Abstract
Background:
Silicon carbide ceramics obtained by reactive infiltration of silicon (SRI) have many industrial applications especially involving severe and high temperature conditions. In this study, the oxidation behavior in air of Si-SiC-ZrB2 systems at a high temperature (1500°C) for dwelling times of up to 48 hours was examined.
Methods:
The oxidation process was analyzed on the basis of elemental maps and X-ray diffraction patterns taken, respectively, on the core and on the surface of the specimens, together with weight gains and the average thicknesses of the resulting scale. Further, flexural strength at room temperature was examined as a function of different oxidation times.
Results:
The main chemical reactions and phase transformations involved in the oxidation process are reported. Several oxides were detected on the surface: zirconia, silica, zircon and 3-zirconium monoxide. All of the samples showed a parabolic oxidation kinetics, suggesting that the controlling mechanism was the diffusion; however, even after 48 hours, the oxidation process was not finished – indeed, all of the samples continued to gain weight.
Conclusions:
The oxidation of Si-SiC-ZrB2 material produced via SRI was slower compared with previously investigated ZrB2-SiC composites processed with a different techniques and tested in similar conditions. The oxidation mechanism was found to be consistent with the convection cells model.
Introduction
Ultra-high temperature ceramics (UHTCs) are a class of materials which show high stability in harsh environments involving high temperatures. They include many transition metals such as carbides, borides and nitrides; refractory metals (such as Hf, Nb, Ir, Ta) and oxides (such as HfO2, ZrO2, UO2). UHTCs show melting temperatures higher than 3000°C, good thermomechanical properties and good thermal stability, which allow them to be used continuously at temperatures higher than 1600°C (1). Scientific interest in UHTCs has substantially increased in recent decades; they are very promising materials, especially for aerospace applications such as thermal protection systems (TPSs) and the hot parts of reusable atmospheric re-entry vehicles. The latter require materials showing a good oxidation resistance at temperatures usually higher than 1500°C to be safer and enable faster re-entry. Multiphase boride and silicon carbide ceramics, including ZrB2-SiC-Si ceramics, are promising materials for these kind of applications (2-4). In addition, these materials find other applications such as in porous burners and solar receivers (5).
The processing methods for diboride often involve high temperatures and/or high pressures to obtain highly dense materials. Their high melting points and the strong covalent nature of their bonds make the densification process difficult to complete. Different techniques can be used for their sintering, such as hot pressing, hot isostatic pressing, pressureless sintering, self-propagating high-temperature synthesis or spark plasma sintering. All of these involve temperatures higher than 1800°C required for the activation of the grain boundary diffusion mechanism (6, 7).
A relatively new way to obtain nearly fully dense composite materials using a “mild process” is silicon reactive infiltration (SRI), which was originally investigated by Hillig et al (8). Reaction-bonded SiC ceramics show low synthesizing temperatures, short processing times and reduced dimensional changes induced by the densification process (near-net shaping) with respect to those observed using both sintered without pressure and hot-pressed sintered SiC. Moreover, the production of reaction-bonded SiC does not require fine and high purity SiC starting powders; the low raw materials cost represents a considerable advantage from an economic point of view. The main drawback of these techniques is the presence of unreacted silicon in the sintered material: to obtain a complete and rapid infiltration of the preforms, the volume of their porosity has to be higher with respect to the stoichiometric amount of Si required to react with carbon. For this reason, an excess of silicon, in the range from 10 to 20 vol% is present to fill the pores, thus yielding a nonporous body.
The oxidation behavior of ZrB2 and its composites is a key factor in aerospace applications because it can cause a dramatic worsening of the materials’ properties and then critical damage to the vehicle itself (9-13).
In our previous work (14), we investigated the properties of Si-SiC-ZrB2 materials after short oxidation times. They showed the following layered structure: a surface borosilicate layer rich in silica, an underlying borosilicate glassy layer including ZrO2 particles (BSZ) and the unoxidized bulk material. It should be noted that the formation of the “SiC-depleted region,” which many authors report for oxidized ZrB2-SiC materials, was not observed (14-16).
In the present work, the properties (such as bending strength) of Si-SiC-ZrB2 materials produced via SRI and subjected to high temperature and oxidative conditions for long oxidation times were investigated.
Owing to the presence of unreacted free silicon in the matrix, the main target applications of these multiphase materials are currently foreseen as a coating or as a matrix in fiber-reinforced composites in a specific range of temperatures (1400°C-1600°C) or in applications where no mechanical loads are applied (e.g., TPSs or cellular ceramics) (5).
Materials and methods
Materials and sample preparation
Samples were produced using commercial ceramic powders and a solid plastic binder as starting materials. Alpha silicon carbide (grade UF 05; Stark, Goslar, Germany), with an average particle size d50 of 1.4 μm and a specific area of 4-6 m2/g, and zirconium diboride (grade A; Stark, Goslar, Germany), with an average particle size d50 of 0.3-5.0 μm, were used as starting powder. The particle size distribution data were provided by the powder producer using the laser light diffraction technique according to American Society for Testing and Materials (ASTM) B822.
To produce the green body, a phenolic novolac powder (average particle diameter of ~64 μm; Momentive, Columbus, OH, USA) was mixed with the solid charge in distilled water for 4 hours in a rotary mixer with glass spheres.
To study how the constituent materials would affect the oxidation behavior, 3 compositions with different relative amounts (% volume) of SiC and ZrB2 were selected. The ceramic components were then mixed with a fixed amount (40% volume) of the phenolic resin (first part of Tab. I).
Volume fractions of constituent materials
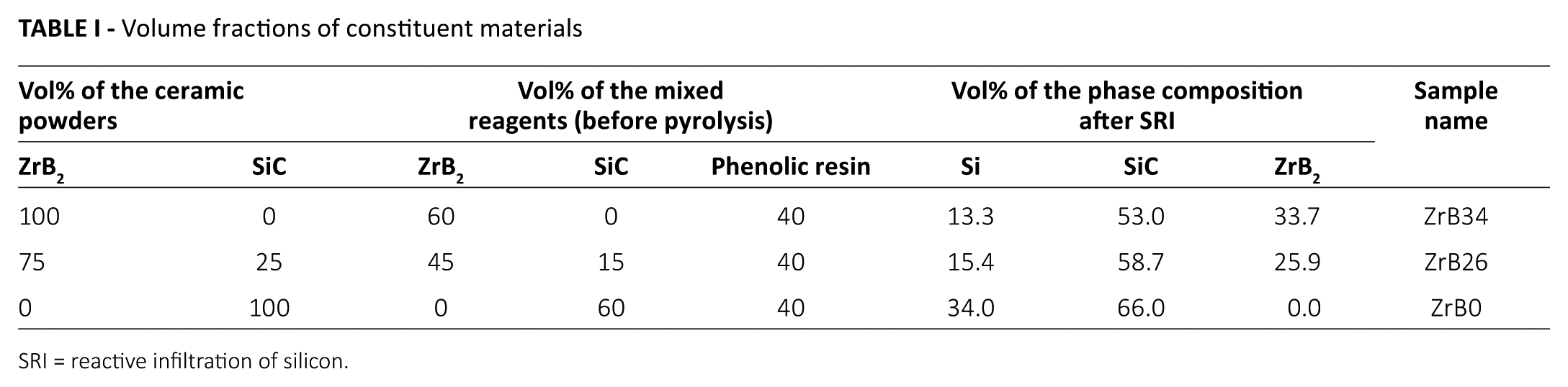
SRI = reactive infiltration of silicon.
