Abstract
Background:
Mold fluxes are mainly prepared using cement clinker, quartz, wollastonite, borax, fluorite, soda ash and other mineral materials. Quartz, as one of the most common and essential materials, was chosen for this study to analyze itseffects on crystallization temperature, critical cooling rate, crystal incubation time, crystallization ratio and phases of flux film.
Methods:
We used the research methods of process mineralogy with the application of the single hot thermocouple technique, heat flux simulator, polarizing microscope, X-ray diffraction, etc.
Results:
By increasing the quartz content from 16 mass% to 24 mass%, the crystallization temperature, critical cooling rate and crystallization ratio of flux film decreased, and the crystal incubation time was extended. Meanwhile, the mineralogical structure of the flux film changed, with a large amount of wollastonite precipitation and a significant decrease in the cuspidine content until it reached zero. This showed a steady decline in the heat transfer control capacity of the flux film.
Conclusions:
The reason for the results above is that, by increasing the quartz content, the silicon-oxygen tetrahedron network structure promoted a rise in viscosity and restrained ion migration, inhibiting crystal nucleation and growth, leading to the weakening of the crystallization and a decline in the crystallization ratio.
Introduction
Lubrication and heat transfer properties of continuous casting mold flux, closely related to crystallization behavior and mineralogical structure of the mold flux film, are important to reduce slab surface defects and ensure the smooth operation of the casting process1–3. This is especially true for crystallization properties and mineralogical structure, both of which give perfect control of the melt viscosity and the heat transfer coefficient, making it possible to reduce slab surface defects such as longitudinal cracks, inclusions and breakouts4,5. Therefore, the systematic study of crystallization behavior and the mineralogical structure of the mold flux film is becoming increasingly essential.
Differential thermal analysis is the most common traditional method for studying the crystallization temperature of mold flux. It is based on the thermal effect of the sample during the cooling process and the formation of an exothermic peak on the differential thermal curve, to determine the crystallization temperature of the mold flux. But the crystallization process and crystallization rate of flux film cannot be monitored and observed online. But a device for measuring the crystallization temperature of mold flux based on the single hot thermocouple technique (SHTT) can give data on the mold flux in the production process and show the crystallization process, using image processing technology. Furthermore, the microstructure, mineralogical phase and crystallization rate of flux film can be quantitatively studied with a polarizing microscope.
To date, considerable research on the effects of chemical composition on the crystallization properties of the mold flux film has been conducted. As shown in the studies of the crystalline phase of flux films conducted by Zhu et al 6 and Yang et al 7 , Al2O3 can promote melilite precipitation and inhibit cuspidine precipitation. Han et al 8 noted that moderate amounts of Li2O, Ti2O and Fe2O3 can inhibit wollastonite precipitation and promote melilite precipitation, leading to a decline in the heat transfer control capacity of flux films. Zhao et al 9 changed the ratio of CaO and SiO2 to adjust the basicity of mold fluxes, so that with the rise of basicity, the crystallization rate and the interface thermal resistance of the flux films increased, which resulted in an increase in the thickness of the flux films and an decrease in the heat flux density.
However, the effects of the cement clinker, blast furnace slag and natural minerals of the mold flux on crystallization behavior of the flux films have not been reported in previous studies. Thus, a series of studies of the raw mineral materials and crystallization behavior of mold flux film were conducted by the National Natural Science Foundation of China10–13. In this present study, mold flux samples were prepared with raw materials such as cement clinker, quartz, wollastonite, borax, fluorite and soda ash. The influence of quartz on crystallization behavior and microstructural characteristics of mold flux film were systematically studied using the SHTT, polarizing microscope, X-ray diffraction (XRD), etc.
Materials and methods
Flux preparation and properties test
Experimental mold fluxes were obtained based on various mineral materials, and all of the materials were ground and sieved through 200 mesh. Moreover, on the basis of the common minerals of mold flux and analysis of the chemical components of each raw material as shown in Table 1, experimental schemes of the mold flux were established (Table 2).
