Abstract
Background
The objective of the present study was to compare solubility and pH of 6 direct pulp capping materials.
Methods
Specimens of each material – i.e., Dycal, Calcicur, Calcimol LC, TheraCal LC, MTA Angelus and ProRoot MTA – were prepared and immersed in water. Solubility was determined after 24 hours and 2 months and analyzed statistically using a 1-way ANOVA and post hoc Tukey test. pH values were measured 3 and 24 hours after manipulation.
Results
All direct pulp capping materials showed low solubility; the pH of tested materials ranged from 10 to 12 and showed a nonsignificant increase/reduction after 24 hours.
Conclusions
Within the limitations of this in vitro study, the direct pulp capping materials studied showed different solubility even if no changes were recorded over time. All of the materials showed a very alkaline pH.
Introduction
Direct pulp capping involves the application of a dental material to the exposed pulp in an attempt to act as a barrier, protect the dental pulp complex and preserve its vitality (1). The ultimate goal of direct pulp capping materials is the induction of hard tissue formation by pulp cells (2). Materials used in the vital pulp treatment must be placed in contact with the pulp tissue (3, 4), so they should be nontoxic and biocompatible; they should also provide good sealing (5, 6) and low solubility (7, 8). The last may have a great impact on the success rate of the restorative procedure. Calcium hydroxide–based and calcium oxide–based materials are the most popular materials for direct pulp capping because of their ability to release hydroxyl (OH) and calcium (Ca) ions upon dissolution (9, 10). Unfortunately, these materials are soluble and increase local pH with the formation of a necrotic layer at the material–pulp interface (11, 12). Dycal® (Dentsply Tulsa Dental, Johnson City, TN, USA) is a self-setting, radiopaque calcium hydroxide–based material (13). Its alkaline pH stimulates the formation of secondary dentine when the material is in direct contact with the pulp. Calcicur® (Voco GmbH, Cuxhaven, Germany) is a water-based calcium hydroxide paste with a high pH (12.5). Calcimol LC® (Voco GmbH, Cuxhaven, Germany) is a light-curing radiopaque single-component material containing calcium hydroxide. TheraCal LC® (Bisco Inc., Schaumburg, IL, USA) is a light-cured resin-modified calcium silicate–filled liner material containing approximately 45% calcium silicate particles (type III Portland cement) and 45% resin. Radiopaque Portland cements, commonly named mineral trioxide aggregate (MTA) cements (ProRoot MTA®; Dentsply Tulsa Dental, Johnson City, TN, USA; and MTA Angelus®; Angelus, Londrina, PR, Brazil), are therapeutic, endodontic repair calcium silicate materials. MTA is a powder that contains trioxides and hydrophilic particles, which sets in the presence of moisture (14). Since its introduction in dentistry, MTA has been the focus of several studies aiming to describe its components and properties (15). These studies allowed the indications of MTA to be extended beyond its original purpose as a material to seal perforations, to its use as a retrograde filling material (16), also as a pulp capping cement in pulpotomies (17). MTA cements favor the mineralization process in human dental pulp cells, thus having the potential to be used as capping materials (18). Several studies on MTA have demonstrated that this material possesses many ideal properties: low solubility, low cytotoxicity, tissue biocompatibility and ability to induce mineralized tissue formation (19). According to the manufacturer (20), ProRoot MTA® (Dentsply Tulsa Dental) is composed of 75% Portland cement clinker, 5% dehydrated calcium sulfate (gypsum) and 20% bismuth oxide. Conversely, white ProRoot-MTA® (Dentsply Tulsa Dental) contains 80% Portland cement clinker and 20% bismuth oxide, while MTA Angelus® (Angelus) is produced by adding bismuth oxide to white Portland cement clinker to improve radiopacity (21) – it does not contain calcium sulfate, to improve the setting time. This cement shows alkaline pH and calcium ion release, similar to ProRoot MTA® (Dentsply Tulsa Dental) (22). Despite the good physicochemical and biological properties, MTA cements present some shortcomings – i.e., the long setting time, which may favor the solubility and the displacement of the applied material. The alkalinizing power of a pulp capping material represents a key property: the release of hydroxyl ions during the hydration reaction creates an adverse environment for bacterial survival and proliferation (23, 24). In addition, alkaline pH is known to cause an inflammatory reaction with the formation of reparative dentin and favors the formation of hydroxyapatite (25). The objective of the present in vitro study was to evaluate for each pulp capping material (Dycal® from Dentsply Tulsa Dental; Calcicur® from Voco GmbH; Calcimol LC® from Voco GmbH; TheraCal LC® from Bisco Inc.; MTA Angelus® from Angelus; and ProRoot MTA® from Dentsply Tulsa Dental) the solubility at 24 hours and at 2 months after the preparation of the specimens; while pH changes were verified at 3 and 24 hours after preparation.