The specimens were first pyrolyzed in an inert atmosphere at 1000°C for 1 hour and further densified according to the SRI technique. Samples were placed on carbon wicks which were themselves put on a BN-painted graphite plate. Silicon flakes were placed on the samples’ surface. The furnace was heated at 1500°C in vacuum with a residual pressure of 10−2 mbar; the maximum temperature was reached quickly and was maintained for 1 hour (for details see (15)). The system was then slowly cooled to room temperature.
The relative volume amounts of Si, SiC and ZrB2 phases have been determined from both image analysis and an X-ray diffraction (XRD)–based external standard method, as already described in our previous paper (14).
The image analysis was done on different regions of the samples to differentiate the areas containing ZrB2 phase from those including Si-based ones. This technique is not sensitive enough to clearly distinguish the areas occupied by Si or SiC. However, the evaluation of the amount of unreacted silicon represents a key factor in the investigation of this kind of composite materials. To separate the contributions coming from Si-based phases and thus quantify the relative amount of free silicon and silicon carbide, a quantitative method based on XRD was applied (14, 17, 18). Five standards were prepared to cover the whole range of the expected silicon amounts. They were prepared by mixing silicon carbide powder with different amounts of silicon (≥99.7% purity, average particle size <150 μm; Schuchardt, Műnchen, Germany), at 3, 5, 10, 15 and 25 wt%.
A calibration curve fitting the standards’ composition vs. integrated areas of silicon XRD peaks was obtained. The most intense peak for crystalline silicon at 2θ was 28.5° (corresponding to the plane (111)) was taken as reference. The calibration curve was then used to calculate the silicon content (wt%) in each sample on the basis of the integrated area of the Si peaks. These weight percentages were then converted into relative volume amounts using the densities of Si, SiC and ZrB2, which are 2.3, 3.18 and 6.2 g/cm3, respectively. The second part of Table I summarizes the relative amounts of the 3 phases (vol%) for all of the prepared samples.
Plates with dimensions of 90 × 40 × 4 mm were cut with diamond tools to produce 25 × 5 × 2 mm bars for the following oxidation and bending tests.
Oxidation tests
Oxidation resistance was investigated by submitting the Si-SiC-ZrB2 samples to temperatures of 1500°C and maintaining them for 4, 8, 24 and 48 hours, in static air and at atmospheric pressure. The oxidation tests were conducted in a resistance heated furnace (HT64/17; Nobertherm GmbH, Lilienthal, Germany). The furnace was slowly heated and cooled to avoid thermal shock on the samples and preserve the integrity of the oxide layer.
The oxidation behavior of Si-SiC-ZrB2 materials were then compared with those of pressureless sintered SiC materials, for which the processing method has been presented in more detail elsewhere (19), and with previously investigated SiC-ZrB2-based composite (6, 13, 20-26).
Characterization
The samples’ microstructures were investigated using an optical microscope (Leica DMLM; Wetzlar, Germany) and scanning electron microscopy (SEM-FEG Assing SUPRA 25; Zeiss, Germany) coupled with energy dispersive spectroscopy (EDS Oxford INCA X-Sight analyzer). The crystalline phases were identified by XRD measurements (PW3830; Philips, Eindhoven, The Netherlands).
The samples’ weights were recorded before and after oxidation tests using an analytical balance (Bc; Orma, Milan, Italy) with an accuracy of 0.1 mg. The rates of mass change for each composition were obtained by taking the mean value calculated for 6 samples.
The flexural strengths of the as-produced and oxidized samples were determined via 3-point bending using a span of 20 mm and a crosshead speed of 0.2 mm/min (Sintech 10/D; MTS, Eden Prairie, MN, USA). For each composition and oxidation time, at least 3 specimens were tested. To estimate the error bars in the graphs, the standard deviation was used.
Results and discussion
In Figure 1A, the results of the oxidation tests are reported in terms of weight gain, while in Figure 1B, the oxide layer thickness for each composition, which increased with dwell time, is shown. The specimen ZrB0 was the material least affected by oxidation treatments; the behaviors of ZrB26 and of ZrB34, both containing a high amount of ZrB2, were very similar.