Chemical composition of materials of mold fluxes (mass%).
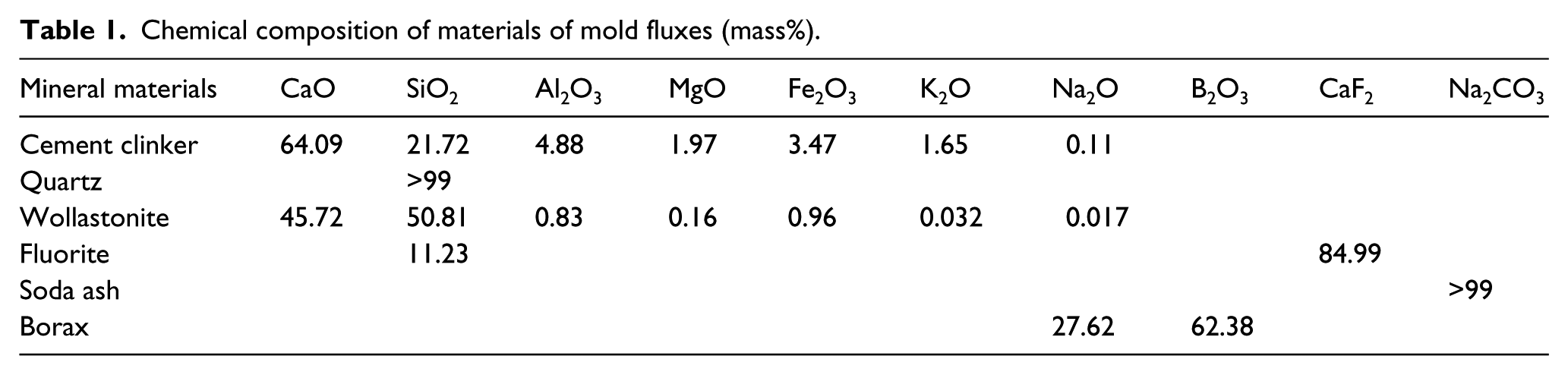
Blending scheme and properties of experimental mold fluxes.
The melting point and viscosity of the samples were measured using an RDS-04 melting property tester (made in China) and HF-201-type viscosity instrument (made in China). As presented in Table 2, the results showed that increasing the amount of quartz and decreasing the cement clinker content led to a gradual rise in the melting point and viscosity of the mold flux.
Establishment of CCT and TTT diagrams
The crystallization behavior of the mold flux was observed to construct the continuous cooling transformation (CCT) diagrams and temperature time transformation (TTT) diagrams using the SHTT. Correlative experiments to characterize continuous cooling crystallization and isothermal crystallization were conducted using the mold fluxes whose material compositions are shown in Table 2.
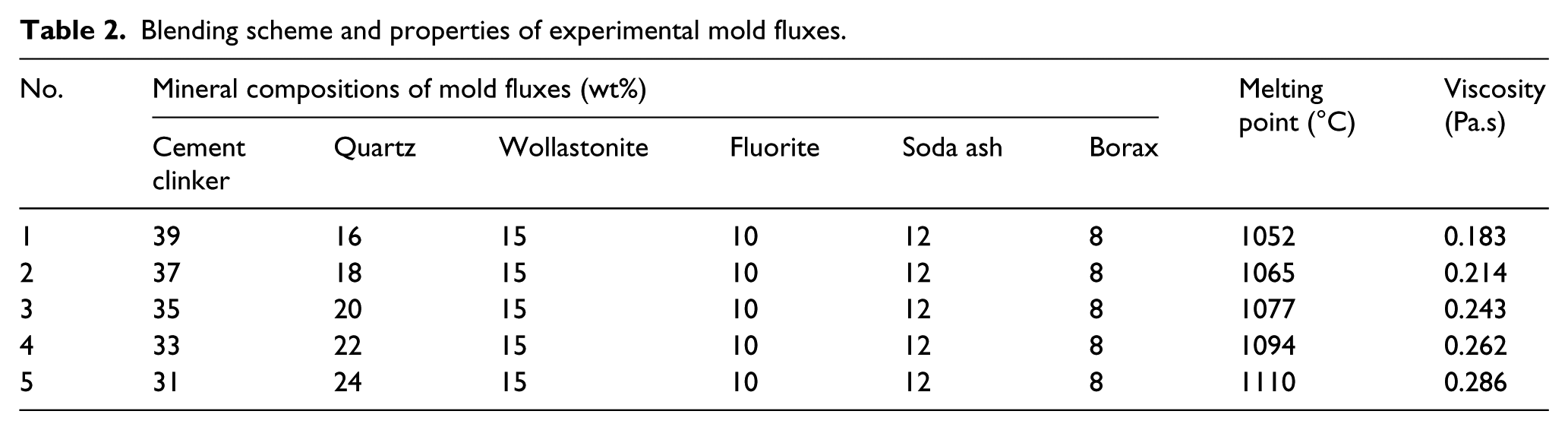
Figure 1a shows the thermal cycle in cooling crystallization experiments for establishing the CCT diagrams. Firstly, the mold flux was heated to 1500°C at the rate of 15°C/s, and melted at 1500°C for 60 seconds to eliminate bubbles and insure the complete melting of the flux. Thereafter, the flux was cooled down to 800°C at different cooling rates, and the whole crystallization process was observed, recording the onset temperature and time of crystallization. The CCT graphs were constructed by recording the crystallization temperature and the onset time at different cooling rates.
(a) Cooling crystallization experiment; (b) isothermal crystallization experiment.
