Abstract
The L1 cell adhesion molecule (L1CAM) is overexpressed in many human cancers and can serve as a biomarker for prognosis in most of these cancers (including type I endometrial carcinomas). Here we provide an optimized immunohistochemical staining procedure for a widely used automated platform (VENTANA™), which has recourse to commercially available primary antibody and detection reagents. In parallel, we optimized the staining on a semi-automated BioGenix (i6000) immunostainer. These protocols yield good stainings and should represent the basis for a reliable and standardized immunohistochemical detection of L1CAM in a variety of malignancies in different laboratories.
Introduction
The L1 cell adhesion molecule (L1CAM; CD171) is expressed in many human cancer tissues including ovarian and endometrial carcinoma, melanoma, pancreatic ductal adenocarcinoma, colon cancer, intrahepatic cholangiocarcinoma, renal carcinoma, gallbladder carcinoma, non-small cell lung carcinoma and certain brain tumors (1–9). Wherever tested in detail, L1CAM expression was found to correlate with a poor clinical outcome (6–12). The association with bad prognosis was attributed to the ability of L1CAM to activate the NFkB pathway, to drive migration and invasion of tumor cells, and to support metastasis formation (13–15). A recent study with more than 1,000 patients has shown that immunohistochemical detection of L1CAM is the best-ever published prognostic factor in the International Federation of Obstetricians and Gynaecologists (FIGO) stage I, endometrioid (histological type I) endometrial cancer. L1CAM detection shows clear superiority over the standardly used multifactor risk score in estimating the chance of recurrence and death (16). In line with this, the biomarker L1CAM could be decisive in advocating adjuvant treatment in early type I endometrial cancers. Therefore, a prospective randomized phase II trial comparing adjuvant chemotherapy to no treatment in patients with L1CAM-positive type I endometrial cancers has been initiated. It is self-evident that for all kind of treatment decisions a reliable and highly reproducible test to determine L1CAM expression (positivity) is required.
Detection of L1CAM in cancers is commonly done using immunohistochemistry (IHC) on formalin-fixed and paraffin-embedded (FFPE) tissues (1, 3, 4, 9, 11, 12). This is one of the most preferred techniques in surgical pathology, due to the readily available tissue samples, which can easily be obtained from routine tissue storage. However, unfortunately, the reproducibility of IHC methods applied to FFPE tissue sections within a single laboratory and among different laboratories is relatively poor. Although IHC is a semi-quantitative method always related to an inevitable observer bias, to allow a maximum of reliability this immunoassay must be performed with a degree of technical rigor and control that matches every other immunologically-based assay. Lack of 'standardization' is now recognized as a major impediment for future reliable diagnosis, prognosis, and utility in cancer treatment selection. Improvements in L1CAM-IHC are urgently needed to guarantee comparability of results among different laboratories.
The aim of our technical brief is to provide an optimized IHC staining protocol for L1CAM on widely used automated platforms using commercially available reagents. This protocol should enable investigators worldwide to detect L1CAM-positive tumors and resolve inconsistent results from different laboratories.
Methods
Based on our previous experience we established L1CAM immunostaining on 2 separate platforms: the fully automated VENTANA™ immunostainer, and the semi-automated BioGenix (i6000) immunostainer.
Optimized staining protocol on Ventana™ immunostainer
A-1: FFPE tissue sections
Collect 3-4 μm sections from a 40°C-44°C flotation bath containing deionized water and mount on Superfrost Plus slides.
Dry slides overnight at 37°C in a convection oven.
A-2: Epitope retrieval
Use HIER BUFFER pH 8.5 (Ventana cell conditioning 1) and heat for 60 minutes.
A-3: Primary antibody/incubation time
Use L1CAM (CD171) monoclonal antibody (mAb) clone 14.10/Cat. No. SIG-3911-1000 (concentrated format) diluted 1:100 and incubate for 32 minutes at 37°C.
