigens’ expressions were assessed employing the Kendall's tau-b coefficient. The associations of mucins and carbohydrate antigenic expression with other variables were also tested with non-parametric tests. For categorical variables, the Wilcoxon rank-sum test or Kruskal-Wallis test, including a Wilcoxon-type test for trend across ordered groups were used. In all cases, statistical significance was set at p<0.05. In order to compare the pattern of expression of the antigens, we collected from the same patient paired samples of non-tumoral mucosa, carcinoma, and lymph node metastasis, and we evaluated them by the McNemar test (significance level of p<0.01) with an analysis of overall marginal homogeneity (Stuart-Maxwell test, significance level of p<0.05).
The overall survival (OS) was calculated from the date of diagnosis until last control or date of the patient's death. Progression free survival (PFS) was calculated from the date of diagnosis until first tumor progression date. The evaluation of treatment response was performed using the Response Evaluation Criteria in Solid Tumors (R.E.C.I.S.T).
OS and PFS univariable associations with mucins and carbohydrate expression were evaluated employing the Kaplan-Meier estimator with the log-rank test. A multivariable Cox regression model addressed OS and PFS associations with the score of mucins, carbohydrate antigenic expression, gender (female vs. male), age (<60 vs. ≥60), history of familial CRC, smoking or alcoholism (yes versus no), histological type (adenocarcinoma versus mucinous), recurrence (yes versus no), histological grade (high 3 versus low 1–2), presence of lymphatic, vascular, or perineural invasion (yes versus no), pT stage (1–2 versus 3–4), pN stage (0 versus 1–2), and M stage (0 versus 1). All variables where normalized and significance was set at p<0.05.
All calculations were performed using the SPSS statistical software package v17.0.
Results
Expression of MUC1, MUC2, and carbohydrate-associated antigens in colorectal samples
The immunohistochemical results of the different antigens assayed in colorectal samples of normal mucosa, adenomas, primary tumors, and metastatic lymph nodes are summarized in Table III. Results are expressed as percentages of positive samples among the total number of samples.
Positive/Total (Percentage) Of Normal Mucosa, Adenoma, Adenocarcinoma, And Metastatic Lymph Nodes
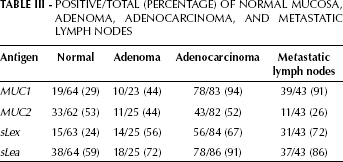
Taking into account the percentage of positive samples and the immunohistochemical score, an increase of MUC1 reactivity was observed from normal, through adenoma, to malignant samples (Fig. 1A); metastatic lymph nodes also showed a high MUC1 expression (Fig. 1A). Statistical analysis using the χ2 test revealed significant differences between normal mucosa and carcinoma (p<0.05), and between normal mucosa and metastatic lymph nodes (p<0.05); in addition, significant differences were found between adenoma and adenocarcinoma (p<0.05), and between adenoma and metastatic lymph nodes (p<0.05).
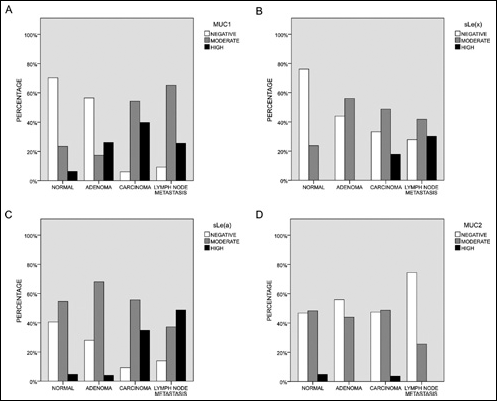
Antigenic expression of MUC1 (A), sLex (B), sLea (C) and MUC2 (D) in normal, adenoma, primary CRC tumors, and metastatic lymph nodes. Percentage of negative (white), moderate (grey), and high (black) expressing cells are depicted.
Similar results were obtained for sLex (Fig. 1B), for which statistical analysis revealed significant differences between normal mucosa and adenocarcinoma (p<0.05), normal mucosa and metastatic lymph nodes (p<0.05), and between primary tumors and metastatic lymph nodes.