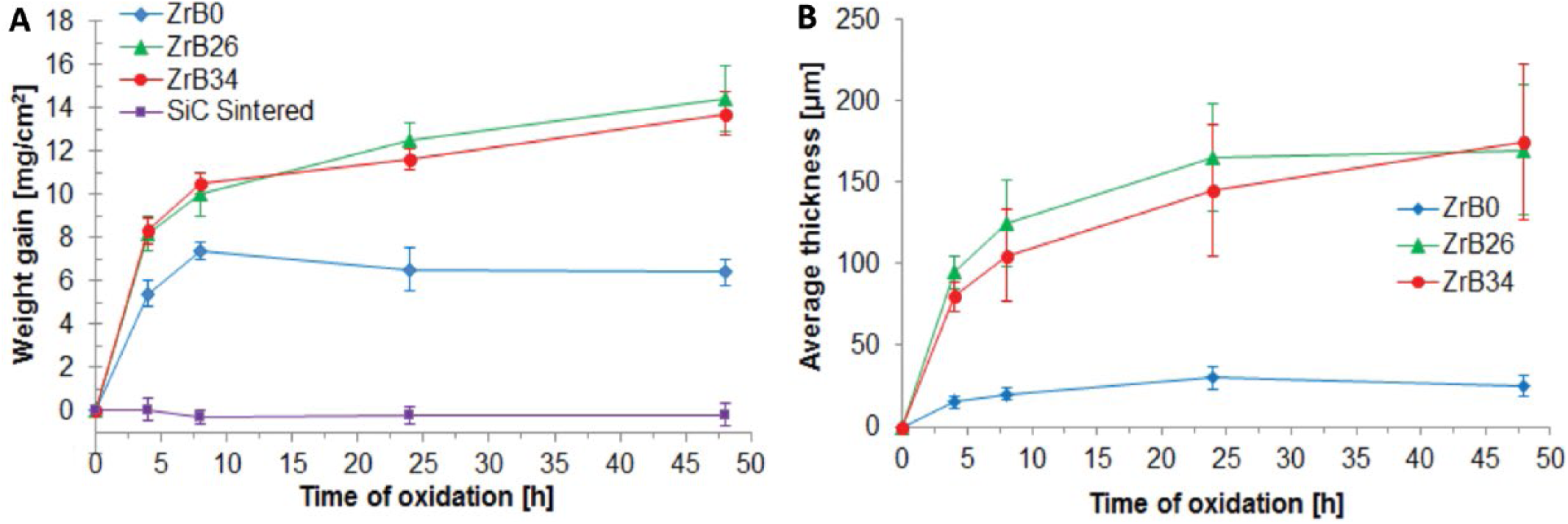
Comparison of samples’ oxidation behavior in terms of (
The oxidation rate of these Si-SiC-ZrB2 materials, evaluated in terms of weight gain, was lower in comparison with ZrB2-SiC systems reported in the literature (13, 21, 24-26) but higher than the values measured for sintered SiC (Fig. 1A). From Figure 1A, it is clear that even after 48 hours, the process of oxidation was not finished; in fact all of the Si-SiC-ZrB2 samples continued to gain weight. A stable passivation the SiO2 oxide layer was achieved only for samples ZrB0 and sintered SiC. This feature was confirmed by XRD characterization (Fig. 2).
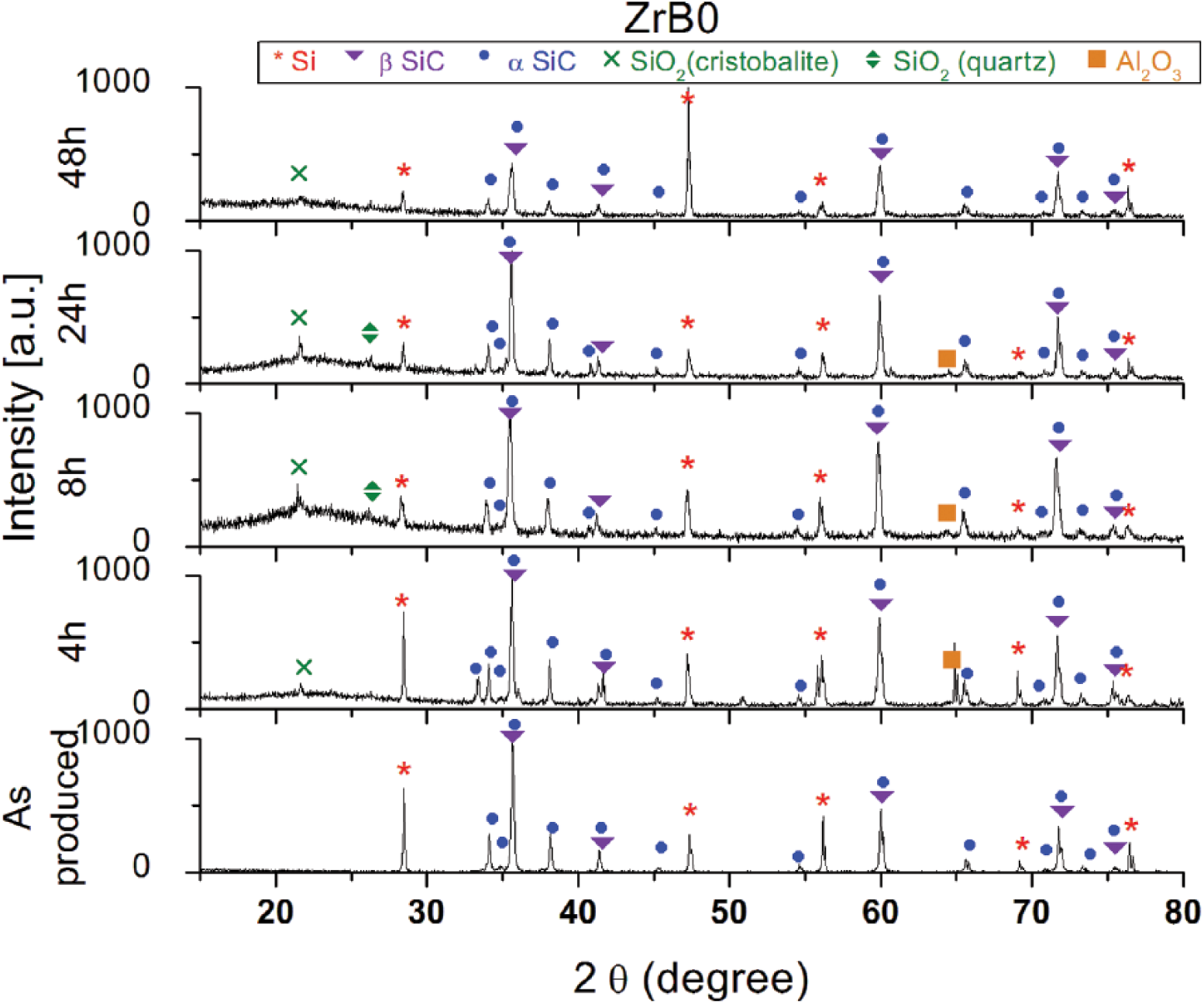
X-ray diffraction (XRD) patterns of ZrB0 at 0, 4, 8, 24 and 48 hours of oxidation at 1500°C.
