Abstract
Background
At present plasma tissue inhibitor of metalloproteinases-1 (TIMP-1) is undergoing validation as a biological marker in colorectal cancer (CRC). The clinical implementation of plasma TIMP-1 in prognosis, prediction, screening and monitoring CRC requires robust information as to the influence of preanalytical factors, including inter- and intrapersonal biological variations. The aim of the present study was to evaluate the possible effects of smoking on the level of TIMP-1 in plasma from healthy subjects.
Materials and Methods
Forty-six never-smokers and 48 daily smokers participated in the 13-week study. Smokers were randomized into 3 groups of 16 subjects each: one group continued to smoke, a second group refrained from smoking and used a transdermal nicotine patch, and a third group refrained from smoking and used placebo patches. Plasma TIMP-1 levels were determined using ELISA.
Results
No significant differences in TIMP-1 levels could be shown between the smoking group, the 2 different abstinent smoking groups and the never-smoking group.
Conclusion
Smoking does not appear to have any influence on TIMP-1 levels in plasma collected from healthy subjects.
Keywords
Introduction
At present the use of soluble biological plasma markers in colorectal cancer (CRC) recommended by international guidelines is limited to carcinoembryonic antigen (CEA). A high serum level of this protein is strongly related to late stage of the disease and therefore valuable for monitoring disease after intended curative resection (1-3). However, CEA is far from able to detect early recurrence of disease in all patients who have been curatively resected (2, 3). Moreover, the relations between serum CEA and the presence of early stages of CRC are not strong enough to recommend the use of CEA in screening programs. Additional biological markers for daily use in CRC treatment are therefore urgently needed.
Of specific interest as a biological marker for CRC is tissue inhibitor of metalloproteinases-1 (TIMP-1), which can be determined in EDTA and citrate plasma (4). TIMP-1 is a multifunctional protein stored in α-granules of platelets, and its primary function is to inhibit matrix metalloproteinases (MMPs). Through this inhibition TIMP-1 plays a prominent role in the process of degradation and remodeling of the extracellular matrix. In addition to this inhibitory role, TIMP-1 has been shown to have antiapoptotic effects, to upregulate angiogenesis, and to stimulate cell growth (5). These additional effects of TIMP-1 might lead to considerations whether the molecule participates in the proliferation, invasion and dissemination of tumor cells (5, 6).
TIMP-1 is undergoing additional validation as a biological marker in CRC, the potential implications being early detection, determination of prognosis, prediction of treatment response, and monitoring of patients following initial treatment (7-11). The level of evidence 1 has just been achieved for plasma TIMP-1 as a preoperative prognostic variable with a pooled analysis presented at the American Society of Clinical Oncology 2012 Annual Meeting (12).
The clinical implementation of plasma TIMP-1 as a useful biomarker in CRC requires robust information on preanalytical factors in sampling, handling and storage, as well as thorough examination of postanalytical factors including inter- and intrapersonal biological variation, to clarify whether a given change in concentration is attributable to the presence or recurrence of CRC. This is of utmost importance to ensure the reproducibility of obtained clinical results (13, 14).
Gender-specific variation has been described with approximately 10% higher TIMP-1 levels in men than women (4). Moreover, TIMP-1 correlates weakly but significantly with age (4). Limited, nonsignificant intrapersonal variation over time has been observed, while physical exercise has no effect on plasma TIMP-1 levels (15). The levels of TIMP-1 appear to be increased among patients with various benign diseases including diabetes, cardiovascular disease and autoimmune diseases (5). Surgical trauma has a significant but transient impact on TIMP-1 levels: it causes an increase in TIMP-1 immediately after surgery which is followed by a stepwise decline, reaching preoperative levels 30-60 days after surgery (7, 8).
It is still unknown whether smoking affects TIMP-1 levels. Therefore, smoking might be considered a potential confounder in the interpretation of plasma TIMP-1 levels. The aim of the present study was thus to evaluate the effects of smoking on plasma TIMP-1 levels in healthy subjects.