Materials and Methods
Solubility Test
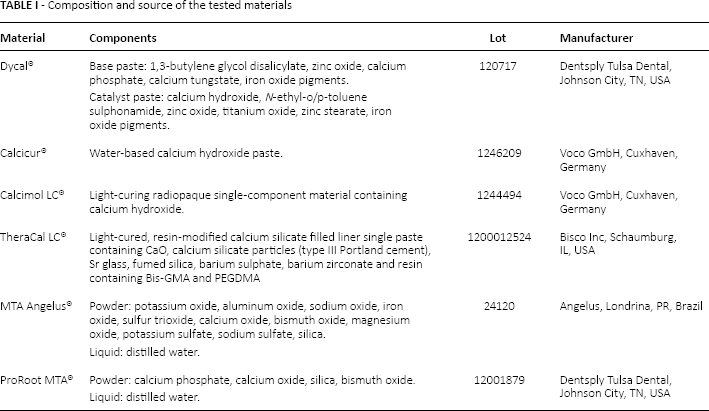
Six pulp capping materials were selected for this study: Dycal® (Dentsply Tulsa Dental), Calcicur® (Voco GmbH), Calcimol LC® (Voco GmbH), TheraCal LC® (Bisco Inc), MTA Angelus® (Angelus) and ProRoot MTA® (Dentsply Tulsa Dental). The components of each material and its manufacturer are reported in Table I.
Composition and source of the tested materials
Stainless steel ring moulds with an internal diameter of 20 ± 0.1 mm and a height of 1.5 ± 0.1 mm were used for sample preparation. All moulds were cleaned with acetone in an ultrasound bath for 15 minutes. All moulds were weighed 3 times prior to use (accuracy ± 0.0001 g) on a Mettler AE-163 (Mettler Toledo) balance, which was used throughout the experiment. The moulds were placed on a glass plate and filled to slight excess with the mixed materials. The same operator mixed all materials in accordance with the manufacturers’ instructions. After filling the moulds, another glass plate covered with a Mylar strip was placed on top of the moulds, exerting a light pressure in order to remove any excess. Dycal® (Dentsply Tulsa Dental), Calcicur® (Voco GmbH), MTA Angelus® (Angelus) and ProRoot MTA® (Dentsply Tulsa Dental) samples were cured and left to set on a grating in a cabinet at 37°C and 100% relative humidity. Calcimol LC® (Voco GmbH) and TheraCal LC® (Bisco Inc) samples were cured with a LED curing light unit (Celalux 2 High-Power LED curing-light; Voco GmbH, Cuxhaven, Germany) for 20 seconds on both surfaces at a light intensity of 1,700 mW/cm2. Six sets of specimens for each material were prepared in 1 operation. All samples were left to set for 24 hours on a grating in a cabinet at 37°C and 100% relative humidity. The samples in their moulds were then exposed to air for 15 minutes and weighed 3 times, and then the average reading was recorded to 3 decimal places. The specimens of each material were individually placed in tarred bottles, containing 5 mL of distilled water. The bottles were then transferred to an oven at 37°C where they remained for 24 hours. They were removed from the oven and rinsed with distilled water, which was then collected in the same bottle. The water was evaporated at a temperature slightly below boiling point. Bottles and residues were dried in an oven at 105°C, cooled in a desiccator and weighed. The differences found between this weight and the original bottle weight were divided by the initial dry weight of the specimens and multiplied by 100. The result was recorded as solubility (26, 27). The solubility test was performed again at 2 months using the same method. The solubility of the set pulp capping materials should not exceed 3% mass fraction (ISO 6876 clause 4.3.6). Results were analyzed by analysis of variance (ANOVA). A statistically significant difference was set at a p value of <0.05.
Differences in solubility percentages were calculated, and a statistical analysis was performed with computer software (Stata 7; Stata Corp., College Station, TX, USA). To assess the normality of the distributions of the means, the Kolmogorov-Smirnov test was applied; then ANOVA was used to determine whether significant differences existed among the groups. For the post hoc test, the Tukey test was used. Significance for all statistical tests was set at a p value of <0.05.