As shown in Figure 1b, the heating mode of the TTT experiments was consistent with that of the CCT. But then after heating to 1500°C for 60 seconds, the flux was cooled to each target temperature in 5 seconds and kept isothermal. In this way to keep isothermal temperature, the graphics data were collected per second by the photographic system, which can be used to judge the start and end time of the crystallization. Thus the TTT diagrams were constructed by recording the onset time of crystallization at different isothermal temperatures.
Flux film preparation and microstructure analysis
The flux films were extracted from the heat flux simulator, and samples were prepared as follows: The graphite crucible was placed in the furnace of the simulator and preheated to 1300°C. Thereafter 350 g of the experimental mold flux was placed into the graphite crucible, and the temperature was set to 1400°C isothermally for 10 minutes until the mold flux melted completely. Then, a water-cooled copper sensor was inserted into the graphite crucible, and the contact time between the copper sensor and the liquid mold flux was recorded by the computer. After 45 seconds, the copper sensor was automatically removed from the graphite crucible, and the flux films that had adhered to the copper sensor were obtained.
A part of the flux film samples were polished and made into slices for observation by polarizing microscope. The mineral compositions and contents, crystallization ratio and microstructure of the flux film were evaluated using an ZEISS Axio Scope.A1 polarized light microscope. Further, another part of the flux film samples was ground and sieved through 400 mesh, and tested by BDX-3200 XRD to verify its mineral composition.
