Abstract
The mechanical properties and biocompatibility of titanium alloy medical devices and implants produced by additive manufacturing (AM) technologies – in particular, selective laser melting (SLM), electron beam melting (EBM) and laser metal deposition (LMD) – have been investigated by several researchers demonstrating how these innovative processes are able to fulfil medical requirements for clinical applications. This work reviews the advantages given by these technologies, which include the possibility to create porous complex structures to improve osseointegration and mechanical properties (best match with the modulus of elasticity of local bone), to lower processing costs, to produce custom-made implants according to the data for the patient acquired via computed tomography and to reduce waste.
Keywords
Introduction
Metals commonly used for biomedical applications possess specific characteristics: They must be nontoxic, nonimmunogenic, nonthrombogenic and noncarcinogenic, and exhibit a high corrosion resistance (1, 2). Body fluids contain a high concentration of chloride ions and various amino acids and proteins that lead to corrosion phenomena: Different oxide-reduction reactions take place between biological fluid and metallic surfaces, inducing their modification and possible ion release inside the body. This can cause several negative effects, such as allergies or carcinoma (3).
Moreover, mechanical properties also play a key role, depending on the specific application. For example, stents and stent grafts require materials with high plasticity and enough rigidity to allow expansion and keep dilatation, while orthopedic implants must have excellent toughness, elasticity, strength and fatigue resistance. In the case of joint replacement and dental implants wear can create serious damage to the surface and also debris inside the body: In such conditions, it is generally required to use hard and fatigue-resistant materials.
As a consequence, the principal metallic biomaterials are stainless steels, cobalt-chrome alloys, and titanium-based alloys, as summarized in Table I. Stainless steels are in widespread use due to their corrosion resistance, elevated strength and relative low price (4-6). The most commonly employed stainless steel is 316L (AISI classification): Examples of applications are cases of craniofacial and bone fixation in the form of pins, plates, intermedullary nails, rods or screws, as described by Disegi and Eschbach (7). On the other hand, Co-Cr alloys, owing to their high degree of hardness and wear resistance, have been employed successfully for orthodontic applications and artificial joints, such as knee and hip replacement prosthesis (4, 5), or tibial trays and acetabular cups (8, 9). Also titanium and titanium-based alloys have been widely applied in the orthopedic field, thanks to their excellent corrosion resistance to body fluids and their Young modulus, which are more similar to bone compared with other alloys (2).
Tensile properties of the main metallic biomaterials
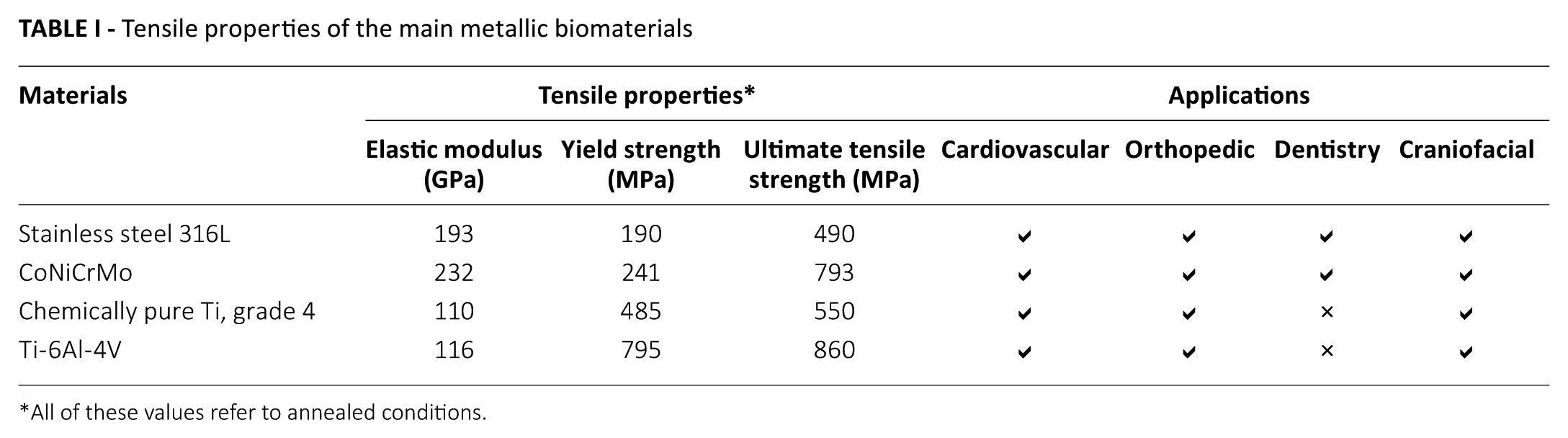
All of these values refer to annealed conditions.