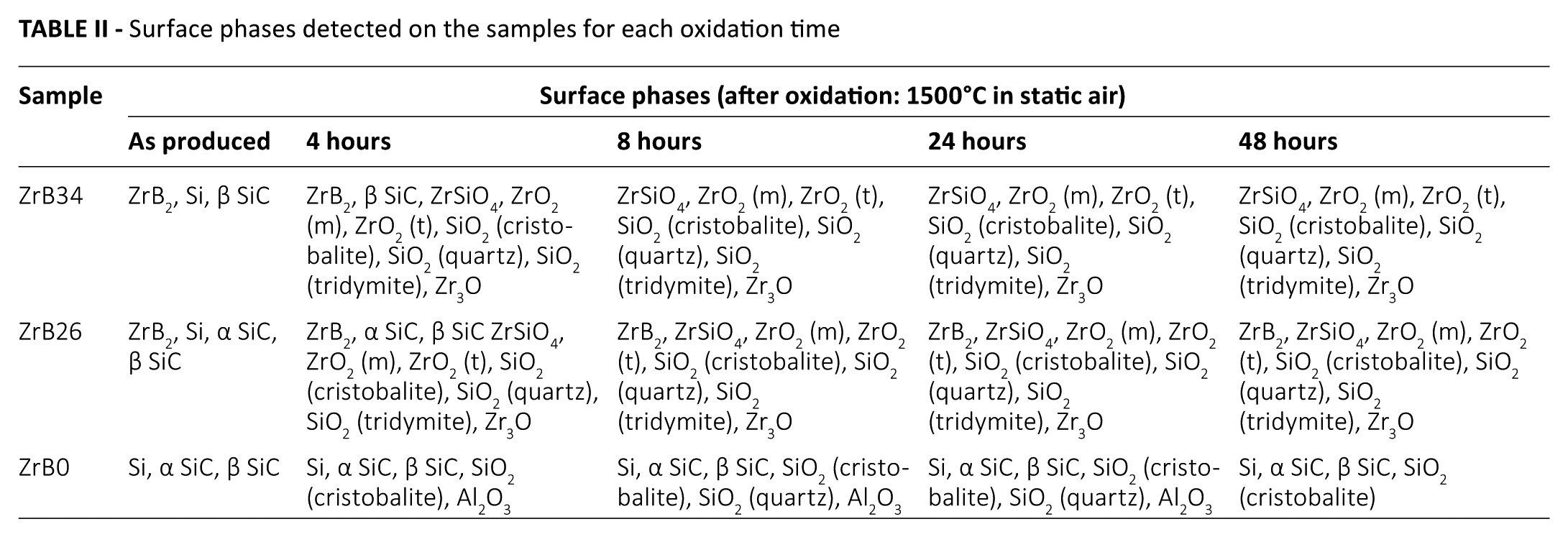
XRD patterns for ZrB2-containing specimens (Figs. 3 and 4) show the formation of several oxides on the exposed surface – namely, zirconia (both monoclinic and tetragonal), silica (cristobalite, tridymite, quartz), zirconium silicate (ZrSiO4) and 3-zirconium monoxide (Zr3O); in Table II, all of the different phases detected on the sample surfaces are summarized. These oxides were predicted by the state diagrams and were already present after 4 hours of oxidation. B2O3 was not detected due to its volatilization, which starts above 1100°C. The main chemical reactions of oxidation, which occur up to 1500°C in all Si-SiC-ZrB2 systems are:
2 SiC (s) + 3 O2 (g) → 2 SiO2 (s) + 2 CO (g) Reaction 1
Si (s) + O2 (g) → SiO2 (s) Reaction 2
2 ZrB2 (s) + 5 O2 (g) → 2 ZrO2 (s) + 2 B2O3 (l) Reaction 3
B2O3 (l) ⇄ B2O3 (g) Reaction 4
ZrO2 (s) + SiO2 (s) → ZrSiO4 (s) Reaction 5
3 ZrB2 (s) + 5 O2 (g) → Zr3O (s) + 3B2O3 (l) Reaction 6
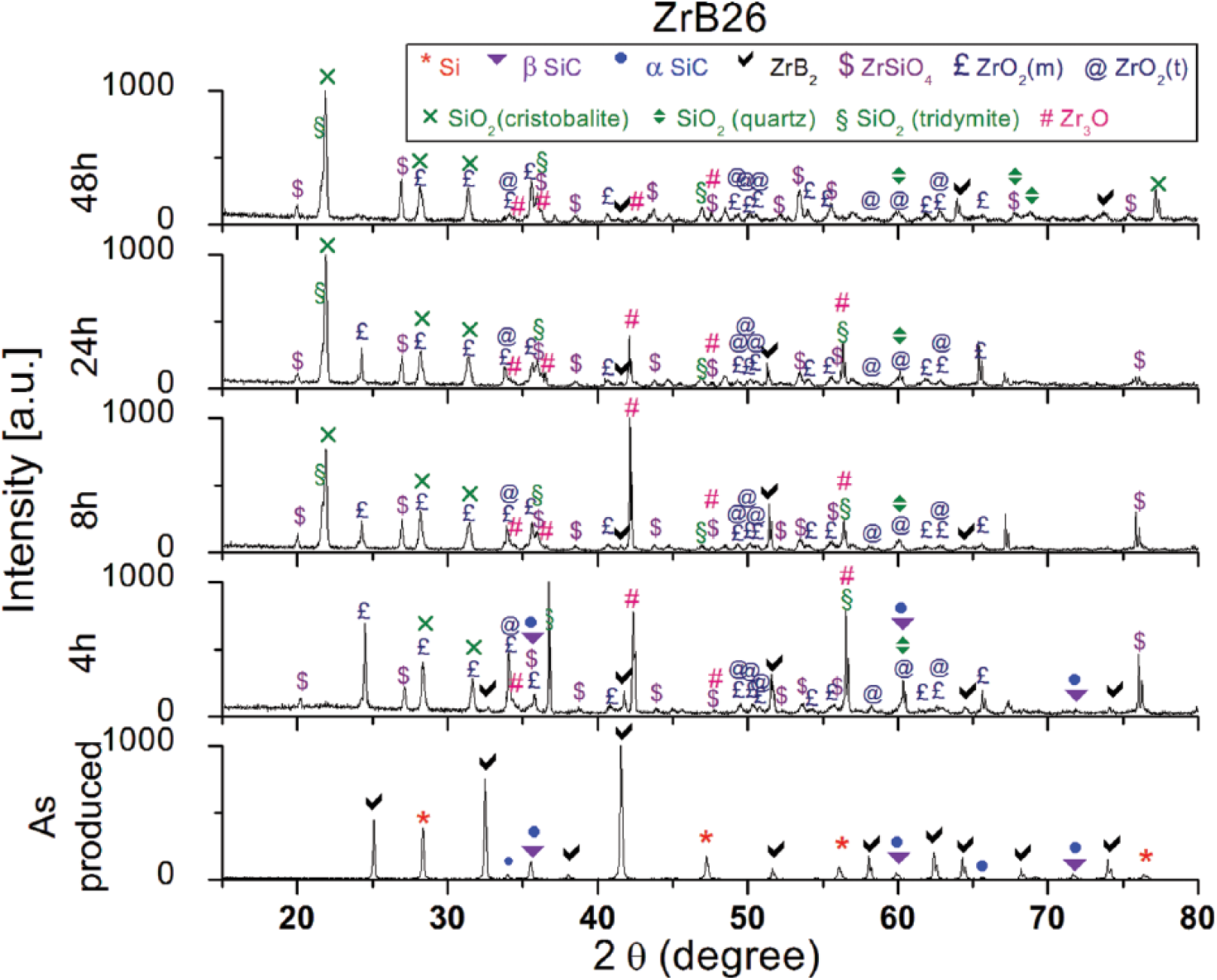
X-ray diffraction (XRD) patterns of ZrB26 at 0, 4, 8, 24 and 48 hours of oxidation at 1500°C.