Results and discussion
Effect of quartz on crystallization temperature and critical cooling rate
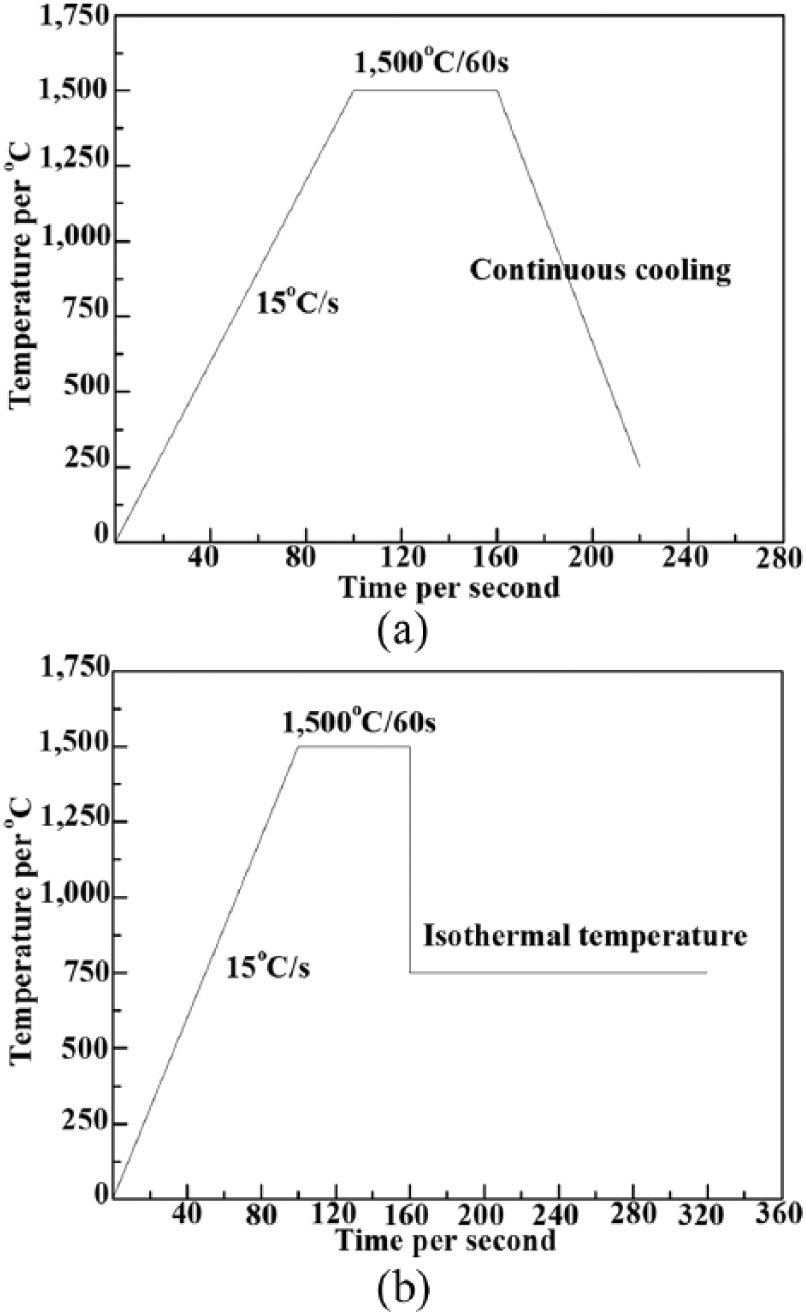
It is well known that the crystallization temperature of the flux film is defined as the onset temperature when it begins to crystallize during the cooling process 14 . The crystallization temperature of a mold flux is a function of the material composition and cooling rate 15 . Generally speaking, measurements or comparisons of crystallization temperatures of mold fluxes with different components, are usually determined under the same cooling conditions 16 . CCT diagrams of experimental mold fluxes are shown in Figure 2a. The relationship between quartz content and critical cooling rate shown in Figure 2b was determined from the maximum cooling rate indicated in Figure 2a.
(a) Continuous cooling transformation (CCT) of fluxes with 16%-24% quartz; (b) relation between quartz content and critical cooling rate.
The results presented in Figure 2a show that the crystallization temperature decreased with an increase of the cooling rate for the same mold flux and decreased with increasing quartz content under the same cooling rate. As can be seen from Figure 2b, the critical cooling rate curve exhibits a downward trend with an increase in quartz content. By adding an additional 2% of quartz content in the mold flux, the critical cooling rate dropped 3°C/s on average. Consequently, the driving force for crystallization of the flux film was weakened, resulting in poor crystallization ability.