Until now, the majority of metallic implants have been fabricated using traditional processes including forging, investment casting, hot rolling and machining (10). Several fabrication methods are required because not all of the implant alloys can be easily and economically processed into a final shape in the same way (11). However, in the last 30 years a new kind of manufacturing technology, grouped under the name of additive manufacturing (AM), has been developed. As the name implies, unlike subtractive manufacturing, the new approach is based on adding material only where it is needed for building the part (12). According to ASTM 52921 of the International Organization for Standardization (13) in fact, AM can be defined as the “process of joining materials to make objects from 3D model data, usually layer upon layer.”
AM technologies use a focused heat source to selectively melt raw material and consolidate it to fabricate a part. The processes can be divided on the basis of the raw material (plastics, metals or ceramics), its starting state (powder or wire), heat source (electric resistance, laser or electron beam) and deposition method (spreading or depositing). The most widespread AM techniques for metals are powder bed fusion (PBF) processes (14), such as selective laser melting (SLM) and electron beam melting (EBM); and direct deposition processes, such as laser metal deposition (LMD) (15-17). Thanks to these techniques, also materials difficult to be machined like titanium-based alloys, if available in powders, can be turned into near net shape components.
Different industrial sectors, from automotive (18) and aerospace (19) to jewellery (20) and biomedical (17) industries, have adopted AM processes because of the numerous advantages they offer to producers and customers. The most relevant benefits include the product customization (21), the reduction of tooling costs, waste and energy consumption (22) and the possibility of having on-demand manufacturing and new supply chain approaches (12). Moreover, in the production of orthopedic products, AM technologies offer the possibility to realize complex shapes (novel shapes, hollow structures, functionally graded parts) able to improve implant compatibility, patient wellness with shorter surgery durations and better corrosion resistance favoring tissue ingrowth, cell migration, adhesion and growth (23).
The aim of the present study was to provide an overview of the major benefits and issues in using AM metal powder–based processes, in particular SLM, EBM and LMD, for the production of implants and prostheses made of titanium-based alloys.
Titanium alloys in the biomedical field
The first relevant applications of titanium as a biomaterial for dental and surgical devices go back to the end of World War II, thanks to improvements in the titanium manufacturing processes (24). Titanium-based alloys are mostly used for their significantly high strength to density ratio and resistance to corrosion and fracture-related properties. Commercially pure titanium (CP-Ti) and Ti-6Al-4V alloy are the preferred titanium-based materials for implant fabrication. Titanium is a transition element, with a melting point of around 1678°C, which undergoes an allotropic transformation in the solid state at 882°C, referred to as the β-transus temperature (24), in which a change from the α phase (hexagonal closed-packed structure, hcp) to a β one (body-centered cubic structure, bcc) occurs. Thanks to the presence of a stable and an inert oxide layer, which spontaneously forms on its surface, CP-Ti has an elevated corrosion resistance and biocompatibility, and is widely considered to be the most compatible metal for the human body. Furthermore, by adding α-stabilizer elements (Al, C, O, N, B) or β-stabilizer elements (Fe, Mo, V, Cr, Ni, Cu, W, Co, Nb, Ta) the α →β transition temperature can be modified. Therefore, depending on the chemical composition and allotropic structure, titanium alloys are classified as α alloys, near α alloys, metastable β alloys, β alloys and α+β alloys (24-26). Alpha and near α alloys exhibit low values of tensile strength at room temperature but superior corrosion resistance. Compared with the α phase, the β phase induces a lower resistance to plastic deformation and higher ductility, and a significant anisotropy of physical and mechanical properties (Tab. II). The α + β alloys possess higher strength due to their biphasic microstructure, whereas β alloys possess a low elastic modulus and superior corrosion resistance (25, 27).Therefore, by modifying the manufacturing process conditions (thermal treatment and thermomechanical processing conditions), it is possible to alter the microstructure and directly influence the mechanical properties (28).
