Abstract
Background:
This study investigated the design and osseointegration process of transitive porous implants that can be used in humans and all trabecular and compact bone structure animals. The aim was to find a way of forming a strong and durable tissue bond on the bone–implant interface.
Methods:
Massive and transitive porous implants were produced on a direct metal laser sintering machine, surgically implanted into the skulls of sheep and kept in place for 12 weeks. At the end of the 12-week period, the Massive and porous implants removed from the sheep were investigated by scanning electron microscopy (SEM) to monitor the osseointegration process.
Results:
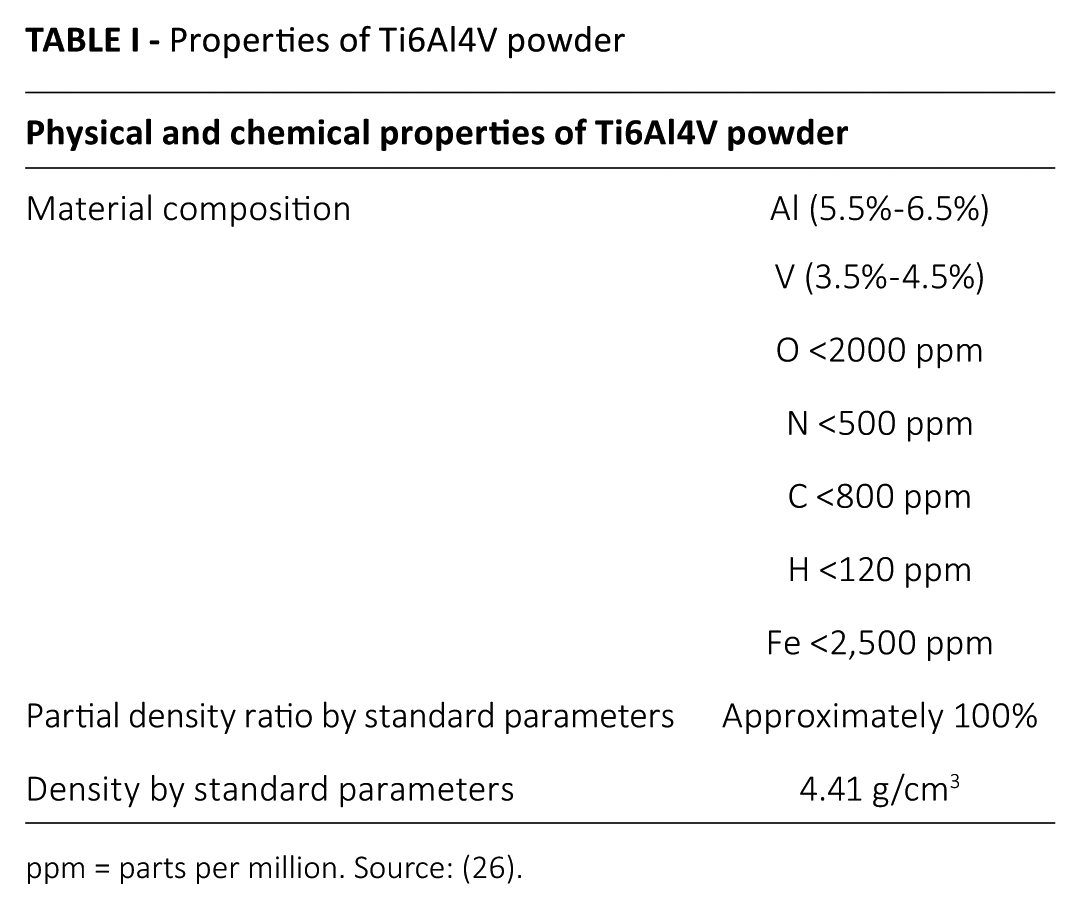
In the literature, each study has selected standard sizes for pore diameter in the structures they use. However, none of these involved transitional porous structures. In this study, as opposed to standard pores, there were spherical or elliptical pores at the micro level, development channels and an inner region. Bone cells developed in the inner region. Transitive pores grown gradually in accordance with the natural structure of the bone were modeled in the inner region for cells to develop. Due to this structure, a strong and durable tissue bond could be formed at the bone–implant interface.
Conclusions:
Osseointegration processes of Massive vs. porous implants were compared. It was observed that cells were concentrated on the surface of Massive implants. Therefore, osseointegration between implant and bone was less than that of porous implants. In transitive porous implants, as opposed to Massive implants, an outer region was formed in the bone–implant interface that allowed tissue development.
Keywords
Introduction
Artificial implants are used to treat bone structure defects due to disease, cancer, trauma, etc. (1-3). Materials used in implant manufacturing are usually metals, ceramics, polymers or composites (4-7) and, in general, can be classified as metals and nonmetals (8). Metals are generally preferred in terms of biocompatibility, ductility and strength. The most commonly used metals as implant materials are stainless steel, chrome-cobalt and titanium-based alloys, but titanium is preferred because of its high strength and corrosion resistance, low weight, good biocompatibility and osseointegration properties (9, 10). Titanium and its alloys are said to be the most effective among biomaterials, and they are widely used in numerous implant applications (11).