In the case of sLea, the highest reactivity was found in primary tumors and metastatic lymph nodes, although a moderate expression was found in all groups (Fig. 1C). Significant differences in sLea expression were found between normal mucosa and primary tumors (p<0.05), between adenoma and CRC (p<0.05), and between adenoma and metastatic lymph nodes (p<0.05).
On the other hand, 53% of normal mucosa and 52% of CRC samples expressed MUC2, and this reactivity was significantly lower in metastatic lymph nodes; the intensity of the reaction was moderate in most samples (Tab. III, Fig. 1D).
Figure 2 shows microphotographs corresponding to MUC1, sLex, sLea, and MUC2 immunohistochemical expression in samples of normal mucosa (A, E, I, and M), adenoma (B, F, J, and N), CRC primary tumors (C, G, K, and O), and metastatic lymph nodes (D, H, L, and P).
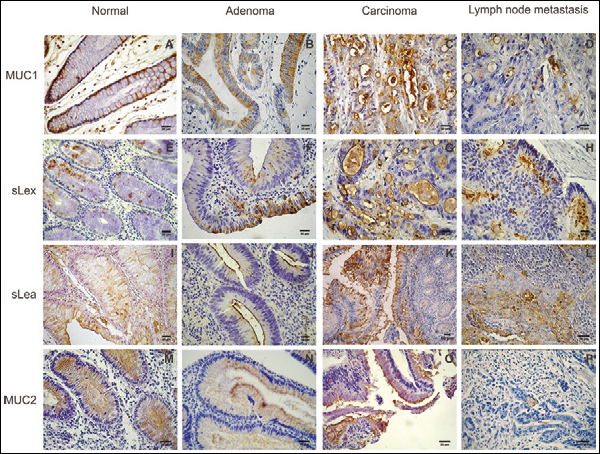
Microphotographs of immunoperoxidase staining of normal, adenoma, CRC primary tumor, and metastatic lymph node samples. Staining with anti-MUC1: (A) a normal sample showing some columnar reactive cells at the basal and lateral areas with a moderate intensity; (B) in an adenoma sample, a mild reaction can be seen mainly at the apical part of most cells; (C) most cells of a CRC primary tumor sample showing a strong reaction with a mixed staining at the membrane and cytoplasm; (D) a strong reaction in neoplastic cells of a metastatic lymph node. Staining with anti-sLex MAb: (E) a moderate and granular reaction in the cytoplasm of some normal cells; (F) a group of cells showing a strong and granular staining; (G) a strong staining can be seen at the apical membrane in most cells of a primary CRC tumor; (H) reaction of metastatic cells with a strong and mainly apical pattern. Staining with anti-sLea MAb: (I) normal cells showing a mixed moderate reaction; (J) an aden oma sample showing a linear reaction restricted to the apical membranes; (K) most neoplastic cells show a strong and mixed reaction; (L) metastatic reactive cells are clearly visible. A strong and mixed reaction is depicted. Anti-MUC2 MAb staining: (M) normal columnar and goblet cells are stained with a moderate and mixed reaction; lumen content is also stained; (N) moderate and apical reaction in an adenoma sample; (O) a mainly apical reaction; (P) a metastatic lymph node showing a negative reaction.
The pattern of expression was exhaustively analyzed by considering non-tumoural mucosa specimens and metastatic lymph nodes matching the results with paired malignant specimens of the same patient. Most malignant cells from primary tumors and metastatic lymph nodes showed a MUC1 reactivity that comprised the whole cytoplasm of the tumor cells, while in non-tumoural mucosa MUC1 was located at a basal level; significant differences were found when comparing non-tumoural mucosa and primary tumor (Stuart-Maxwell test, p=0.007).
With regard to sLex, staining of tumor samples showed a strong, apical, and mixed reaction of most specimens, while paired normal mucosa showed a weak, apical staining in a few samples (non-tumoural mucosa vs. primary tumor, p=0.017).