Mechanical properties of titanium alloys for biomedical applications
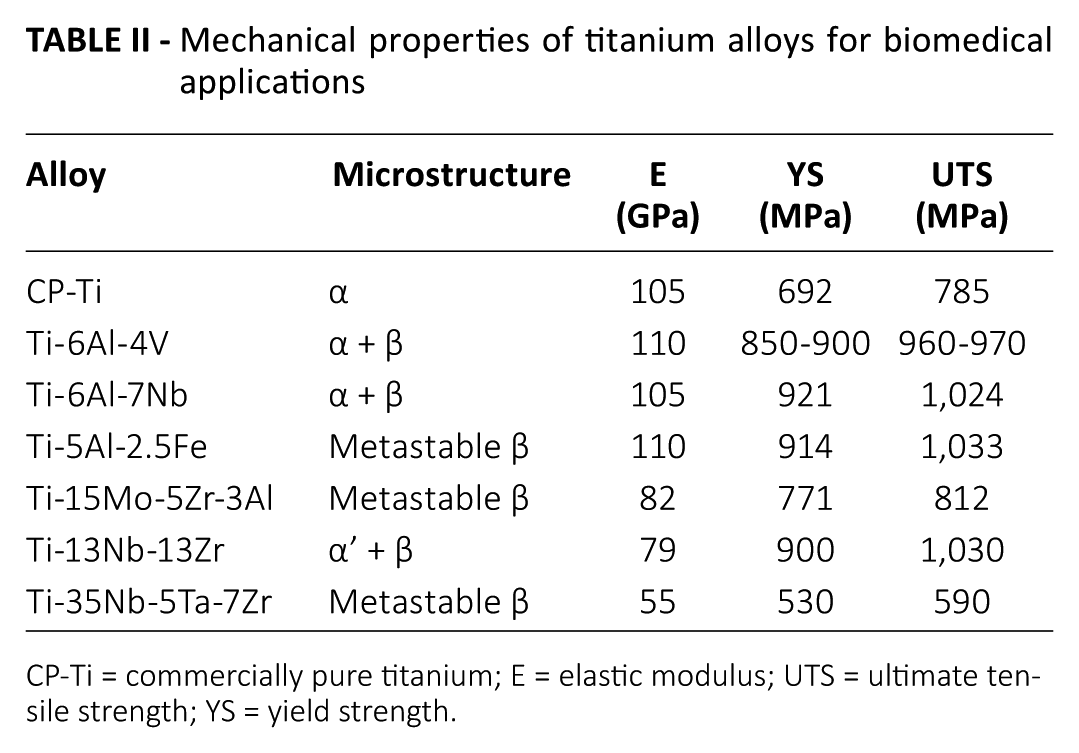
CP-Ti = commercially pure titanium; E = elastic modulus; UTS = ultimate tensile strength; YS = yield strength.
Among the different alloys, Ti-6Al-4V has been used for several biomedical devices on account of its high strength and fatigue properties at room temperature, together with a higher resistance to corrosion compared with the other biomedical alloys (29). Notwithstanding its good biocompatibility, in some cases, ion realizing can occur, inducing cytotoxicity problems (30) and limiting the number of possible applications. Thus, other Ti-based alloys such as Ti-Ta-Zr, Ti-Nb-Zr and Ti-Fe-Ta, have recently been investigated as possible alternatives (26, 31-33). Okazaki et al (29) compared the ions released in rat tibia tissues from Ti-15Zr-4Nb-4Ta with different metals, from 316L SS to Co-Cr-Mo and and Ti-6Al-4V alloys. The (Zr + Nb + Ta) concentration appeared 20% inferior to the (Al + V) one (28), suggesting a promising corrosion resistance, coupled with a suitable mechanical behavior, for some alternative Ti alloys.
The most widely employed Ti-based implants, made of CP-Ti and Ti-6Al-4V, are generally manufactured via conventional forming operations (forging, cold and hot forming, hydroforming), machining processes (drilling, milling, turning), and alternative machining techniques such as laser cutting and water-jet cutting. Due to their relatively low ductile yield and high tensile strength, machining technologies of titanium and its alloys represent a considerable manufacturing challenge. The lower elastic modulus may cause a greater “springback” effect, which is a geometric change made to a part at the end of the forming process. Therefore, there is a need to have rigid setups can induce high pressure and temperature in the tool contact zones. This can represent a problem since at higher temperatures the titanium becomes more chemically reactive. In addition, overheating of the surface can result in interstitial absorption of nitrogen and oxygen that can create a series of microcracks which reduce the performance and the fatigue properties. For such reasons, the conventional cutting speed values for titanium alloys machining are relatively low, implying longer and more expansive production processes.
The numerous drawbacks of the traditional production routes of Ti-based materials offer to AM technologies the occasion to demonstrate their ability to realize parts with comparable or higher features with a not negligible cost reduction (34). Furthermore, the complex thermal evolutions and the high temperature gradients of the different AM processes, combined with the allotropy of Ti and its alloys, give the possibility to largely modify their microstructure and mechanical properties.
In recent years, AM favored the develop of metallic foams and porous devices limiting conventional tooling and manufacturing issues, obtaining mechanical properties that can be adjusted to make them comparable to human bone (35-41). The stiffness values of the titanium prostheses can be lowered to values between 3 and 40 GPa (cortical bone values), limiting stress shielding and improving prosthesis osseointegration. In this way, the elevated biocompatibility of titanium can be used without provoking any bone cell resorption, providing also outstanding tensile properties.
In the following sections, after a brief presentation of SLM, EBM, and LMD technologies, research studies focusing on Ti-6Al-4V AM processing are reported, to underline the relations between AM process and material microstructures and properties.
Additive manufacturing metal powder–based processes
In metal powder–based AM processes, parts are manufactured layer-by-layer by melting metal powder under the effect of an electron or a laser beam. These processes, as in all AM technologies, start from the creation of a 3-dimensional (3D) computer-aided design (CAD) model which may be created in a CAD system or obtained from reverse engineering, such as with 3D scanners or computer tomography. The 3D CAD model is subsequently converted into standard .STL file format, and a slicing algorithm slices the .STL model into a 2D layers parallel to the build direction.