Osseointegration of the implant is affected by the mechanical properties of its material, design and manufacturing method. Implants are manufactured by conventional methods in standard structures, shapes and dimensions, and these are most commonly used in surgical operations. However, standard implants sometimes do not completely fit the individual anatomical structure of the patient (12, 13), resulting in failure to achieve the best outcomes for the patient. Therefore, it is essential to produce tailored implants that are completely suitable for the patient’s anatomical structure. Custom-made implants decrease complications and enhance the healing process due to better geometrical coherency and osseointegration.
In addition to a custom-made design, a porous structure enabling tissue development is also desired in implants. The internal structure of human bone displays an outer region consisting of a microporous cortex layer and an inner region consisting of a variable porous and sponge-like structure (14, 15). The structure of the skull bone resembles other bone structures, but its platy bones consist of a sponge-like structure. It is covered by compact bones having variable thickness in the inner and outer regions. There is a porous structure between these 2 compact layers. Pore voids are covered by a thin cellular layer (14). Modeling and design of custom-made implants coherent with anatomy and cell development are required in skull implants; however, conventional manufacturing methods cannot meet that demand. Additive manufacturing methods developed in recent years are a remedy for that problem.
Nowadays, additive manufacturing methods have been used to produce implants using 2- or 3-dimensional (3D) numerical models obtained from computed tomography, magnetic resonance imaging or a 3D scanner. Additive manufacturing involves adding materials on top of each other, layer by layer. This method produces parts that cannot be produced by conventional methods.
Although additive manufacturing methods might be identical, they vary with respect to raw materials and the approach taken to bonding the layers. Additive manufacturing methods include stereolithography, selective laser sintering (SLS), fused deposition modeling and selective laser melting (SLM) (16). It has been observed in reports in the literature that there has been an increased focus on this topic in recent years (17-19).
The production of real parts using SLS and SLM additive manufacturing methods has been researched in various fields, such as medicine, mold-making, aeronautics and space technology. SLS/SLM systems were developed as a rapid prototyping technology in the early 1990s (20). In recent years, there has been an increased use of a method called additive manufacturing. With the further development of SLS and SLM systems, it has become possible to produce a part using metal powders. The mechanical properties of parts manufactured by this method have been equal to, or sometimes greater than, those manufactured by conventional methods (21, 22).
This present study investigated the design and osseointegration process of transitive porous implants that can be used in humans and all trabecular and compact bone structure animals. The design of the implant included inner and outer regions in a cylindrical and planar shape. The implant was designed as a single piece, providing for the attachment of 2 regions. Semispherical and elliptical pores at the micro level were formed in the outer region. Channels were formed in the micropores to enable cells to develop in the inner region. Transitive pores, gradually increasing in size according to the natural structure of bone, were modeled for cells to develop in the inner region. The aim was to provide the formation of a strong and durable tissue bond on the bone–implant interface. It was predicted that such an implant design would extend implant revision time, provide a better geometric coherency and improve quality of life for the patient.
Mechanical tests were conducted to determine optimum parameters, and the implants were produced based on those. Later, cranioplasty was performed to transfer Massive implants into 3 sheep and transitive porous implants into 3 other sheep. The sheep skulls were monitored for 12 weeks, and the health of the sheep was constantly monitored by expert veterinarians. Finally, the implants were removed and investigated for osseointegration using scanning electron microscopy (SEM).
Materials and methods
Ti6Al4V metal powder was used in the study. Based on information about implant design, manufacturing and experimental animal application, Massive and transitive porous implants were designed using Massive Works® and Netfabb® software. Implants were produced on an EOS M 280 direct metal laser sintering (DMLS) bench and then implanted into the skulls of the 6 experimental sheep.
Material
Ti6Al4V metal powder was used as the implant material, the chemical composition of which is shown in Table I. Ti6Al4V is widely used in many fields, such as defense, aerospace, chemistry and medicine due to its high strength and corrosion resistance values, despite its molecular mass (23-25).
Implant design
The implant design was created with outer and inner regions in a cylindrical and planar shape. The implant was designed as a single piece, providing for the attachment of 2 regions, as shown in Figure 1. Semispherical and elliptical pores at the micro level (500 µm) were formed in the outer region. Cells developed in micro pores were drawn to ensure that they would develop inside the inner region through cell development channels (200 µm). Porosity was introduced in the inner region to ensure the survival of cells developing from the channels to the inside (500-1,000 µm). The outer region of the implant was sketched by Massive Works® software through a special controlled (semispherical structure) porous model, as shown in Figure 2. Inner regions of the implant were sketched using KRZ unit pore structures in Netfabb® software. As shown in Figure 3, transitive porous designs were completed by combining the outer region with the inner regions.