Effect of quartz on crystal incubation time
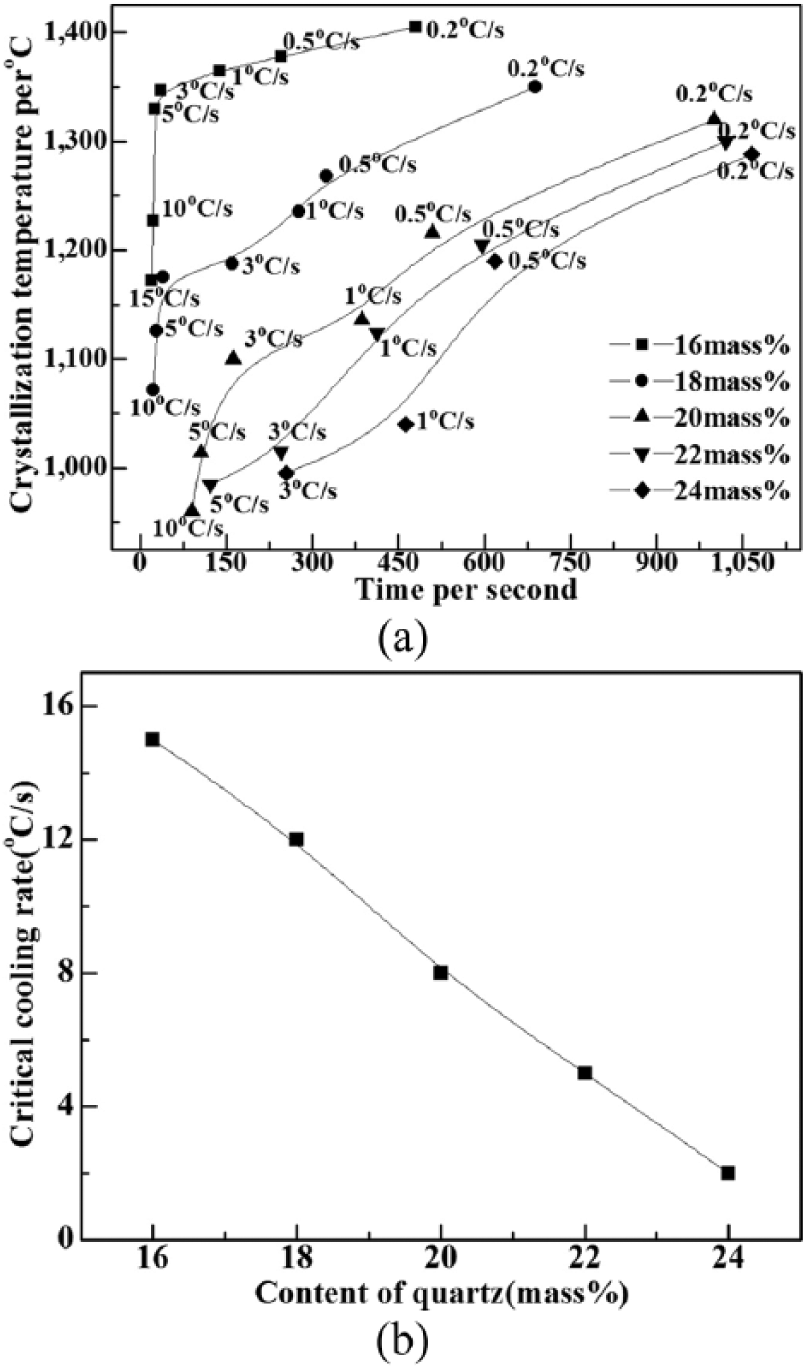
The crystallization rate and the rate of crystalline growth can be characterized by the crystal incubation time. In general, a shorter crystallization incubation time leads to a faster crystalline growth and nucleation rate. Crystallization starting curves for TTT generated based on isothermal crystallization experiments are presented in Figure 3. This shows that the TTT curves of mold fluxes shifted in the direction of an extended crystal incubation time and the crystallization temperatures dropped with the increase of quartz content from 16 mass% to 24 mass%.