Although metal AM processes share the same additive approach, each process has its specific characteristics in terms of useable materials, processing procedures and applicable situations. In the biomedical field, the uses of additive technologies are rapidly expanding and are expected to revolutionize health care. Applications include tissue and organ fabrication, and creation of customized prosthetics, implants, and anatomical models. Among the metal AM technologies, powder-based processes, such as laser powder bed fusion (LPBF), also known as SLM and EBM; and directed energy deposition (DED), such as LMD, are increasingly being used in biomedical applications. Table III shows some of the main geometrical features and material properties obtainable by the 3 technologies.
Geometrical features and material properties obtainable by selective laser melting, electron beam melting and laser metal deposition
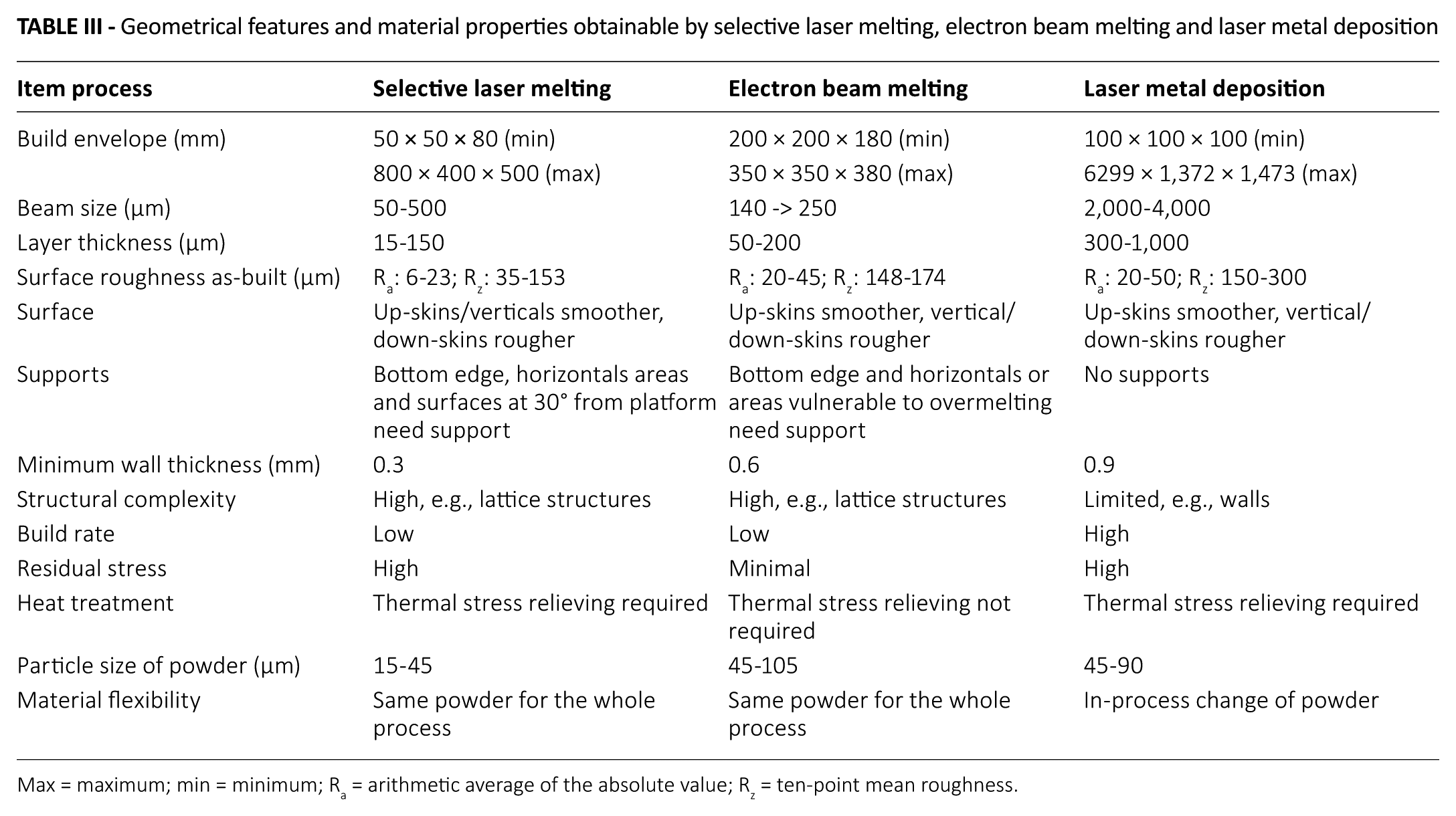
Max = maximum; min = minimum; Ra = arithmetic average of the absolute value; Rz = ten-point mean roughness.