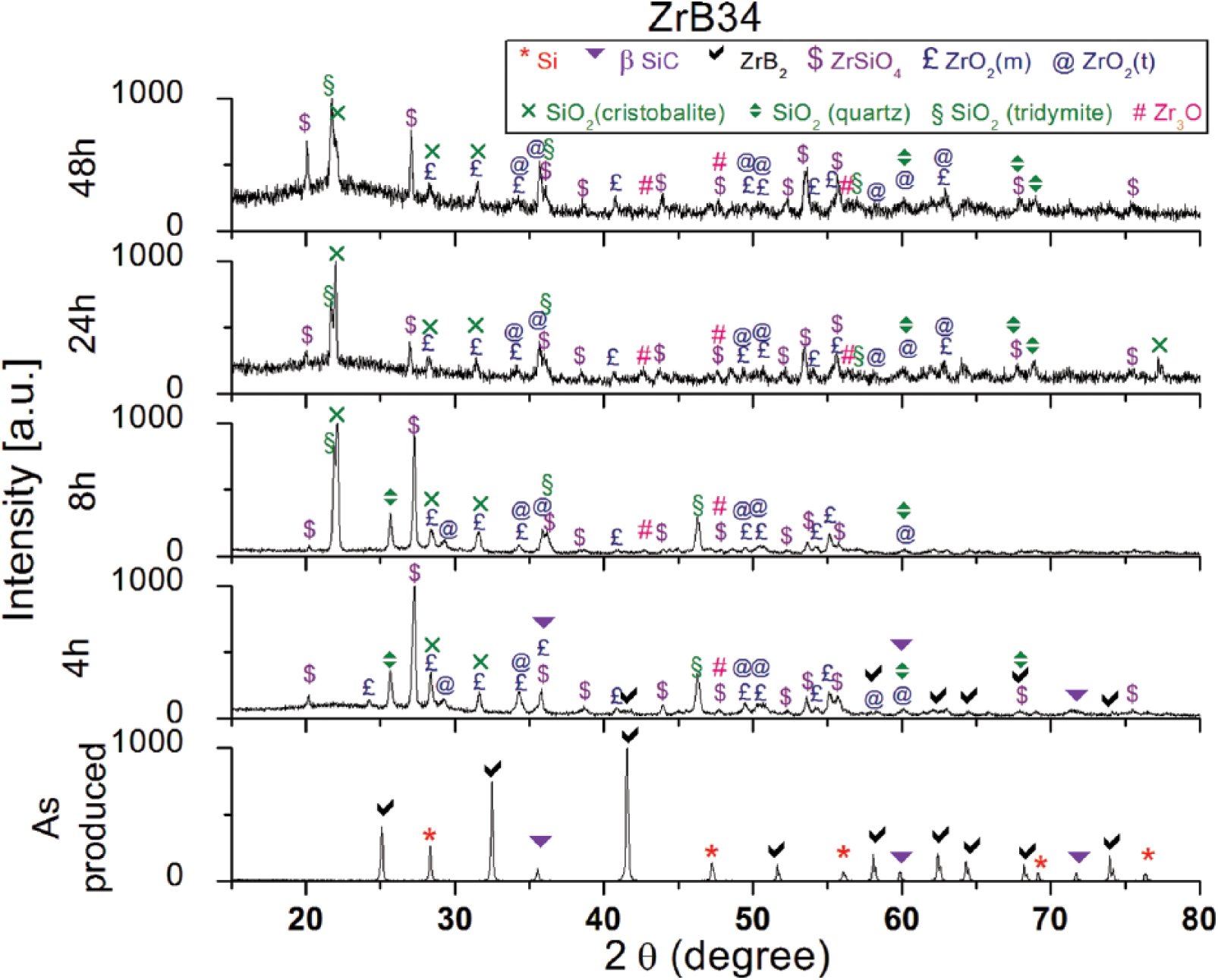
X-ray diffraction (XRD) patterns of ZrB34 at 0, 4, 8, 24 and 48 hours of oxidation at 1500°C.
Surface phases detected on the samples for each oxidation time
After 8 hours, Si-SiC bulk material below the scale was undetectable by XRD, and only traces of ZrB2 could be found in ZrB26 (Figs. 3 and 4). The formation of crystalline SiO2 is not well-understood, but it could be due to the presence of a not negligible amount of Si in the amorphous silica. Indeed, when the silicon concentration reached saturation value, the precipitation of crystalline SiO2 from the amorphous form was to be expected (27). In addition, at temperatures above 1400°C, for longer times and in the presence of impurities, the crystallization of SiO2 becomes important, reducing the diffusion rate of gaseous products. The oxidation of both ZrB2 and SiC leads to the formation of a surface silica-rich glassy scale consisting of ZrO2 and amorphous silica.
The coexistence of these 2 phases results in a further reaction between them, as shown in Reaction 5. Veytizou et al (28) reported that the presence of both ZrO2 and amorphous silica, in addition to the presence of interstitial silicon, can cause the precipitation of ZrSiO4: the interstitial silicon can indeed diffuse and dissolve into crystalline ZrO2 until the solubility limit is reached. The formation of this phase, called zircon (23), has been reported by several authors (13, 23, 25-27, 29-32).
The reference ZrB0 material (Fig. 2) showed a very different oxidation behavior. Even after 48 hours of thermal treatment, the peaks related to the unoxidized bulk were well defined, and only the oxide crystalline phase was silica. SiO2, initially present in both amorphous and crystalline phases, as oxidation continued, tended to become totally glassy. The peaks related to the alumina phase were due to a contamination of the sample from the furnace.