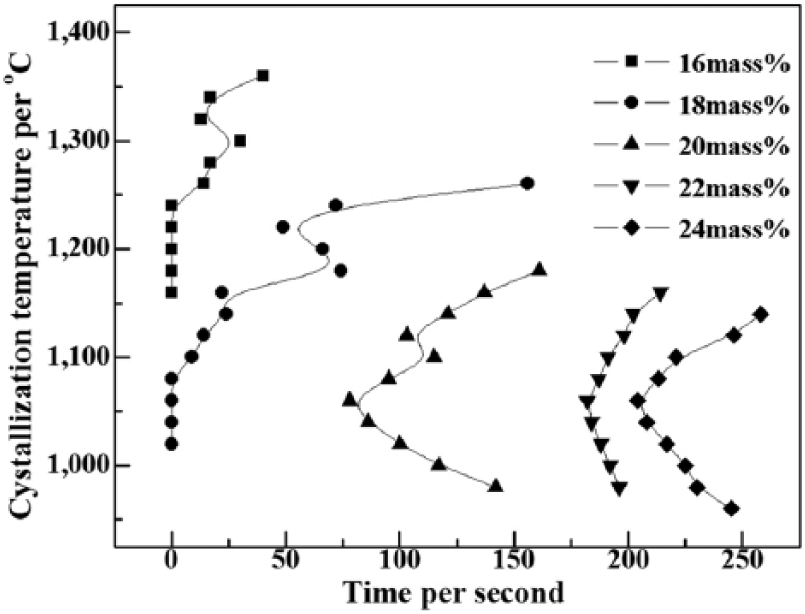
Temperature time transformation (TTT) of mold fluxes with 16%-24% quartz.
The TTT curves indicate that the crystal incubation time was extended by about 60 seconds by adding an additional 2% quartz content, and the crystallization temperature clearly dropped from 1370°C to 1140°C. Furthermore, The TTT curves of the samples appear in the form of 2
Effect of quartz on crystallization ratio and phases
The microstructure of the flux film plays a significant role in the thermal resistance of the flux film and the heat flux in the mold17,18. The mineral composition, phase content and crystallization rate of flux films were evaluated using an ZEISS Axio Scope.A1 polarized light microscope; the results are presented in Table 3. It was observed that the main phases of the flux films were wollastonite and cuspidine, and that the crystallization ratio of flux films and volume percentage of crystalline minerals had a great magnitude of range with increasing quartz content.
Mineralogical composition, proportion and crystallization ratio of flux films.