Selective laser melting
SLM or laser powder bed fusion (Fig. 1) is a process that uses the energy of a laser beam, usually an ytterbium fiber laser which can operate at up to 1 kW, to create 3D metal parts by fusing fine metallic powders. A fine metallic powder layer is applied to the building platform by a blade, and the laser beam melts the powder according to the 2D cross-section in a controlled inert environment. Next, the building platform is lowered, and a new layer is applied. The process is repeated until the part built height is completed. The layer thickness can vary from 15 to 150 µm. The beam focus is controlled by the galvanometer, and the movement of the beam is controlled by the F-theta lens.
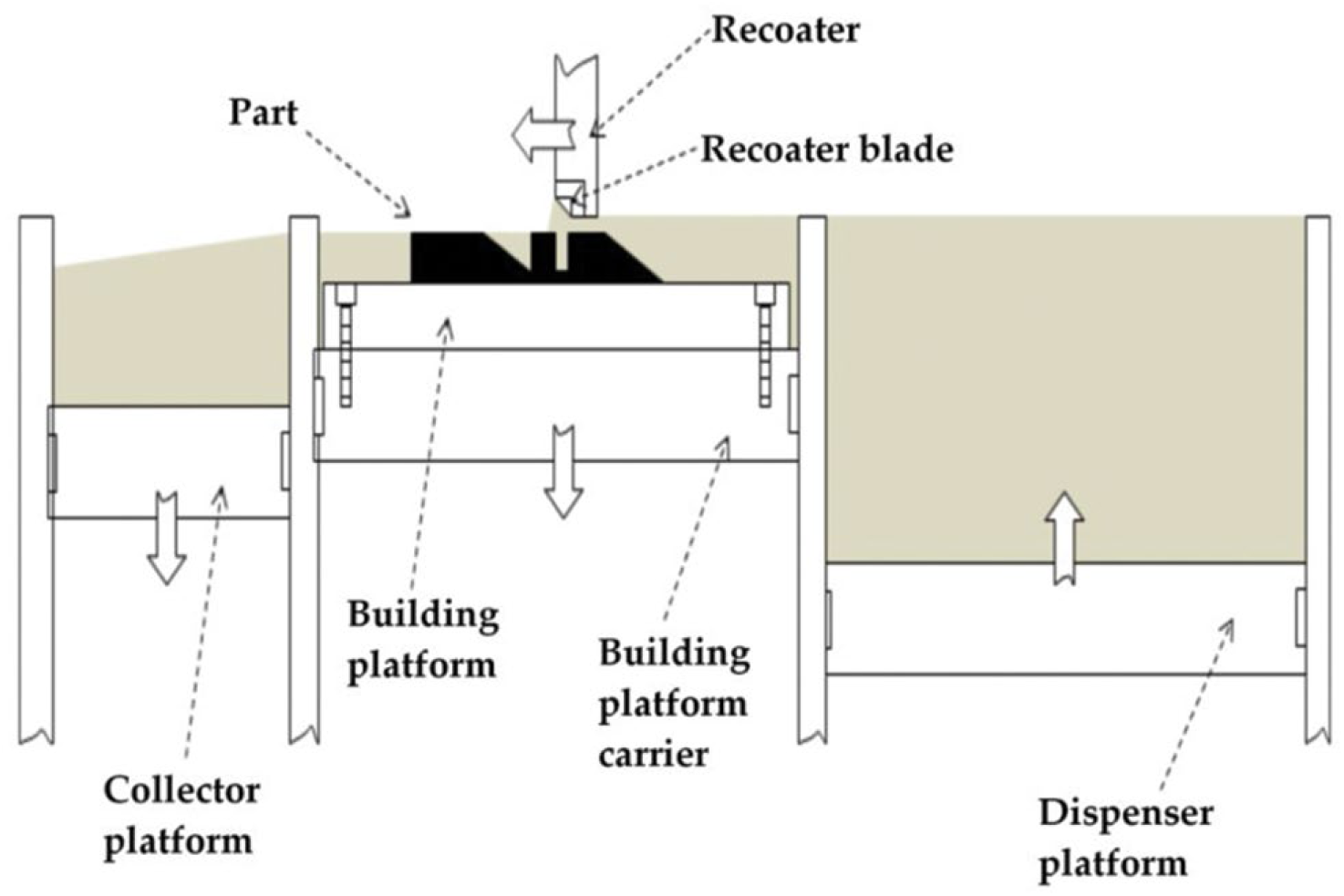
Schematic representation of selective laser melting (SLM) machine.
Compared with cast and forged components, a part produced by the SLM process will have excellent mechanical properties thanks to characteristics of grain refinement, extended solid solubility, chemical homogeneity and reduction in quantity and size of phase segregation. However, due to the Marangoni convection induced by high thermal capillary forces, the melt pool may be unstable causing microstructures uncontrollability (42). The process parameters – laser power, scan speed and scan-line spacing – directly affect the melting behavior of the metallic powder. For this reason, the process parameters need to be carefully controlled to successfully fabricate high-quality biomedical devices.