On the other hand, the sLea antigen was weakly expressed at the apical part of non-tumoural mucosa while almost all tumor and lymph node samples showed a higher intensity with a non-apical, cytoplasmic, or mixed pattern (no statistically significant differences were found).
In the case of MUC2, the non-tumoural mucosa samples showed a characteristic cytoplasmic reaction mainly in goblet cells, with a moderate to intense staining, as well as staining at the glandular lumen content; in carcinoma samples, this pattern changed to a weak or moderate reaction with an apical staining restricted to tumoural glands, most lymph nodes being negative (non-tumoural mucosa vs. primary tumor, p=0.001).
Relationship between antigenic expression and histological diagnosis in primary tumors, metastatic lymph nodes, adenoma, and normal mucosa
The Kendall and Tukey tests were employed to determine the possible correlation of the reactivity scores of each antigen according to the different histological specimens. The results obtained showed that MUC1, sLex, and sLea had an increased expression from normal, through adenoma and carcinoma, to metastatic lymph node samples (p<0.01). On the other hand, MUC2 showed its lowest levels of reactivity in metastatic lymph nodes, with a significant difference compared to normal samples (p<0.05). A multiple comparison analysis based on the Tukey test showed that MUC1 and sLex discriminated among 3 groups: normal, adenoma, and adenocarcinoma.
Association of antigenic expression with clinical and histological characteristics in CRC patients
In order to elucidate if MUC1 and carbohydrate antigenic expression in CRC patients was associated with histological and clinical features, a correlation analysis was carried out employing the Kendall and χ2 tests (Tab. IV). A significant positive correlation was found between pT stage and MUC1 (p<0.02), while a significant but negative correlation was detected between pT stage and MUC2 (p<0.005).
Association Between Muc1 And Muc2 Expression Scores And Pt Stage Obtained By Kendall Test
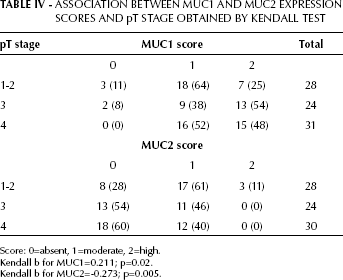
Score: 0=absent, 1=moderate, 2=high.
Kendall b for MUC1=0.211; p=0.02.
Kendall b for MUC2=-0.273; p=0.005.
The correlation between familial history of CRC and antigenic expression showed a positive and significant association with sLea reactivity (p=0.021). Tobacco smoking was the only risk factor here considered that showed a positive correlation with MUC1 expression (p=0.033), while alcohol abuse showed a negative correlation with sLea.
Relationship of antigenic expression with tumor recurrence and dissemination
According to the χ2 test, the increase of sLex expression showed an association with recurrence (p=0.07); when tumors were classified according to the intensity of the reaction, a significant difference was found between tumors with no or low expression of sLex (p=0.04). Furthermore, a higher expression of sLex was observed in patients that developed distant metastasis compared to those with local recurrence (p=0.014).
Relationship with survival
To study the influence of antigenic expression and co-expression, a survival analysis was carried out (Fig. 3). The median survival time for these series of CRC patients was 5.38 years (range 0.4–17.4 years). Considering the antigenic detection and their relationship with OS, sLex showed a significant difference between negative and strong staining (p=0.021; Fig. 3A). Other antigens did not show any statistically significant association. On the other hand, alcohol consumption (p=0.008), recurrence (p<0.001), and distant metastasis (p<0.001) were significantly associated with OS (Figs. 3B, C, and D, respectively).
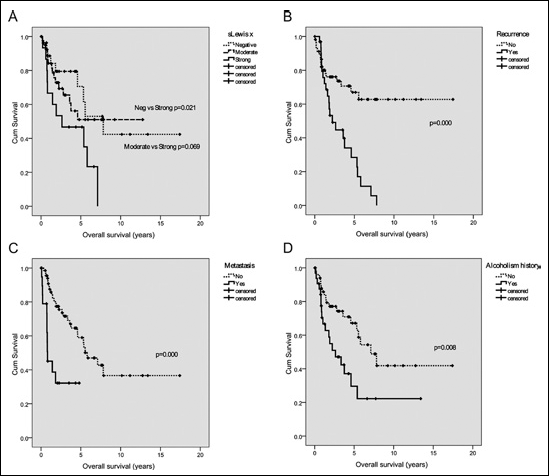
Kaplan-Meir analysis of CRC patients’ OS. Significant differences in sLex score (A), presence of recurrence (B), distant metastasis (C), and alcohol abuse (D) were assessed by the long rank test.