Further, for sample ZrB0, the trend of the measured oxide thickness appeared to be much lower than the respective gains in weight. This is clear from Figures 5 and 6, and it can be explained by the fact that the oxide layer was more compact, homogeneous and almost devoid of porosity. In the reference material, indeed, the thickness of the scale, even after 48 hours of treatment, did not exceed 30 μm. After 8 hours, however, a parabolic oxidation behavior was observed for each curve. This suggests that after a first stage involving the formation of an external glassy scale, diffusion is the mechanism which controls the oxidation rate, as previously observed for different ZrB2-based composites (33-35). The 2 relevant mechanisms of oxygen diffusion in SiO2, in this range of temperatures, are the diffusion of interstitial oxygen and the network exchange of ionic oxygen, known as the “network exchange diffusion” or “ionic diffusion.”
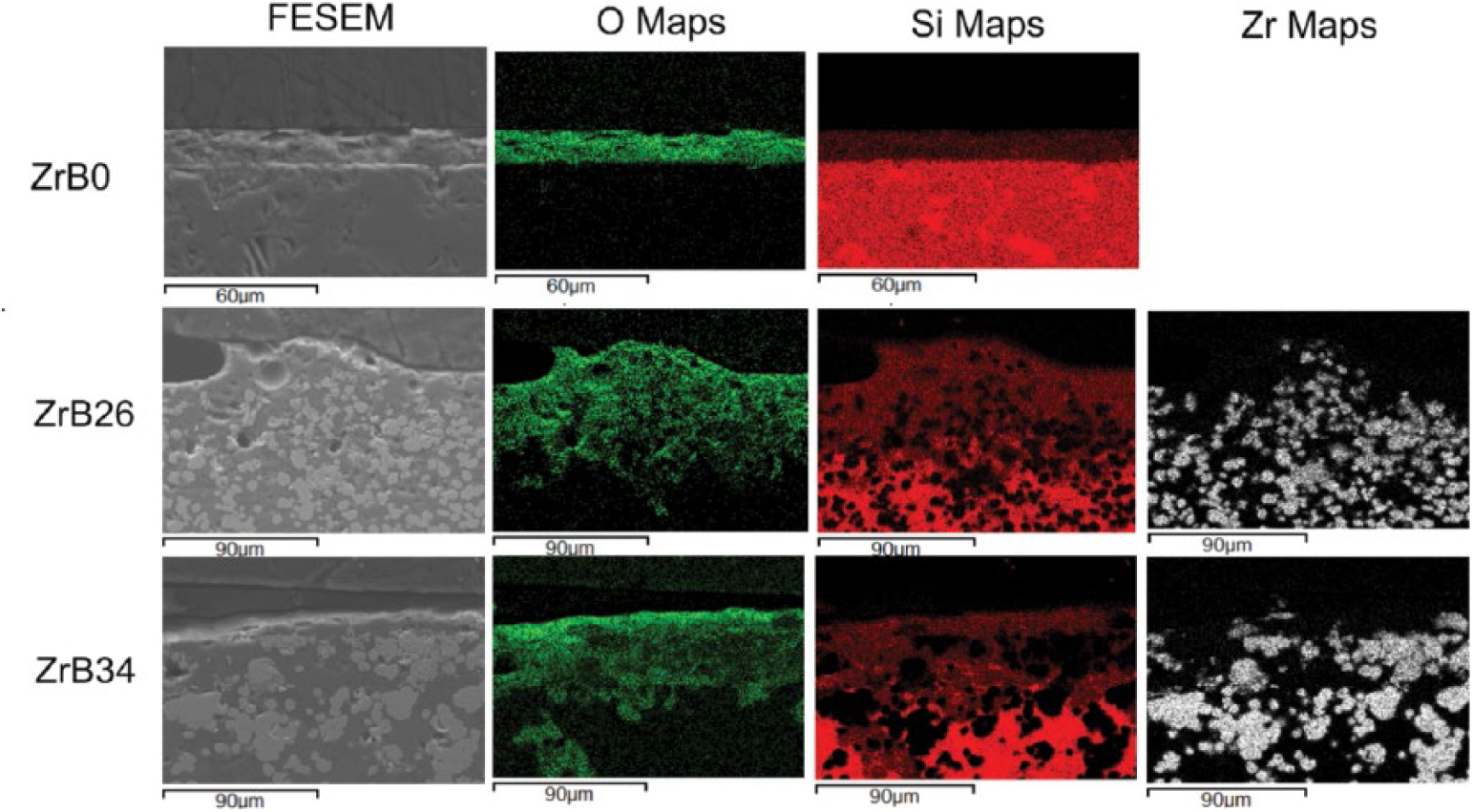
Field emission scanning electron microscope (FESEM) images and elemental maps of sample scales after 4 hours at 1500°C in air.
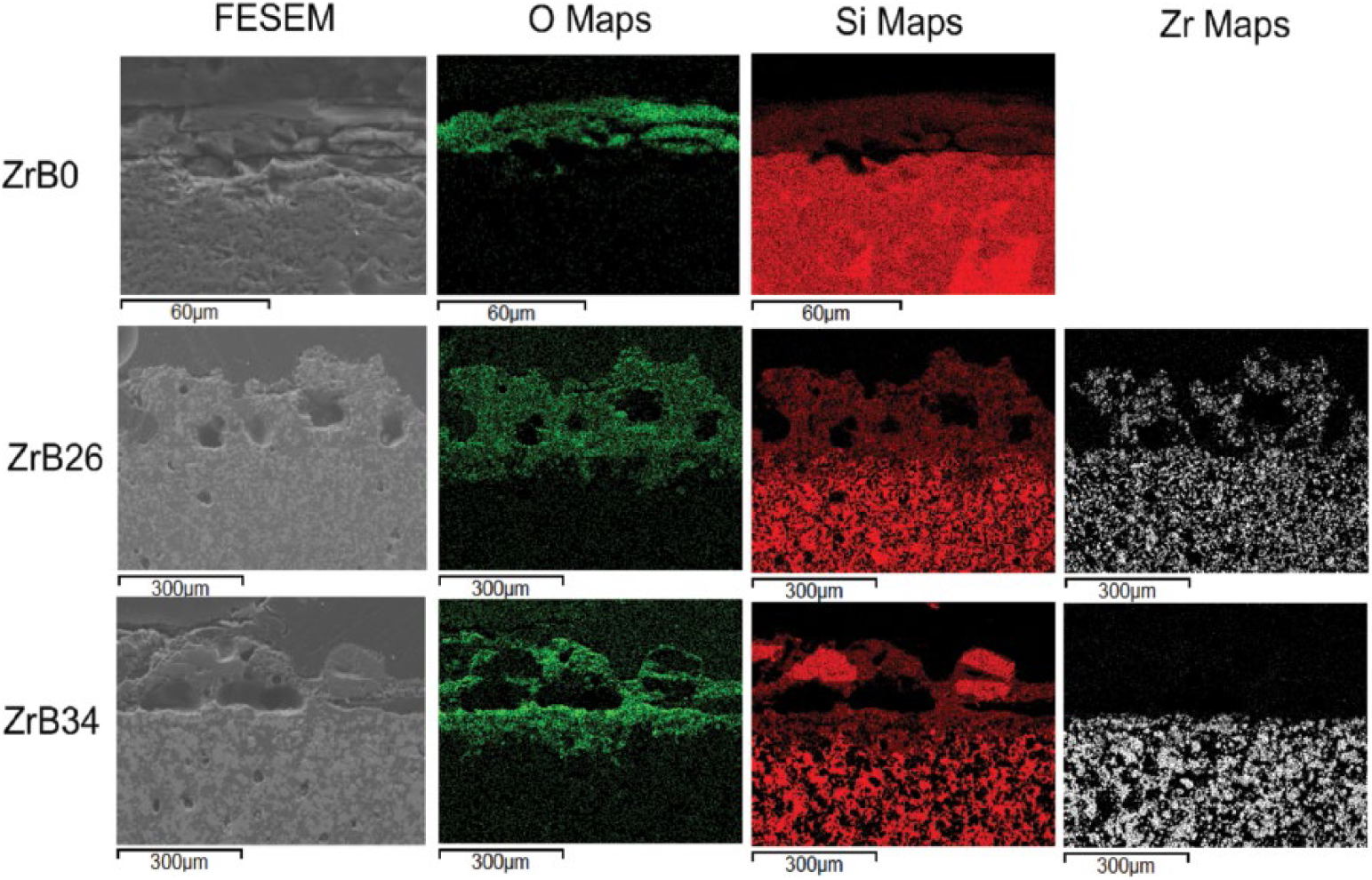
Field emission scanning electron microscope (FESEM) images and elemental maps of sample scales after 48 hours at 1500°C in air.