Electron beam melting
EBM or electron powder bed fusion processing, which was conceived and patented by Arcam AB (Sweden), utilizes electron beam energy to melt the metal powder (Fig. 2). Each layer is obtained through the following steps: spreading of the powder, preheating and sintering using a strongly defocused beam that gives mechanical stability and electrical conductivity to the powder layer, melting using a focused beam, and lowering of the build platform by 1 layer thickness that can vary from 50 to 200 µm. The EBM process takes place in a vacuum (base pressure of about 1 × 10−5 mbar) and at high temperature, specific for the material used, and produces stress-relieved components with a microstructure free from martensitic structures. For these reasons, EBM materials present microstructural features better than cast and comparable to wrought material.
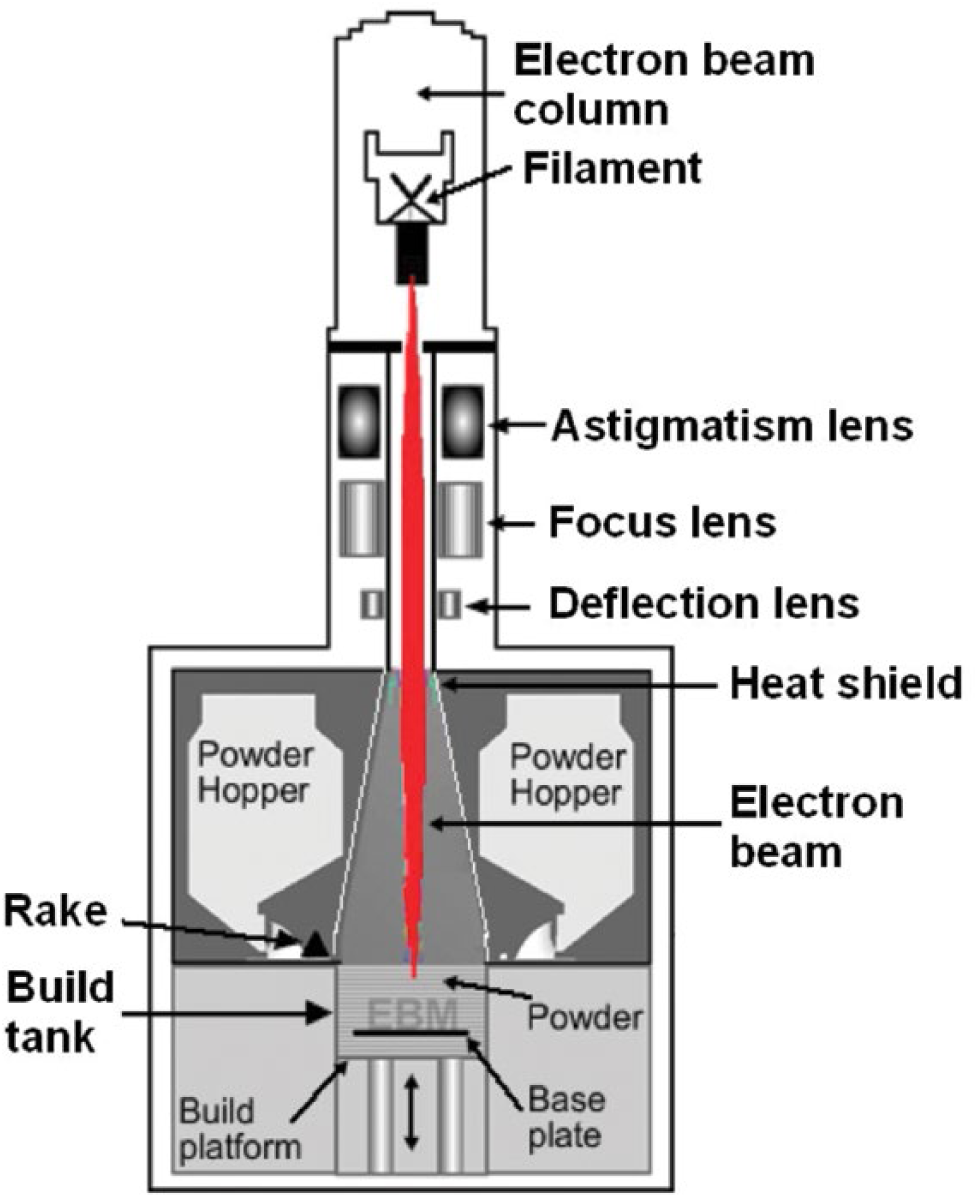
Schematic representation of electron beam melting (EBM) process.
During the melting process, a partial pressure of He is introduced to 2 × 10−3 mbar to maintain the chemical specification of the built material. So this process is suited for materials with a high affinity to oxygen such as titanium alloys. Sometimes the type of microstructure achieved with this technology is not the one desired. For this reason, the effects of heat treatments on microstructure and properties of the part produced by EBM are mainly studied. The EBM technology is used in the biomedical sector for production of standard and custom orthopedic implants.