When disease-free survival (DFS) was considered, we found a significant association at univariate analysis with pN status (p=0.012), recurrence (p<0.001), and distant metastasis (p<0.001).
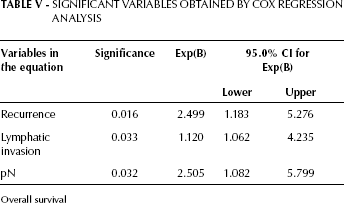
According to the Cox proportional-hazards model, the multivariate survival analysis included the scores of antigenic expression and multiple risks, and the prognostic factors described above. The statistical evaluation showed that pN, lymphatic invasion, and recurrence were strong predictors of survival. MUC1, sLex, sLea, and MUC2 did not have an independent prognostic value, although patients who expressed MUC2 had a better survival, indicated by a hazard ratio of 0.5 (95% confidence interval, 0.24–1.02). Table V and VI show the figures related to, respectively, OS and DFS.
Significant Variables Obtained By Cox Regression Analysis
Overall survival
Significant Variables Obtained By Cox Regression Analysis
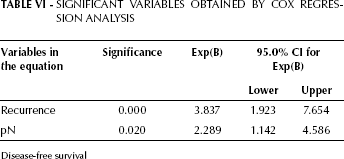
Disease-free survival
Discussion
Recent findings have emphasized the role of mucins in tumor progression (3, 8, 17); previous reports demonstrated that MUC1 and MUC2 may be respectively carriers of sLex and sLea (18). Meanwhile some of the underlying mechanisms that lead to their expression as well as their pathophysiological significance are presently being clarified. In this sense, Sakuma et al (19) found that sLex/sLea expression is strongly induced during epithelial-mesenchymal transition of colon cancer cells, and that c-Myc and CDX2 play key roles in this phenomenon. Furthermore, Gomes et al (20) demonstrated that the expression of sLex in MKN45 gastric cancer cells induced an invasive phenotype with a specific increase in c-Met activation.
The present study was developed to clarify the differential expression of MUC1, MUC2, sLex, and sLea in Argentinian CRC patients, and their relationship with survival. We found a significant increase in the expression of MUC1, sLea, and sLex in primary tumors and metastatic lymph nodes compared with normal mucosa and adenoma specimens. This result was obtained not only by the analysis of the staining of single samples, but also by the extensive study of the matched tumor samples, metastatic lymph nodes, and non-tumoural mucosa of the same patient.
Several lines of evidence point towards a biological role of MUC1 in CRC, since a positive correlation between MUC1 secretion, proliferation, invasiveness, metastasis, and negative prognosis (5, 6) has been described. According to Baldus et al (21), a higher MUC1 immunoreactivity in CRC compared to normal tissue seems to be mainly caused by a reduced glycosylation that leads to a damasking of the MUC1 peptide antigen allowing their immunohistochemical detection. Furthermore, and in accordance with other authors (21), the pattern of MUC1 expression in primary tumor samples and metastatic lymph nodes did not vary among samples. With regard to MUC2 expression, the normal mucosa showed a luminal pattern, while the neoplastic samples showed a cytoplasmic and apical reactivity. In CRC specimens, both sLea and sLex showed a mixed pattern of expression, which is in accordance with the findings of Villar-Portela et al (16).
Concerning the relationship among mucins expression and histological features, of special interest are the correlations we found between MUC1 and MUC2 and pT stage, which were detected by the Kendall and χ2 tests and confirmed by PCA. We also found a negative, significant correlation between MUC2 expression and pT stage, while the opposite was found for MUC1; these associations are in line with the observation that while MUC2 is frequently associated to colorectal normal tissues, MUC1 is associated to CRC (9). In this sense, it has been recently found that in CRC the loss of MUC2 expression was significantly associated with disease recurrence and tumor localization, and that MUC2 positivity significantly predicted longer DFS as well as disease-specific survival (7).