Materials and Methods
Patients
A total of 94 healthy volunteers were included in the study after having provided written informed consent according to the Declaration of Helsinki II. Forty-eight subjects were daily smokers (24 women and 24 men, median age 33 years, range 20-40 years) consuming 20 (10-50) cigarettes per day and having a smoking history of 16 (3-50) pack years. The control group comprised 46 never-smokers (23 women and 23 men, median age 26 years, range 20-40 years). Prior to inclusion, all study participants were subjected to a CO breath test to confirm their smoking status. Exclusion criteria were chronic medical disease, pregnancy, and current or recent medication with corticosteroids or nonsteroidal antiinflammatory drugs. This specific study was part of a larger study of the impact of smoking, smoking cessation and transdermal nicotine patches on wound healing as approved by the Copenhagen Ethical Committee on Biomedical Research, Denmark (#KF 02-037/00) (16).
The study covered a period of 13 weeks. During the first week of the study, all current smokers agreed to smoke 20 standard filter cigarettes per day (Red Princē; Scandinavian Tobacco Company A/S, Copenhagen, Denmark). Subsequently, smokers were randomized in a double-blinded setup into 3 groups according to computer-generated randomization numbers which had been drawn from sealed, opaque, and consecutively arranged envelopes at the day of inclusion. In the first group, the subjects continued to smoke 20 filter cigarettes per day. In the second group, the subjects refrained from smoking and used a transdermal nicotine patch (TNP) (15 mg/16 h plus 10 mg/16 h, Nicorette; Pfizer, Copenhagen, Denmark) 24 hours a day. In the third group, smokers refrained from smoking and used placebo patches. Each subgroup comprised 8 women and 8 men.
Venous blood was collected from the smoking subjects between 8 and 10 AM 2 days after allocation to continuous smoking or abstention. The subjects had been fasting from midnight, except for pure water intake, and had refrained from smoking. The blood collection was repeated after 4, 8 and 12 weeks between 8 and 10 AM. From 6 of the 46 never-smokers (3 men and 3 women) blood was collected under the same conditions and at the same time points as in smoking subjects to control for potential biological and periodical effects. In the remaining 40 never-smokers (20 men and 20 women) blood was collected only once.
Specimen characteristics
Blood was collected in Vacutainers̄ (B&D, UK) using a light tourniquet and subsequently processed according to a predefined standard operating procedure. The centrifuged samples were separated into serum and plasma, and kept frozen at −80°C until analysis.
Assay methods
TIMP-1 protein levels were determined in EDTA plasma using an in-house, rigorously validated kinetic rate ELISA demonstrating low intra- and interassay coefficients of variation (17). The ELISA platform measures total TIMP-1, both the free form and the form in complex with MMPs. All samples were assayed in duplicate and the mean values were used in the subsequent statistical calculations. Finally, platelets were counted by an automated analysis (Sysmex E-2100) to compare the counts with the levels of plasma TIMP-1.
Compliance with the smoking and nonsmoking regimens was assessed using 3 methods. Firstly, blood analyses of carboxyhemoglobin using heparinized blood (ABL 700; Radiometer A/S, Copenhagen, Denmark) and cotinine (HP 5890A gas chromatograph; Hewlett-Packard, Los Angeles, CA) were undertaken. Secondly, twice-weekly CO breath tests (MicroSmokerlyzer; Bedfont Instruments, Rochester, UK) were performed. Validated compliance with abstention was defined as a CO breath test <6 ppm and a carboxyhemoglobin fraction <0.03 (18). Thirdly, structured interviews on compliance were conducted, reporting the number of cigarettes smoked by those randomized to continuous smoking and the use of nicotine or placebo patches by those randomized to abstinence.
Statistical analysis methods
Data were initially analyzed for compliance with the study plan and data from noncomplying subjects were discarded. Random-effects models for repeated measurements were applied to evaluate plasma TIMP-1 levels. According to the subjects’ actual smoking status at the time of blood sampling, the results were presented as mean and standard error of the mean (SEM) or standard error of the difference. The correlation between TIMP-1 and platelet count was calculated as Kendall's τβ. Finally, carboxyhemoglobin and cotinine levels were compared among groups by the Mann-Whitney
Results
Ninety of the 94 subjects (96%) completed the study. Four smokers, 1 of whom was randomized to continuous smoking and 3 to abstention, withdrew from the study because they did not wish to continue participation. In addition, data from 2 subjects randomized to abstention were discarded because they failed to meet the compliance criteria. In total, 88 subjects were available for analysis.