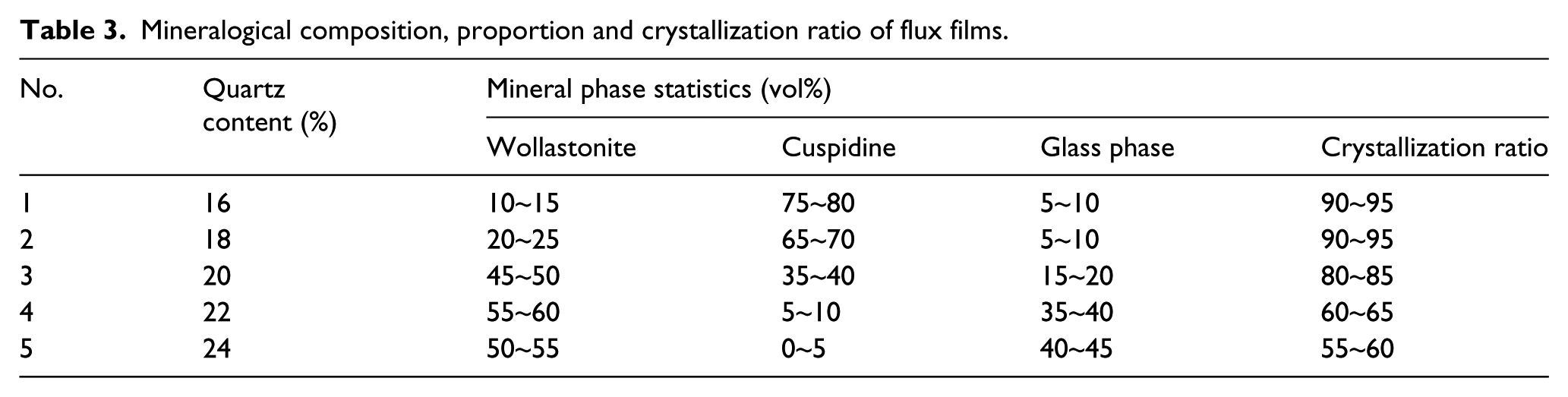
When the quartz content of the mold flux was less than 18%, the crystallization ratio of the flux films was maintained at 90%~95%. Two types of crystalline mineral (wollastonite and cuspidine) appeared in the flux films, and cuspidine was the dominating phase, which accounted for 65%~75% of the flux films. Moreover, there was also a small amount of glass phase in these flux films. When the quartz content of the mold flux was more than 20%, the cuspidine content of the flux films gradually decreased until it disappeared, and crystalline phases began to give priority to wollastonite. which was consistent with the results of the XRD analysis indicated in Figure 4. The content of crystalline minerals changed by a great magnitude, indicating that the increase in quartz exerted a significant effect on promoting wollastonite precipitation and in inhibiting the generation of cuspidine.
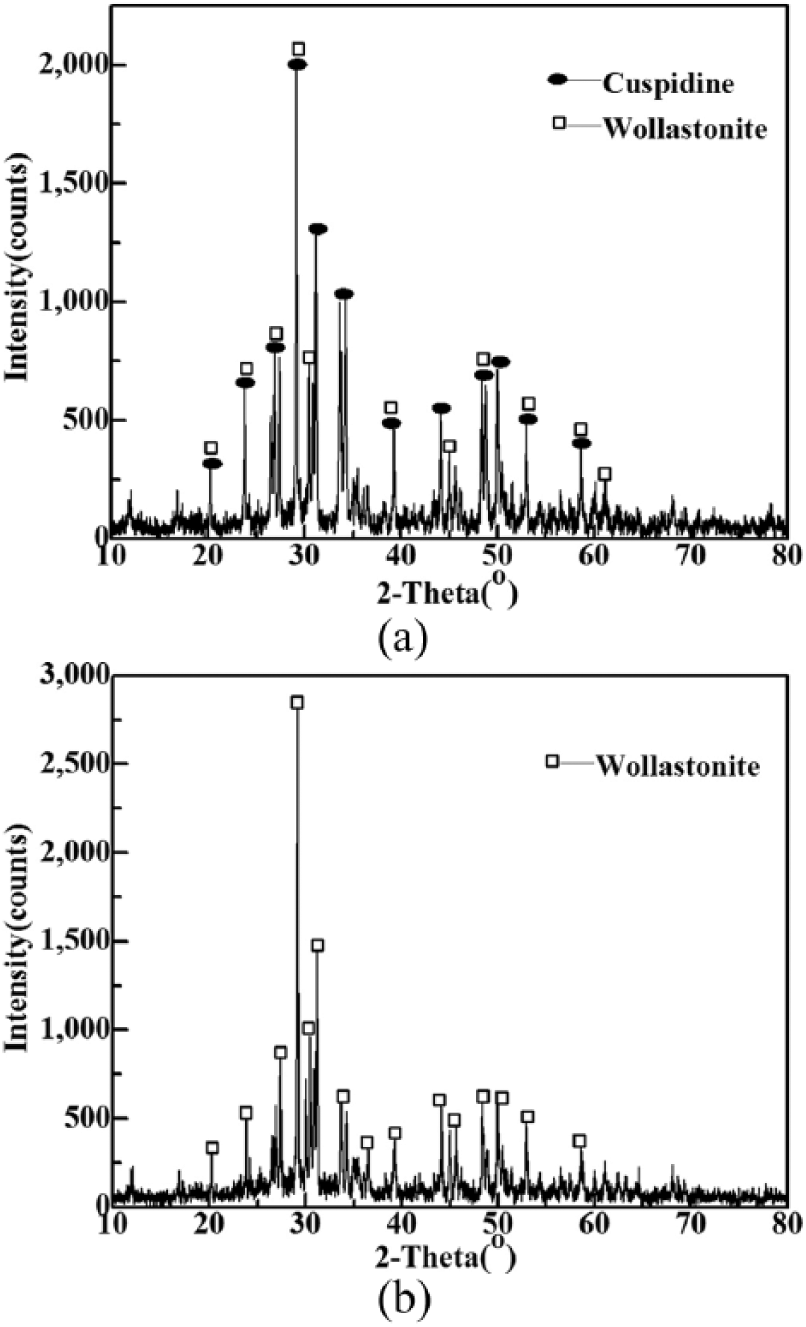
X-ray diffraction (XRD) patterns of flux films for quartz series. (a) Flux film of sample 2; (b) flux film of sample 4.