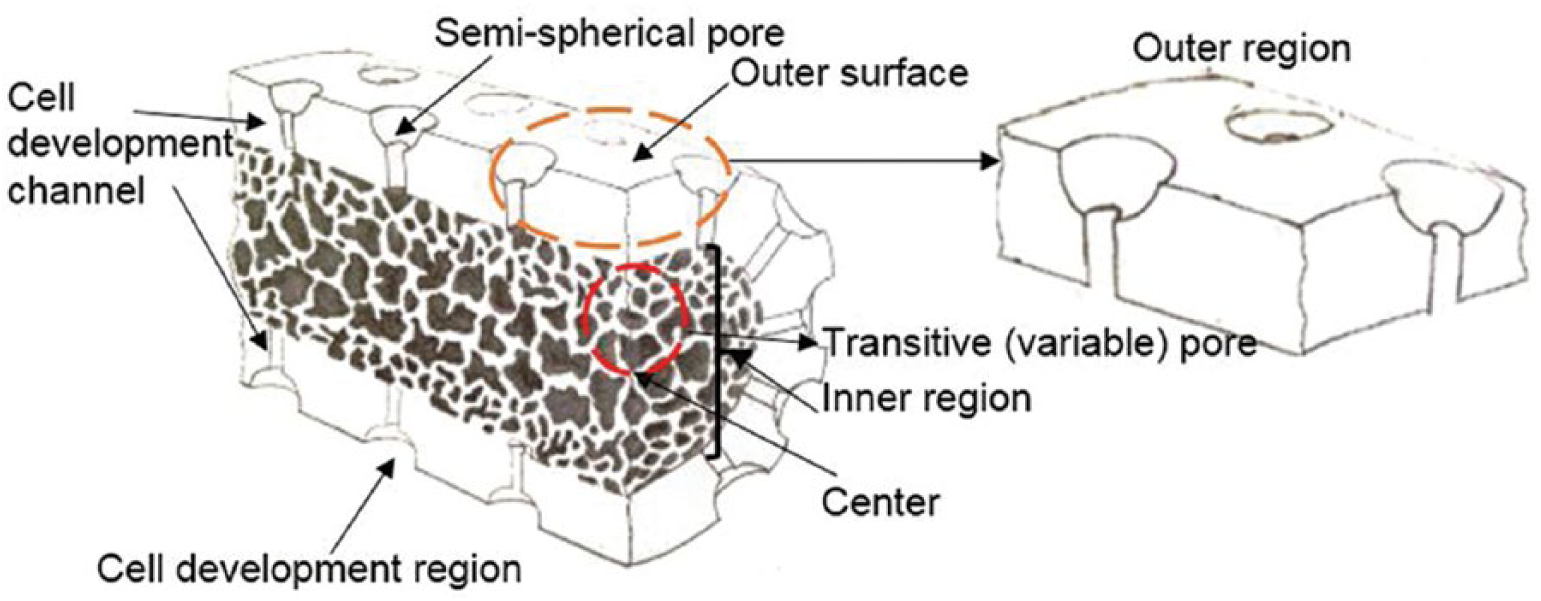
Cylindrical transitive porous structure.
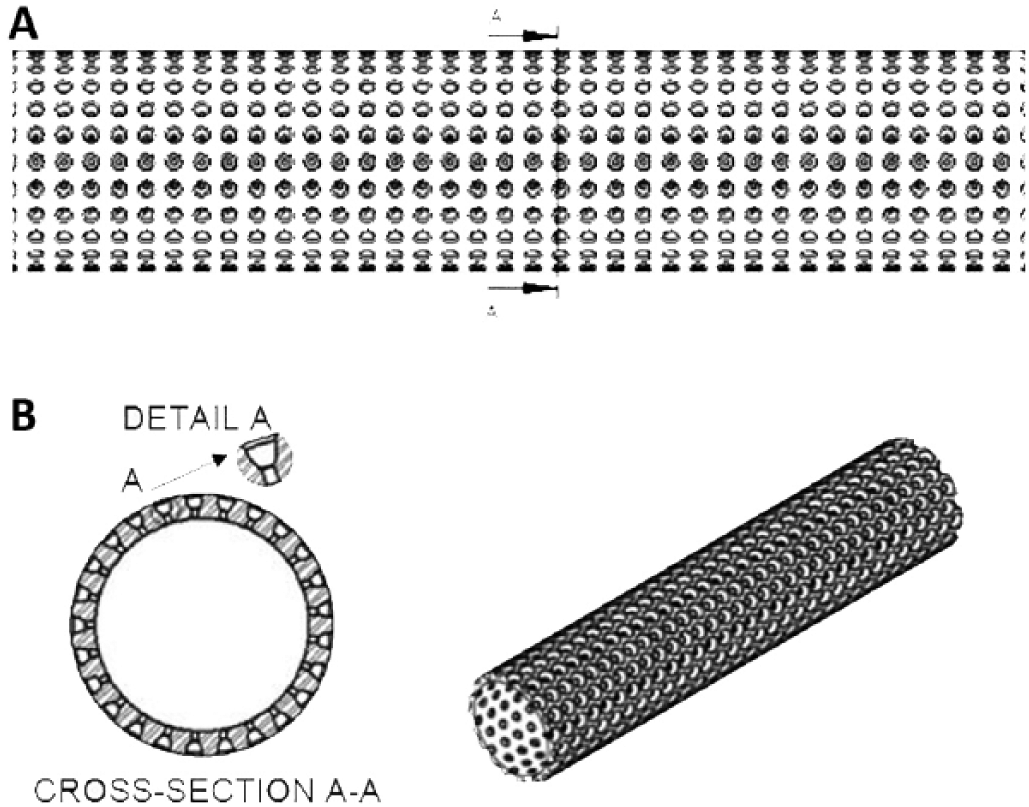
The outer region of an implant: (
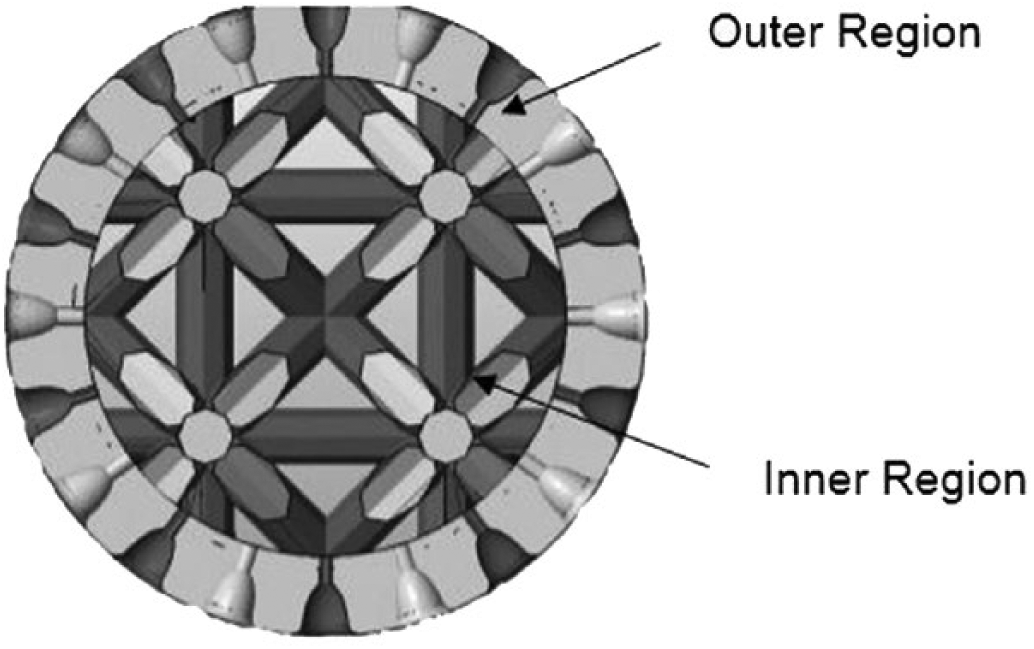
Cross-section of transitive porous model of the implant.
Implant manufacturing
Defects were formed in each experimental animal by surgical operation. CT scans of bone-defective regions were made. The skulls of the sheep were 3D modeled using CT data with Mimics® software. The 3D model of the skull of the sheep was transferred into 3-Matic® software to design a specific implant that was completely coherent with the defective region (Fig. 4).
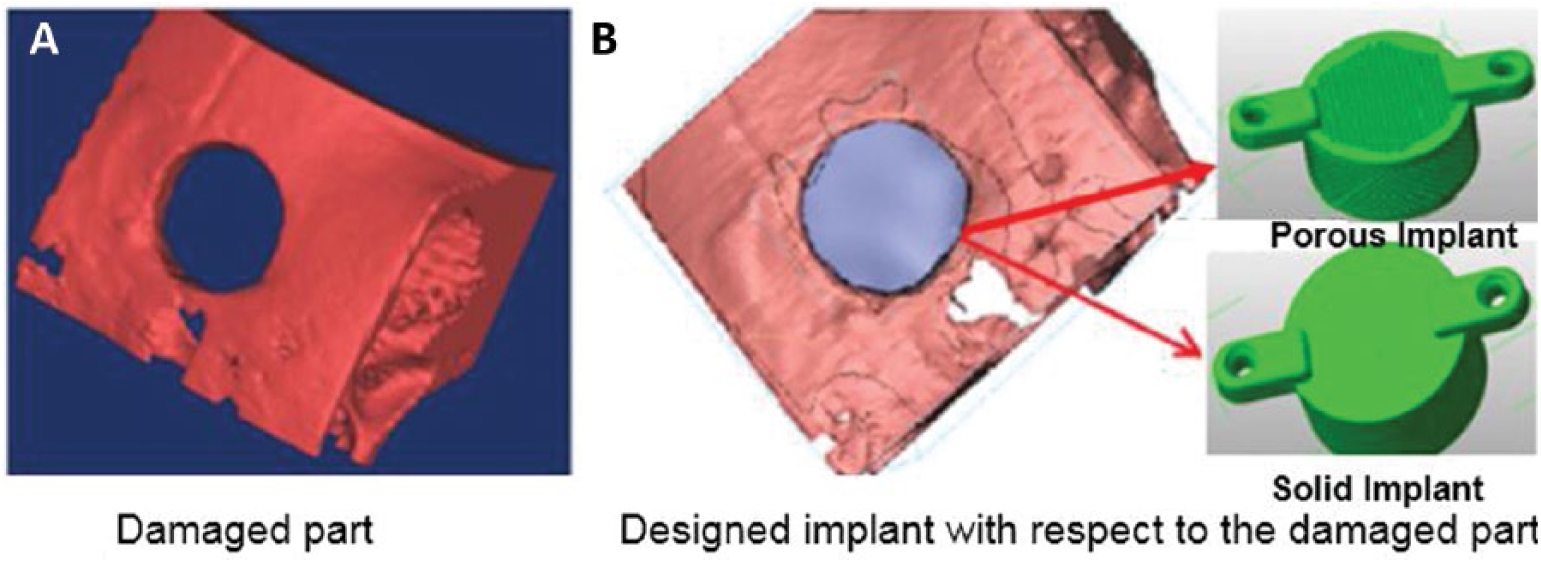
Specific implant designed for the defective region: (
Designs for Massive and transitive implants coherent with the specific damaged region were made. Massive and transitive implants were produced from Ti6Al4V metal powder using the EOS M 280 DMLS equipment. A standard EOS Ti64 powder with a diameter of 30 to 50 μm was used in implant manufacturing. The layer thickness was 30 µm. Optimum processing parameters were obtained from mechanical test results with respect to strength and impact resistance: laser power was 170 W, scanning rate was 1,250 mm/s, scanning distance was 0.07 mm and energy density was 1.94 J/mm2. All implants were manufactured on a single platform using the optimum processing parameters. The ambient temperature was 22.25°C, and relative humidity was 34.65%. The temperature in the manufacturing cabinet was approximately 40°C.