The carboxyhemoglobin and cotinine values confirmed that the abstainers complied with the study plan (data not shown). Four weeks after smoking cessation, the carboxyhemoglobin fraction in abstainers decreased and remained at a minimum throughout the study. Depending on the use of TNP or placebo patches in abstainers, the cotinine levels also decreased and remained lower than in the continuous smokers throughout the study. Subgroup analysis of values in the TNP and placebo groups confirmed compliance with TNP and placebo patches, respectively.
No significant differences in TIMP-1 levels between the continuous smokers, the abstainers, and the never-smokers were found. Data are shown in Table I. The plasma TIMP-1 levels did not correlate with the platelet counts (τβ=0.07, p=0.109) as shown in Figure 1.
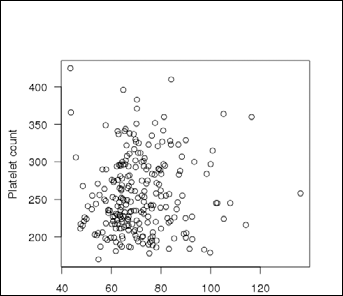
Correlation between plasma levels of TIMP-1 and platelet count. τβ=0.07; p=0.109.
Plasma Levels Of Timp-1 According To Groups
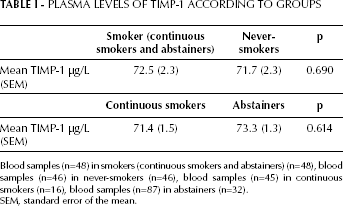
Blood samples (n=48) in smokers (continuous smokers and abstainers) (n=48), blood samples (n=46) in never-smokers (n=46), blood samples (n=45) in continuous smokers (n=16), blood samples (n=87) in abstainers (n=32).
SEM, standard error of the mean.
Discussion
The results of the present study showed that smoking did not influence plasma TIMP-1 levels in healthy individuals. The reliability of the results in the present study heavily depends on the compliance with abstention from smoking in the abstainers. In addition to structured interviews, this was thoroughly controlled by blood analysis of carboxyhemoglobin and cotinine and CO breath tests. Abstaining subjects with carboxyhemoglobin fraction and CO breath tests above the defined validated compliance level were discarded, retaining only TIMP-1 values from abstaining subjects who refrained from smoking for further analysis.
TIMP-1 is stored in the α-granules of platelets (5). To further validate our conclusion that smoking does not influence the plasma TIMP-1 level, a correlation analysis was performed between plasma TIMP-1 levels and platelet counts in whole blood. A correlation could not be shown, suggesting that release of TIMP-1 from the platelets was limited in the test samples. This finding eliminates platelet count as a confounder of the results of the present study.
Because of the multifunctionality of the TIMP-1 protein with effects on extracellular matrix, apoptosis, angiogenesis and cellular growth, it has been suggested that TIMP-1 plays a crucial role in the growth and dissemination of malignant diseases. Increased plasma TIMP-1 levels have been reported in patients with CRC, making TIMP-1 a potentially useful biological marker for early detection of CRC. Other biological protein markers have been shown to be under the influence of smoking, an example being surfactant protein A, where smoking increases the measured protein level (19). We therefore analyzed whether smoking might have an effect on plasma TIMP-1 levels and we could not prove it. Similar results have recently been shown for the 2 TIMP-1 binding partners MMP-8 and MMP-9 (20).
The present data indicate that smoking status does not necessarily need to be considered a dependent factor in the evaluation of plasma TIMP-1 values as a biological marker in patients with CRC or other diseases. The results may be of particular value in future population screening procedures based on blood samples.
It is concluded that smoking does not influence the levels of plasma TIMP-1 in healthy subjects. However, the results must be validated among patients with CRC or other diseases where plasma TIMP-1 may be a valuable biological marker.
Footnotes
Acknowledgments
We thank Birgitte Sander Nielsen and Vibeke Jensen for technical assistance. The Strategic Research Council, the Danish Cancer Society, the Danish Foundation for Advanced Technology, the Kornerup Fund, the Aase & Ejnar Danielsen Fund, the Aage and Johanne-Louis-Hansen Fund, the Arvid Nilsson Fund, a the Walter Christensen Fund, and the Kathrine & Vigo Skovgaard Fund provided financial support.