Figures 5 and 6 show the field emission scanning electron microscope (FESEM) images taken on the edge section for each specimen subjected to oxidation for different dwelling times. A first characteristic, common to all samples, was the change in features of the oxide layer with increasing oxidation time. In the first stages – i.e., after 4 and 8 hours – the scales appeared compact and still fitting rather well to the bulk underneath. After 24 and 48 hours, the scales were very porous and irregular, but still adhering well to the unoxidized substrate.
The high porosity can be explained not only by the volatilization of some phases, but also by the fact that changing volume can also produce porosity and cracks. Some cracks were observed: they could have been due either to the difference between the thermal expansions of ZrO2 and SiO2 or to their volume changes, which accompanied the structures’ transition as reported in Reactions 8 and 9.
The presence of different polymorphic phases of silica and zirconia, as shown in the XRD spectra (Figs. 3 and 4), can be explained by equilibrium transformations that happen during cooling (Reactions 8 and 9).
ZrO2 (tetragonal) ⇆ ZrO2 (monoclinic) Reaction 8
SiO2 (cristobalite) ⇆ SiO2 (tridymite) ⇆ SiO2 (quartz) Reaction 9
Zirconia has 3 polymorphs: The monoclinic form is stable at room temperature. For temperatures higher than 1170°C, the tetragonal form becomes stable, followed by the cubic one at even higher temperatures (>2370°C). The oxidation tests were done at a temperature of 1500°C, so both the monoclinic and tetragonal forms were present. Similar considerations can apply to the different SiO2 polymorphs which involve transition temperatures up to 1500°C.
The scales of the ZrB2-containing specimens are mainly made of silica and particles of zirconia, as can be deduced from the elemental maps where the silica phase is wrapped around the ZrO2 particles. For the sample ZrB26, the oxidation mechanism firstly proposed by Karlsdottir and Halloran is applicable (22). They suggested a convection mechanism in which the borate flow is driven by an increase of volume as the result of ZrB2 oxidation. In fact the oxides formed have a different molar volume with respect to that of the substrate. In the literature for metals oxidation, the so-called Pillings-Bedworth Ratio (PBR) is defined as the ratio between the molar volume of formed oxide and that of unoxidized material. The oxidation of SiC causes the formation of mainly silica (Reaction 2). Considering the molar volumes of SiC and SiO2, which are 12.47 cm3/mol and 27.43 cm3/mol, respectively, the PBR of silica is 2.2.
In the case of ZrB2, its oxidation leads to the formation of both ZrO2 and B2O3. It is thus necessary to define partial PBRs for boron and for zirconium on the basis of the molar volume of ZrB2 (18.54 cm3/mol), B2O3 (55.2 cm3/mol) and ZrO2 (20.92 cm3/mol) (36).
The partial PBR for B is 2.98, and this is associated with the formation of an oxide in a liquid form which can flow. The PBR for Zr has a lower value of 1.13, and this is linked to the formation of an oxide (ZrO2) with a rigid solid structure. The formation of liquid oxide leads to a large volume increase, so this liquid phase must be squeezed out to the surface. The increase of volume upon oxidation is therefore the driving force for liquid flow.
The main differences between this model and our samples are represented by the lack of boron fingering on the surface and by the absence of the porous ZrO2 layer under the external layer. The absence of boron oxide on the surface is justifiable given the long oxidation times at 1500°C which cause the complete evaporation. The second aspect, the absence of the columnar structure of zirconia, has been confirmed in other works, in which it was demonstrated that it was formed only with percentages of ZrB2 above 50 vol% (16, 34, 37, 38) as in the study of Karlsdottir and Halloran (22, 23). From the elemental maps of ZrB34, this sample seems not to follow the convection cells model; indeed no zirconia particles can be detected in the scale. XRD patterns (Fig. 4) show how zirconia and zircon are present on the external oxide layer of this sample, but the intensity of the related peaks is lower when compared with ZrB26. The lower height of the peaks belonging to the zirconia, may thus be explained by the presence of many small crystallites, which are not appreciable at low magnifications in the elemental maps. It is therefore possible to suppose that this mechanism also takes place in this type of sample, but to a lesser extent.
In all of the micrographs, the SiC depletion layer was not observed; this is in agreement with what was previously pointed out by Williams et al (16). In that study, it was found that the formation of SiC-depleted layers after isothermal exposure at high temperatures could be reduced by increasing the relative amount of SiC, which is not observed in composites containing volume percentages of SiC >50 vol%.