As is presented in Figures 5 and 6, mineral modality, particle size and microstructure of flux films were measured using an ZEISS Axio Scope.A1 polarized light microscope. When the quartz content of the mold flux was less than 18%, the main mineral crystals of the flux films were granular and spearhead-shaped cuspidine, and there was only a small amount of short columnar-shaped wollastonite in the flux films (Figure 5a and b). In contrast, the degree of development of wollastonite in sample 2 was better than that in sample 1, and this showed that the crystallization of wollastonite can be promoted by adding quartz content. When the quartz content of the mold flux was more than 20%, the cuspidine crystals were inhibited, and the main modalities appeared to be granular or spearhead shaped in local areas (Figure 6a and b), whereas the crystal modalities of wollastonite were more prone to be fibrous shaped or leaf shaped (Figure 6c and d).

ZEISS Axio Scope.A1 polarized light microscope images of microstructures of flux films with different quartz contents. (a) Structure of flux film of sample 1; (b) structure of flux film of sample 2; (c) structure of flux film of sample 3; (d) structure of flux film of sample 4.

ZEISS Axio Scope.A1 polarized light microscope images of minerals and morphological characters of flux films with different quartz content. (a) Cuspidine with granular shape; (b) cuspidine with spearhead shape; (c) wollastonite with fibrous shape; (d) wollastonite with leaf shape.
According to the above analysis, with the quartz content of the mold flux increasing from 16 mass% to 24 mass%, the crystal incubation time was prolonged, and the crystallization ability of the flux film weakened, whereas the melting point and lubrication performance were enhanced. When the quartz content exceeded 20 mass%, the crystallization ratio and the cuspidine content of the flux films clearly decreased, thereby resulting in a reduction of the interfacial thermal resistance of the flux films, which is adverse to reduce the longitudinal cracks on slab surface. Consequently, the quartz content of mold flux should be controlled no more than 20 mass%, to achieve the balance of heat transfer and lubrication of flux film.
Conclusions
The crystallization temperature of a mold flux decreases with increasing quartz content under the same cooling rate. The critical cooling rate of the mold flux presents a downward trend, and the crystal incubation time is decreased with increasing quartz content from 16 mass% to 24 mass%. Moreover with the addition of 2% quartz content, the melting point and viscosity of the mold flux are elevated by 14.5°C and 0.026 Pa·s on average.
Quartz has a significant effect on restraining cuspidine and generating wollastonite, resulting in a reduction of the interfacial thermal resistance of flux films. When the quartz content exceeds 20 mass%, the crystallization ratio and the cuspidine content of flux films clearly decrease, which is adverse to reduce the longitudinal cracks on slab surface.
To achieve the balance of heat transfer and lubrication of mold flux films during the casting process, a moderate increase in the quartz content (less than 20 mass%) in the mold flux is suggested for crack-sensitive steel slab casting.
Footnotes
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Natural Science Foundation of China (no. 51774140, no. 51174073 and no. U1360106) and the Science and Technology Research Project for the Higher School of Hebei Province of China (no. QN2016126).