The implants manufactured from Ti6Al4V metal powder were cleaned with compressed air. Images of the manufactured Massive (T1, T2 and T3) and porous (G1, G2 and G3) implants are shown in Figure 5. The implants were subjected to stress relief annealing before cutting from the platform. First they were heated up to 650°C in 1 hour, and then kept at that temperature for 3 hours, before being cooled to room temperature in the oven. Implants specifically designed for the defective region were surgically implanted into the sheep by cranioplasty.

Photographs of porous (G1, G2 and G3) and Massive (T1, T2 and T3) implants.
Ethics statement
The implantation study was carried out on sheep with the approval of the Chairmanship of Suleyman Demirel University Local Ethical Committee on Animal Experiments (B.08.6.YÖK.2.SD.0.05.0.07.00-48), and all experiments were conducted in accordance with Veterinary Faculty Veterinary Hospital, Mehmet Akif Ersoy University, guidelines.
Experimental animal practice
There were 9 sheep involved in the experiment: 3 in the control group, 3 implanted with porous implants and 3 with Massive implants. Implantation was done at the Veterinary Faculty Veterinary Hospital, Mehmet Akif Ersoy University. The sheep were anesthetized with 50 mg/kg intramuscular (IM) ketamine (Ketalar; Pfizer®) + 5 mg/kg IM xylazine (Rompun®). After achieving sufficient anesthetic depth, they were stabilized on the operation desk. The frontal regions of the sheep’s heads were trimmed and disinfected with a 10% povidone-iodine solution (Betadine®). A 3-cm linear cut was made with a no. 15 scalpel blade. Periosteum was reached by blunt dissection and dissected by scalpel. Craniectomy was made on the bone by a knob-tipped sharp tool on which a hand perforator was mounted. Sufficient amount of bone to form a defect was removed by craniectomy. Tissue necrosis due to heat during the craniectomy was minimized by wetting during the operation with loj saline. The wound was washed to remove possible foreign substances, and bleeding was controlled. After completing surgical operations on the bones, the skin was closed with a 3-0 polypropylene suture ensuring that the wound lips would be opposite to each other. The suture region was cleaned with a 10% povidone-iodine solution, and the operation was completed.
CT scans of the sheep were obtained courtesy of the Medicine Faculty Radiology Department, Süleyman Demirel University. The defective cranial regions were converted into 3D format. Except in the control group (n = 3), porous implants (G1, G2 and G3) were stabilized to the defective cranial region of 3 sheep, and Massive implants (T1, T2 and T3) were stabilized to the defective cranial region of another 3 sheep (Figs. 6A and B).