A-4: Amplification kit: to increase the signal intensity and the primary antibody dilution
Use Ventana kit Cat. No. 760-080
A-5: Ultraview universal DAB detection kit
Use Ventana kit Cat. No. 760-500.
B-1: FFPE tissue sections
The same as
B-2: Epitope retrieval
Use Tris/EDTA buffer (Zytomed, ZUC029-500) and heat in a pressure cooker (Biocare Medical, DC2002-CE) for 1 minute and 4 seconds at 122°C. Cool down for 20 minutes at room temperature, rinse with distilled water for 2 minutes and wash with buffer.
B-3: Primary antibody/incubation time
Use L1CAM clone 14.10 diluted 1:40-1:100, and incubate for 60 minutes at room temperature.
B-4: No amplification step is needed
B-5: Visualization
Use Polymer conjugate (Nichirei Biosciences Inc., 414151F) and incubate for 30 minutes at room temperature. Wash with buffer and incubate with DAB (Zytomed, DAB 530) for 10 minutes at room temperature.
Results and Discussion
Our optimized staining protocols resulted in a clear and distinctive staining of the tumor mass, whereas the stroma showed no reactivity for L1CAM (Fig. 1).
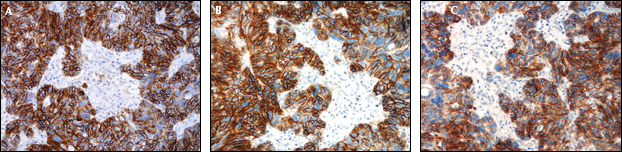
Control sections of a serous ovarian carcinoma stained with anti-L1CAM (14.10).
Several additional factors need to be considered to increase the staining intensity, decrease the nonspecific background, and minimize inconsistent L1CAM staining.
Pre-Analytical Factors
Tissues should be fixed in 10% phosphate-buffered formalin depending on the size of the tissue block and the type of tissue. Fixation between 18 and 24 hours seems to be ideal for most applications. Under-fixation can lead to edge staining, with strong signal on the edges of the section and no signal in the middle; over-fixation can mask the epitope. Adequate fixation is key to achieving reproducible and optimal antigen retrieval.
Sections
Sections of 3-4 μm should be cut and mounted on Superfrost plus slides. Once mounted the slides should be dried to remove any water that may be trapped under the section by incubating at 37°C overnight.
Analytical Factors
Primary antibody
The staining was done side by side on the semi-automated platform of BioGenics (i6000). The specific antibody clone selected was clone 14.10 (Cat. No. SIG-3911-1000 concentrated format), which was then diluted 1:100 and incubated for 32 minutes at 37°C in the Ventana platform, and for 60 minutes in the BioGenix immunostainer. The tissue specimen used for antibody optimization was L1CAM diffusely positive serous ovarian carcinoma. Nerve bundles strongly express L1CAM and served as a positive internal control in the test tissue.
Heat-induced antigen retrieval
In the Covance datasheet this step was omitted. The unmasking of epitopes was found to be crucial in high-pH retrieval solution. We used HIER BUFFER pH 8.5 (Ventana cell conditioning 1) heating for 60 minutes, and TRIS-EDTA pH 9 using a pressure cooker for the BioGenix.
Post-Analytical Factors
Interpretation
L1CAM staining cannot be interpreted on core tissues in tissue microarrays because of the pushing edge effect of focally L1CAM-expressing cells that complicate the determination of IHC-positive or negative assay.
Conclusive Remarks
In the present protocol we optimized the staining procedure for L1-14.10 on 2 widely used staining platforms. This mAb was chosen because we demonstrated earlier its superior capacity to detect L1CAM by IHC (17). It was not our intention to carry out a comparison of the 2 systems, or to evaluate the pros and cons of each platform. Both systems yielded stainings with similar intensity. These protocols will enable routine labs to use the existing equipment and will guarantee comparable results.