On the other hand, we found that alcohol abuse was related with poor survival; the association between alcohol abuse and CRC is not completely clear since regular wine consumption has been shown to favor the outcome of familial CRC, but not sporadic CRC (22). We also found that MUC1 expression showed a significant association with smoking habit, similarly to the findings of Benjamin et al (23) who described an analogous association of smoking habit and alcohol consumption in neoplastic gastric tissues.
Our results showed that CRC samples exhibited stronger immunoreactivity than normal and adenoma samples. Furthermore, a multiple comparison analysis based on the Tukey test showed that MUC1 and sLex discriminated among 3 groups: normal, adenoma, and adenocarcinoma. The high expression of the sLea antigen in our results may be explained by the fact that, in general, most type-1 chain determinants, such as sLea, are present on mature cells, whereas type-2 chain antigens, such as sLex, are present on immature cells and appear in the course of malignant transformation (24).
In order to ascertain whether the tumoral expression of sLea and sLex was correlated with survival, a statistical analysis was carried out: we found that an intense sLex staining was significantly associated with poor survival. Furthermore, the higher expression of sLex observed in patients who developed distant metastasis compared to those with local recurrence is in line with the prognostic value of the sLex antigen in CRC (14). Our results seem to indicate the involvement of sLex in tumor progression. SLex and sLea are E-selectin glycan ligands expressed on the surface of many types of cancer cells, including CRC (25). It is known that these glycans play crucial roles in hematogenous metastasis as well as in tumor angiogenesis, which can facilitate intravasation and postextravasational proliferation of cancer cells (26).
Many tumor cells have altered biosynthetic pathways of O-glycan, as well as aberrant mRNA levels and activities of glycosyltranferases. The synthesis of sLex and sLea antigens is the result of the coordinated action of α1,3-fucosyltransferases (α1,3-FUT) and α2,3-FUT. Recent reports (27) demonstrated that, in colon cancer tissues, FUT6 is a key regulator of sLex biosynthesis since it is the major α1,3-FUT mediating the addition of the last sugar in the biosynthesis of the sLex antigen. Furthermore, in a gastric cell line (KATO III), sLex is synthesized by FUT6, whereas in gastric MKN45 and colonic HT29 cell lines, it is most likely synthesized by FUT5 (28). It has been also shown that overexpression of ST3GAL3 in the MKN45 cell line induced the expression of both sLex and sLea, while overexpression of ST3GAL4 induced exclusively an increase in the expression of sLex (20, 28).
In conclusion, in our series, we found that MUC1, sLea, and sLex expression significantly augmented in primary tumors and metastatic lymph nodes compared with the normal and adenoma specimens, whereas MUC2 showed an inverse behavior. Importantly, we found that a high expression of sLex was associated with poor survival. In recent years, the interest on these antigens has increased, since new therapeutical approaches may be designed based on the clarified molecular mechanisms involved (20, 27, 29, 30).
Author's contributions
AOZ contributed with obtaining the samples, performing the IHC analysis and the microscopic observations. MER performed the statistical analysis and contributed to the analysis and interpretation of the data. GG contributed to the microscopic observations and acquisition of data. SOD contributed to the statistical analysis. AS-E and MVC contributed to the conception and design of the study, interpretation of the data, and writing of the manuscript.
Footnotes
Acknowledgment
Grateful acknowledgement is expressed to Prof. J. Taylor-Papadimitriou and Prof. J. Burchell (Research Oncology, King's College London, Guy's Hospital, London, UK) for the generous gift of the HMFG1 MAb, and to Dr. Hanai (Kyowa Hakko Tokyo Res Lab, Tokyo, Japan) for the gift of the KM93 MAb. Many thanks are expressed to Miss María Emilia Cermignani for performed English correction of the manuscript and Mr. Juan Carlos Molina for technical assistance.