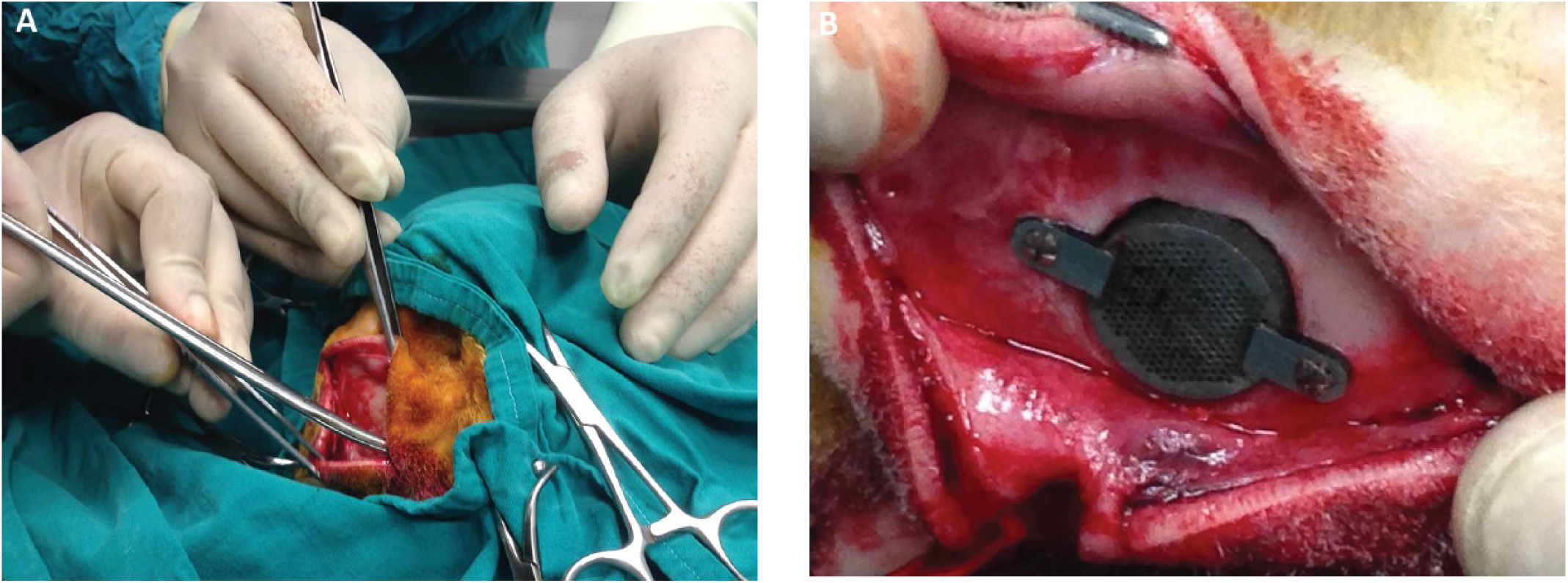
Photograph of implant being applied to the skull of a sheep by surgical procedure: (
The sheep were isolated from each other to protect their skulls after surgery. They were fed with the same amount of food, under same conditions. Twelve weeks were allowed to provide osseointegration.
Results and discussion
After placing Massive and transitive porous implants in experimental animals by surgical operation, they were monitored for 12 weeks by a veterinarian. At the end of the 12 weeks allowed for osseointegration, the sheep were sacrificed, and the implants and a certain amount of bone tissue were removed to investigate implant-bone tissue development. T1, T2 and T3 indices of Massive implants are shown in Figure 7, while G1, G2 and G3 indices of transitive porous implants are shown in Figure 8.
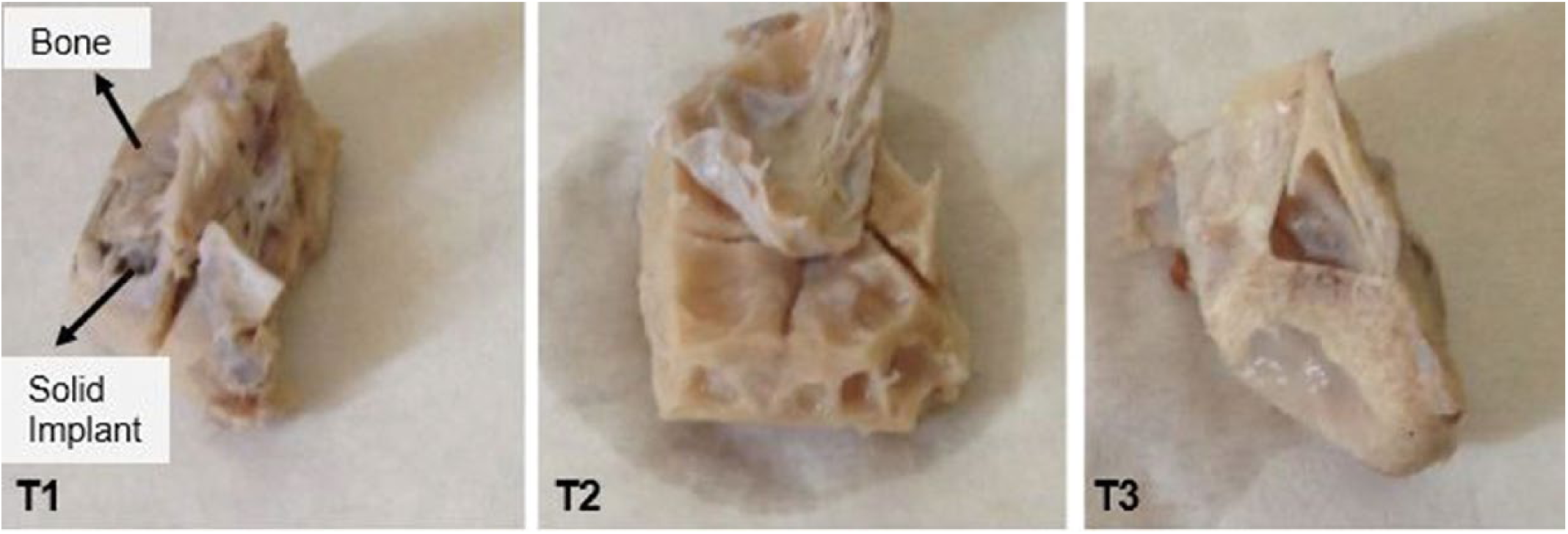
Photographs of Massive implant–bone segments.