Laser metal deposition
LMD is a process in which metal powder is injected into the focused beam of a high-power laser under tightly controlled atmospheric conditions (Fig. 3). The focused laser beam melts the powder and generates a small molten pool in base material. The work piece is moved in the
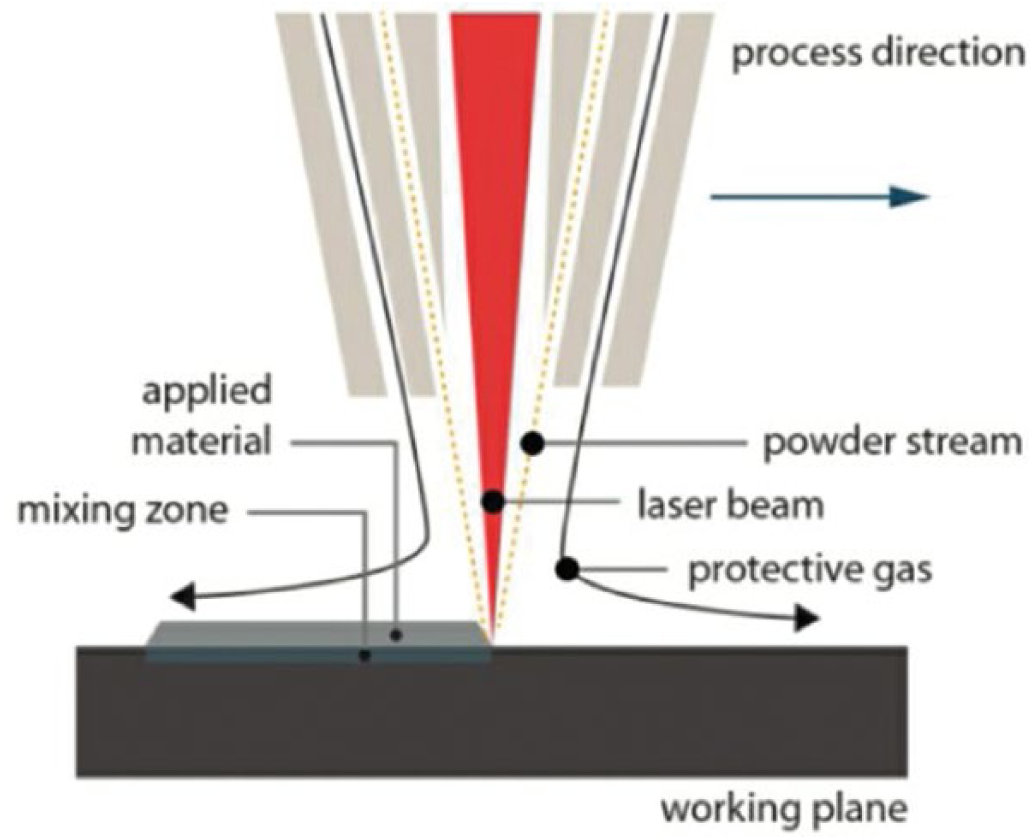
Schematic representation of laser metal deposition (LMD) process.
LMD is a complex process governed by mass and thermal and fluid flow. The processing parameters – laser power, scanning speed, powder flow rate and the shield gas flow rate – greatly influence the properties of the deposited parts, which include deposition height, width and the surface finish and metallurgical and mechanical properties. The process, being dominated by a subtle energy balance which is affected by the part and substrate geometries, ambient temperature, etc., is sensitive to environmental conditions. Furthermore, a constant height is difficult to achieve: typically each layer measures a thickness of 0.3-1 mm.
LMD has been used to produce structures with graded porosity and/or composition, from different biomaterials including titanium, stainless steel and shape memory alloys (46).
Design of cellular lattice structures
AM technologies allow the production of porous surface structures with predictable and predetermined unit cells that are gaining in popularity in the production of biomedical implants thanks to their ability to promote osseointegration and cell proliferation and to achieve suitable mechanical properties, such as compressive strength and elastic modulus, which are similar to those of human bone, so as to avoid stress shielding effects after implantation. However, biomedical implants require precise design of pores and porosities to mimic the different characteristics and mechanical properties of 2 types of bone: cortical bone and trabecular bone. These types of bone have the same composition but different proportions of organic and inorganic materials and degree of porosity. The organization and the combination of cortical and trabecular bones vary according to skeletal region, and according to the applied mechanical loading (47).
In recent years, some researchers have focused their attention on the study of porous titanium structures for biomedical applications to optimize designs and make patient-tailored implants (48-51). From these studies, it was found that, in addition to the pore size and the amount of porosity, the shape of pores can also influence cell proliferation and differentiation (52-55). In general, there are 2 broad categories of cellular structures: stochastic, which have random variations in the shape and size of the cells; and nonstochastic geometries, which have a periodic repetition of lattice structures and can be classified by their shapes and sizes (56-59). Nonstochastic metal structures are preferred because, having no random variations of cell sizes and shapes, they present better mechanical properties (60) and allow a better removal of the unfused powder, compared with stochastic metal foams if fabricated through the powder bed technologies.