To determine whether time influenced the solubility of the pulp capping materials, an analysis of longitudinal data was performed using the t-test for paired data (p<0.05).
pH Measurements
Each specimen was mixed and placed onto cylindrical Teflon moulds of 2-mm height and 10-mm diameter. Each sample was placed into a separate vial containing 10 mL of distilled water. The samples were stored at 37°C, and pH measurement was performed after incubation of 3 and 24 hours, because, as reported below, the samples showed a tendency toward a reduction in their ability to raise the pH value. The pH value was measured by a digital pH meter (HI 2210 pH Meter; Hanna Instruments, USA), previously calibrated (Leouvakul Dental Materials). Six samples were prepared for each group, and the Tukey test was applied to determine whether significant differences existed in pH values after 3 and 24 hours of incubation. To determine whether time influenced the pH values of the pulp capping materials, an analysis of longitudinal data was performed using the t-test for paired data (p<0.05) between different times of incubation (3 and 24 hours).
Results
The results of the solubility test (after 24 hours and 2 months) are listed in Table II. All of the materials fulfilled the requirements of ISO 6876, demonstrating a weight loss of less than 3%. Dycal® (Dentsply Tulsa Dental) and Calcicur® (Voco GmbH) provided the lowest solubility after both 24 hours and 2 months. There was no statistically significant difference in solubility among the other materials tested after 24 hours (p>0.05). Similar results were obtained after 2 months. For each pulp capping material, the weight loss after 2 months was not significantly different from the weight loss after 24 hours: the materials tested provided a low solubility over time.
Solubility after 24 hours and 2 months with standard deviation (SD), of weight loss for each material and for each immersion period
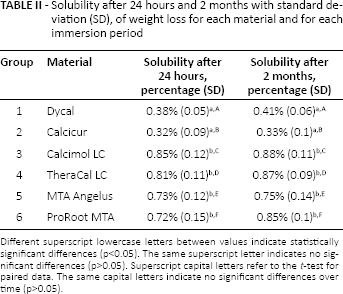
Different superscript lowercase letters between values indicate statistically significant differences (p<0.05). The same superscript letter indicates no significant differences (p>0.05). Superscript capital letters refer to the t-test for paired data. The same capital letters indicate no significant differences over time (p>0.05).
All of the materials tested provided a very alkaline pH after 3 hours (Tab. III); ProRoot MTA® (Dentsply Tulsa Dental) showed the highest value among the materials tested. For Dycal® (Dentsply Tulsa Dental), Theracal LC® (Bisco Inc), MTA Angelus® (Angelus) and ProRoot MTA® (Dentsply Tulsa Dental) a nonsignificant reduction in pH value was recorded after 24 hours (p>0.05); Calcicur® (Voco GmbH) and Calcimol LC® (Voco GmbH) showed a nonsignificant increase in pH value after 24 hours.
pH of the tested materials at 3 and 24 hours
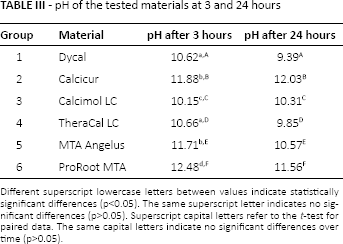
Different superscript lowercase letters between values indicate statistically significant differences (p<0.05). The same superscript letter indicates no significant differences (p>0.05). Superscript capital letters refer to the t-test for paired data. The same capital letters indicate no significant differences over time (p>0.05).
Discussion
Direct pulp capping is the treatment of the exposed vital pulp by sealing the pulpal wound with a dental material to induce a reparative dentinogenic response (23). A pulp capping material must act as a barrier and induce the formation of a new dentine bridge or dentine-like bridge between the pulp and restorative material. In the present study, Dycal® (Dentsply Tulsa Dental), Calcicur® (Voco GmbH), Calcimol LC® (Voco GmbH), TheraCal LC® (Bisco Inc.), MTA Angelus® (Angelus) and ProRoot MTA® (Dentsply Tulsa Dental) were evaluated as direct pulp capping materials. These materials are generally covered with a restorative material which prevents them from direct contact with oral fluids; however, solubility is a very important factor in assessing the suitability of potential substances to be used as liners. One of the major drawbacks of Ca(OH)2-based and CaO-based materials is high solubility and dissolution over time (within 1-2 years after application) in tissue fluids. This leads to the disappearance of the material and the formation of defects in reparative dentin underneath the capping, thereby failing to provide a permanent seal against bacterial invasion (28, 29).