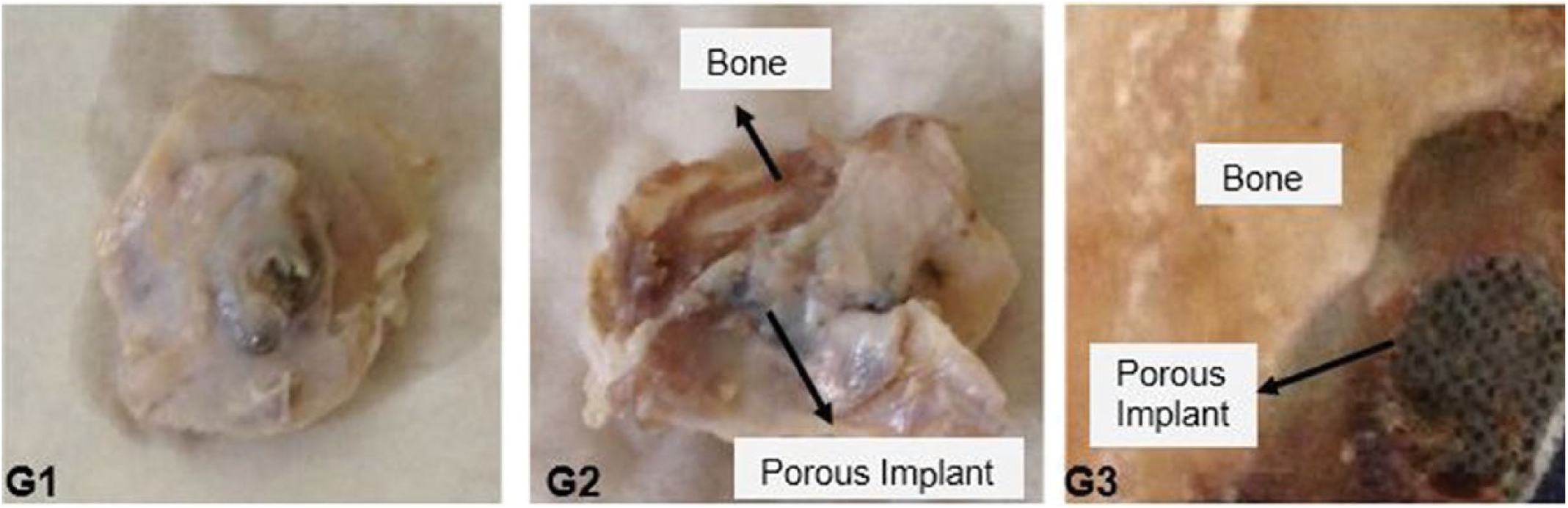
Photograph of porous implant–bone segments.
The implants and surrounding bony tissue were investigated at the Geology Engineering Department Electron Microscopy Laboratory of Hacettepe University. Images were taken with a Carl Zeiss EVO 50 Scanning Electron Microscope. SEM analyses were performed with an accelerated voltage electron beam of 3 to 5 kV, ion beam of 10 kV. Bone–implant integrated segments were evaluated under SEM in terms of material biocompatibility criterion, new bone formation potential and resorption ability.
As indicated in Figure 9A and B, it was observed that cells were concentrated on the surface of Massive implants (T1, T2 and T3). Because the implant was Massive, newly formed bone cells were unable to bond more densely or firmly to the implant. Therefore, osseointegration between implant and bone was less than in the porous implants. The channel structure of the implant increased the bond between bone and implant.