Flexural strength
Many authors have reported that SiC and Si3N4 ceramics maintain a good strength after oxidation treatments (39, 40), but only very few data are available to support this behavior for ZrB2-based ceramics (41-44). Figure 7 shows the effect of oxidation on Si-SiC-ZrB2 ceramics. By increasing the oxidation time, the flexural strength of all samples containing ZrB2 was retained; they showed similar trends that gradually decreased in agreement with the reduced thicknesses of the unoxidized specimen zones. Nevertheless, the strengths after 48 hours of oxidation were between 70%-90% when compared with those of the unoxidized samples. The same trend could be observed for sample ZrB0: its flexural strenght was slightly lower respect to those observed for samples containing ZrB2, but they are consistent with the literature data (45, 46). The decreasing trend can be explained considering the high content of free silicon. Eventually, the ZrB2-based ceramics flexural strength was considerably increased by the addition of a second phase. It must be underlined, however, that the dispersion in this kind of measurements is very high. The fracture surface of sample ZrB26 (Fig. 8) showed a different failure mechanism for the external oxidized layer compared with the inner part. The examination of the bulk material (Fig. 8A) revealed a fully intergranular mode of fracture. In contrast, moving away from the inner part, the external area (Fig. 8B) shows the presence of smooth cleavage facets indicating a modification of fracture mode corresponding to the oxidized layer.
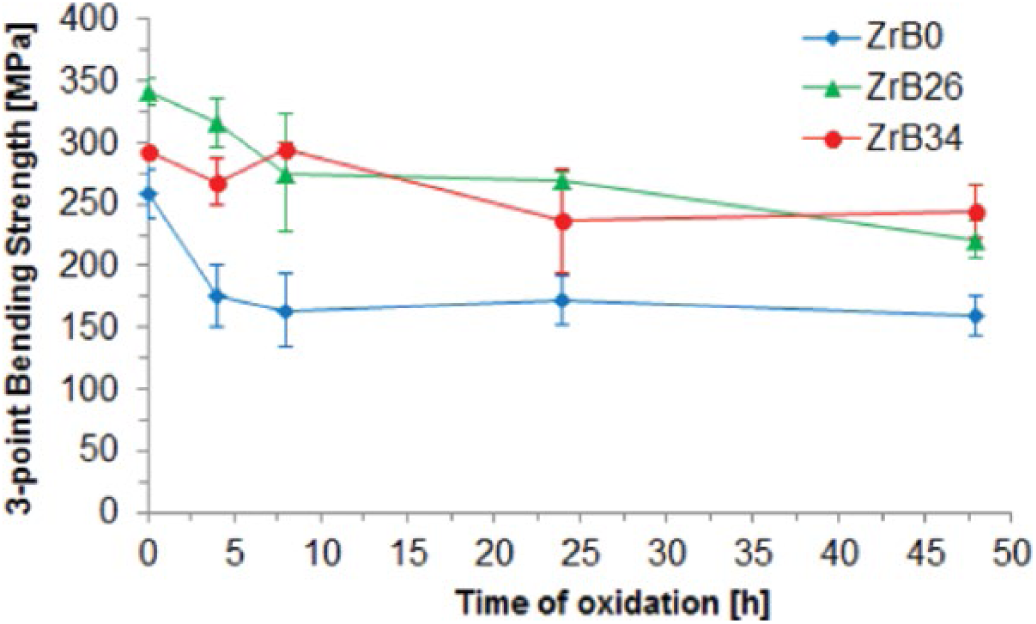
Flexural strength of the samples at different oxidation times.

Field emission scanning electron microscope image of fractured surface of sample ZrB26 scale after 48 hours of oxidation at 1500°C in air: (
Further, the presence of surface defects which may have an influence on the flexural strength should be noted. Other works have reported an increment of strength after oxidation for Si-based materials such as SiC and Si3N4 (40, 41); those results were obtained for lower oxidation temperatures and times compared with the experimental conditions in our tests. This effect is due to the formation of a dense and thin oxide scale which can heal the surface flaws coming from sample processing and machining. This healing effect was effective if the oxide layer was thin, dense and showed a good adhesion to the bulk material. If the oxide was thick and porous, the flaw-healing effect offsets the formation of new surface defects either in the oxide layer or at the interface between it and the underlying unoxidized material.
Guo (42) reported how the first behavior is typical of the nano-sized SiC-ZrB2 composites, while the second one is characteristic of micron-sized SiC-ZrB2 composites, as in our case. This can affect the flexural strength of Si-SiC-ZrB2 materials.
Conclusions
Si-SiC-ZrB2 bulk ceramics were produced by the reactive infiltration technique. A detailed study of the oxidation behavior of this material at high temperature was made: The interaction between Si-SiC-ZrB2 components during the oxidation, their chemical activity with oxygen at high temperatures and the effect of oxidation on the room temperature flexural strength were investigated.
Si-SiC are fully dense materials showing a good oxidation resistance, but their use is limited to temperatures up to 1600°C. For higher-temperature applications, UHTCs are preferentially used, and among them, ZrB2 is one of the most promising materials, generally processed in combination with SiC to obtain a material with good mechanical and oxidation properties, even at high temperatures.
There have been few studies reported in the literature which used a “mild process” to produce dense Si-SiC-ZrB2 composites at relatively low temperatures (1500°C) and under pressureless conditions. In this context, the study of the oxidation behavior of these materials is needed to evaluate their performance at high temperatures under oxidative conditions.
Already after 4 oxidation hours, all of the samples with ZrB2 showed a passivating oxidation layer, and as was evident from their specific weight gains, even after 48 hours, the oxidation process was not finished. This paper reports all of the main chemical reactions of oxidation that occur up to 1500°C and phase transformations on the basis of state diagrams. Several oxides were detected on the surface: zirconia (monoclinic and tetragonal), silica (cristobalite, tridymite, quartz), zircon (ZrSiO4) and 3-zirconium monoxide (Zr3O). Boria was not detected. This study lends support to the oxidation model proposed by Karlsdottir and Halloran (22): the so-called convection cells mechanism, in which the increase of volume of oxide products leads to the squeezing out of BSZ liquid to the surface. In support of what has already been noted in our previous studies (14, 15), an SiC depletion layer was not found in any samples. The oxidation of Si-SiC-ZrB2 material produced via SRI was slower compared with previously investigated ZrB2-SiC composites processed with different techniques and tested in similar conditions.
With regard to the mechanical tests, oxidation influenced the strength of Si-SiC-ZrB2 ceramics. All of the samples showed the same tendency of keeping from 70% to 90% of the original flexural strength, displaying an intergranular fracture.
Footnotes
Acknowledgements
This work was made possible thanks to the contribution of EngiCer SA which provided all of the samples tested. In particular, the authors wish to thank S. Giannella for designing and preparing ceramic specimens for the experiments.
Disclosures
Financial support: No grants or funding have been received for this study.
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