A further problem with calcium hydroxide–containing cements as stated by the literature is that for a material such as calcium hydroxide to be able to exert a biological effect, it must, to some extent, solubilize and dissociate (19). On the contrary, a direct pulp capping material should theoretically provide a hermetic seal of the tubules. However, the literature has shown that a material can dissociate and have reasonable solubility values (27), providing a seal and even adequate biological response (30, 31). ProRoot MTA® (Dentsply Tulsa Dental) and MTA Angelus® (Angelus) produced the highest amount of precipitates, which could have positively influenced the formation of an interfacial layer with tag-like structures that was favored by the greater amount of calcium ions released by the MTA cements (19). Gandolfi et al (23) states that TheraCal LC® (Bisco Inc.) had low solubility, while ProRoot MTA® (Dentsply Tulsa Dental) provided high solubility due to longer setting time. This present study indicated that Dycal® (Dentsply Tulsa Dental), Calcicur® (Voco GmbH), Calcimol LC® (Voco GmbH), TheraCal LC® (Bisco Inc), MTA Angelus® (Angelus) and ProRoot MTA® (Dentsply Tulsa Dental) showed little signs of solubility in water. As for ProRoot MTA® (Dentsply Tulsa Dental), our results are in accordance with the studies of Torabinejad and Parirokh (6) and Danesh et al (32). In contrast, Fridland and Rosado (26) demonstrated with the same method that MTA is capable of partially releasing its soluble fraction to an aqueous environment over a long period of time at a decreasing rate. The authors also confirmed that this soluble fraction is mainly composed of calcium hydroxide, which yields high pH values. According to Vivan et al (19), the MTA Angelus® (Angelus) cements had the highest solubility, while in the present study it was demonstrated that these had low solubility. These differences in solubility rates could be linked to different conditions of mixing and curing. As reported by Torabinejad et al (30) and Fridland and Rosado (27), the characteristics of the resultant set material are likely to be dependent on various factors including water to powder ratio, temperature, environmental humidity and pH, entrapped air and water, the rate of packing and the condensation pressure applied. These aspects are highly variable among the studies because the manufacturer's instructions do not give any information for them. Because of this lack of standardization and use of uncontrolled hand placement methods, the results obtained may be inconsistent.
For the pH measurements, all cements were immersed in water immediately after manipulation (33), and they showed a tendency to a reduction in their ability to raise the pH of most materials between 3 and 24 hours. This is the reason pH measurements were performed only after 3 and 24 hours in the present study (34). Torabinejad et al (35) measured the pH value of gray ProRoot MTA® (Dentsply Tulsa Dental) at 3 hours (12.5), and they state that the peak of alkalinity was reached after 3 hours. This fact confirms what we found about the pH level after 24 hours. The alkalinity reached the top level after about 3 hours, as also found by Torabinejad et al (35), and then it maintained just lower values for a long time. As for Ca(OH)2-based and CaO-based materials, the literature gives contrasting values for the maximum alkalinity reached after manipulation. All of the cements evaluated in this study showed a pH value between 10 and 12 by the release of hydroxyl ions after 3 hours.
Regarding the setting time property, cements with long setting times are more susceptible to dissolution during endodontic surgery, whereas extremely short setting times may represent technical difficulties during clinical application. The literature shows initial and final setting times for ProRoot MTA® (Dentsply Tulsa Dental) of 40 and 140 minutes, respectively (36), which are different than those observed for MTA Angelus® (Angelus) cement by Bortoluzzi et al (20) (12 and 48 minutes, respectively). This parameter did not affect the ability to maintain a very alkaline pH after 24 hours for both ProRoot MTA® (Dentsply Tulsa Dental) and MTA Angelus® (Angelus).
Limitations and Conclusions
The problems encountered in defining the phase of preparation of the specimens could have led to some bias in the results obtained. However, to identify the percentage of solubility, the boiling phase was considered accurate to eliminate water and maintain the material, which is different from other systems such as freezing or drying, which can also be less replicable. Moreover, the boiling method should prevent the formation of supernatants – that is, the liquid that covers material deposited by settling, precipitation or centrifugation. Nevertheless, the solubility of pulp capping materials could be tested with centrifugation, by taking into account the effects of supernatants. In conclusion, the solubility of pulp capping materials seems to be minimal over time, as confirmed by the results at 2 months; but it is quite pronounced in the first 24 hours, thus “making it essential that clinicians prepare and handle the material in accordance with the manufacturer's instructions.
Footnotes
Financial support: The authors did not receive any financial support.
Conflict of interest: Nothing to declare.