SEM image of Massive implant: (
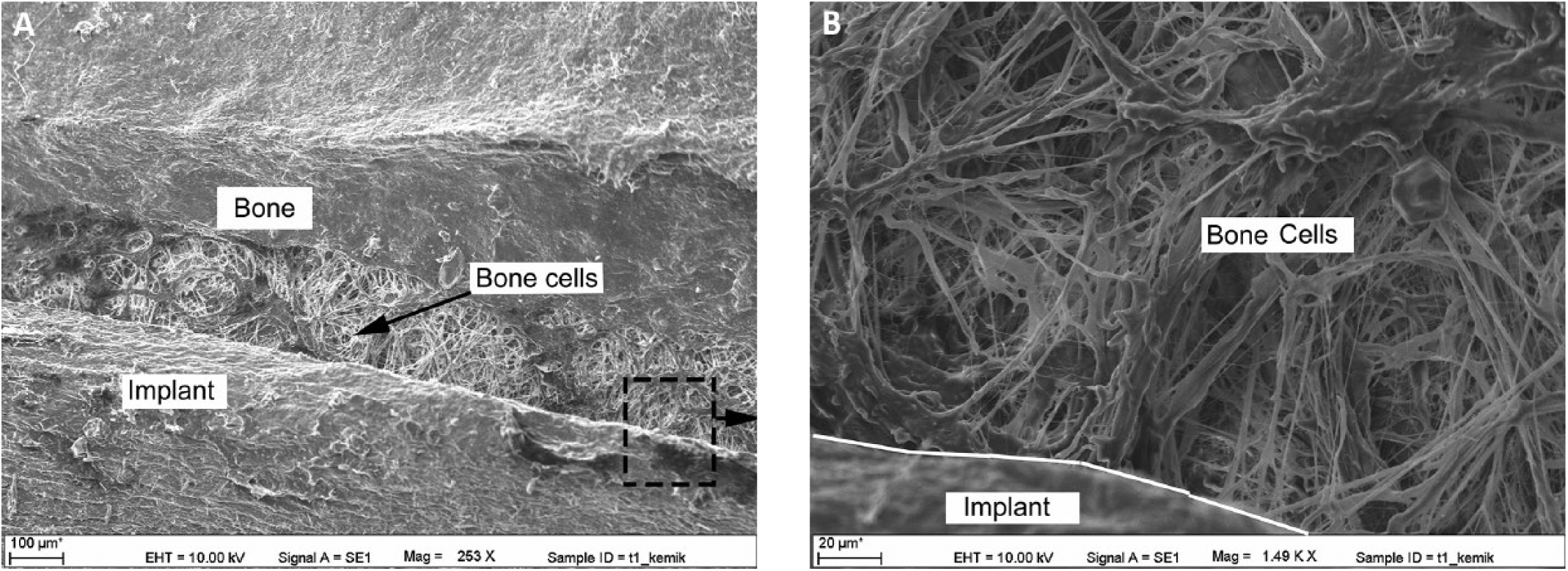
In transitive porous implants, as opposed to Massive implants, an outer region was formed in the bone–implant interface that allowed tissue development. Due to the semispherical and elliptical voids in the outer region of the implant, osteoblast cells moved to the inside of the pore voids (Fig. 10). There was an inner region in which pores grew in the center. It was determined that cells could survive in this inner region. Moreover, the weight of the implant was decreased due to the transitive porosity.

SEM image of cell development channel in the inner region where cells developed of the transitive porous implant.
A magnified cell development channel is shown in Figure 11. Furthermore, images showed that cells inside and around the implant formed hydroxyapatite crystals and covered the implant like a spider web. It was also seen that Ti6Al4V metal powder responded well to, and was compatible with, the body. Functions of tissues between the implant and bone were normal.

Cross-section SEM image of the transitive porous implant.
Bone structures in humans are categorized into 2 groups: cortical (compact) and trabecular (porous). Cortical bones – the main phase – include a minimum 50% porous structure, while trabecular bones have a minimum of 75% porosity. The pore diameter of human bones is between 100 and 500 μm (27). As a result of an integral observation of present porous materials and living tissues, the optimum pore diameter to grow bone is between 300 and 700 µm (28-30). In this study, the designed implant model was transitive porous, growing from the outer to the inner region in accordance with the geometrical structure of the cortical bone of humans, and consistent with descriptions given the literature.
In the literature, porous implant production by additive manufacturing has been a topic of many studies (31-33). In a study conducted by Palmiquist et al (34), Massive and porous implants were manufactured by forming a standard pore using the additive manufacturing method. Bone tissue development was investigated for 6 months after placing implants into experimental animals (sheep). At the end of 6 months, a higher level of osseointegration was achieved for porous implants than Massive ones (34).
In contrast, in the present study, as opposed to standard pores, there were spherical and elliptical pores at the micro level, development channels and an inner region, as shown in Figures 10 and 11. Bone cells developed in the inner region. Transitive pores grown gradually in accordance with the natural structure of the bone were modeled in the inner region for cells to develop. Due to this structure, a strong and durable tissue bond could be formed at the bone–implant interface.
Conclusions
The results of the present study were:
Transitive porous implants were designed to have a geometry similar to that of bone tissue.
The controlled porous (semispherical) structure of the outer region of the implant resulted in the formation of a strong and durable bond between tissues and the implant.
In the inner region, transitive pores grown gradually in accordance with natural structure of the bone were modeled in the inner region for cells to develop, decreasing the weight of the implant.
Strong bone–implant integration in transitive porous implants should increase revision time in both skull and other implants.
Transitive porous implants cannot be produced by any method other than the additive manufacturing method, which makes manufacturing of complex parts easier. It was concluded that it is possible to produce implants having complex geometry from Ti6Al4V metal powders by using the additive manufacturing method.
Transitive porous implants have sufficient quality to be used in all kinds of human and animal applications in engineering and medical sciences.
Footnotes
Acknowledgements
The authors would like to acknowledge the Computer Aided Design – Computer Aided Manufacturing Research and Application Center, Suleyman Demirel University, Isparta, Turkey, for manufacturing the Massive and porous implant specimens, and the Veterinary Faculty, Mehmet Akif Ersoy University, Burdur, Turkey, for surgery on the sheep.
Disclosures
Financial support: This research was supported by Tübitak (113M169), Ankara, Turkey, and by Süleyman Demirel University, BAP (3214-D2-12).
Conflict of interest: None of the authors has any financial interest related to this study to disclose.