Van Bael et al (53) evaluated the effect of nonstochastic structures in terms of pore size and shape, porosity and permeability, on the mechanical properties and in vitro biological outcomes of SLM-produced Ti-6Al-4V scaffolds. The pore shapes affected the permeability of the cells and thus the number of cells attached to the scaffold. The results of this study also showed that a circular cell growth pattern was independent of pore size and shape. This involved the fact that the amount of occlusion of the pore is higher on hexagonal pores compared with triangular or rectangular pores.
Harrysson et al (61) carried out a design by finite element analysis (FEA) and fabrication of titanium hip implants using EBM to reduce stress shielding while maintaining mechanical strength. To reduce the implant stiffness, the solid stems were modified with a periodic lattice structure designed to produce the desired stiffness. The comparison between the simulated model and the built one highlighted the fact that the construction of nonstochastic lattice structures is possible via EBM; however, the orientation of the lattice struts during the manufacture was important. The differences among the surfaces of the struts, with a smooth surface and constant cross-section in the FEA model while there were textured surfaces with slightly varying cross-sections in the fabricated struts, had to be reflected in the design of the implants through use of safety factors. However, this study showed that among the studied configurations (mesh configuration, hole configuration and complete solid), the Ti-6Al-4V stem with mesh appeared to have a better distribution of stress in the proximal portion of the femur.
Ahsan et al (46) investigated the fabrication of porous structures in Ti-6Al-4V by continuous and pulsed LMD. The structures were compared in terms of internal geometry, pore size and part density. The results demonstrated that the 2 methods produced different porous structures: using a pulsed beam method it was possible to obtain a controlled porosity compared with what was produced with a continuous beam. A final irregular structure could be held responsible for premature failure.
Titanium alloys by AM metal powder–based processes
Surface and morphology
It has been demonstrated that osteoblastic cell adhesion, growth and differentiation are supportive for integration of the implant with the bone tissue and are related to surface energy and roughness (62-64). Traditionally, the surface of an implant could be modified by sandblasting, plasma spraying and acid etching to obtain the desired roughness (65, 66). Instead, the implants produced by metal powder–based processes may directly present a suitable surface roughness for osteointegration.
Biemond et al (54) compared the surface topographies and friction coefficients of implants produced by EBM with plasma-sprayed titanium implants and sandblasted implants. The results for attachment, proliferation and differentiation of mesenchymal stem cells on porous surface structures by EBM were promising. This study revealed that the potential bone ingrowth of EBM-produced surfaces was comparable to that of a titanium plasma spray coating. In another study, however, it was demonstrated that the implants fabricated by SLM should be surface treated to further increase bone formation and bridging. In fact, better results were obtained with sandblasted and acid-etched implants compared with the untreated ones (67, 68).
During the process, a large number of spherical Ti particles were partially bonded to the porous walls. After a heat treatment of the specimens at 1300°C in an argon atmosphere to avoid release of these particles, microcavities were formed on the walls, promoting interlocking with the surrounding tissues when implanted in the living body.
A comparative study on trabecular implant surfaces produced by SLM and EBM was carried out by Biemond et al (69). The SLM and EBM specimens presented different morphologies and surface textures: In the EBM specimens, the individual granules could be clearly seen, whereas the surfaces of the SLM specimens were flatter compared with the EBM surfaces. The difference in accuracy between the 2 technologies is probably responsible for the differences in the porosity and surfaces posttreatments.
The conclusions reached in various studies regarding whether or not to make subsequent surface treatments are often contradictory. Ponader et al (70) investigated the responses of osteoblasts to the surface of EBM implants, and they found that the value of the arithmetic average of the absolute values (Ra) of the roughness played a role in the proliferation and differentiation of human osteoblasts: for Ra below 24.9 µm, the surface roughness had a positive effect; when Ra exceeded 56.9 µm, the surface roughness had a negative effect. These results might give an indication of when a surface treatment should be considered for parts produced by SLM and EBM: If the as-built implant already has an Ra less than 24.9 µm, the surface treatment may be unnecessary.
LMD, compared with SLM and EBM, permits the production of structures composed of different biomaterials: Materials with different chemical composition can be changed from layer to layer leading to a graded component (71-73). This allows one to have, e.g., ductility in the bulk and a high level of hardness near the surface. Balla et al (74) deposited pure Zr on Ti, then oxidized the coating using LMD to create oxidized a Zr alloy that exhibited lower friction and superior wear properties for the production of hip and knee implants. Cells on the Ti surface showed a flattened morphology with filopodia extensions covering the entire surface, as opposed to fewer cells on as-deposited Zr coatings. Oxidized Zr coatings and as-deposited Zr coatings showed similar cell morphology, slightly elongated and few filopodia extensions, but the oxidized Zr coatings exhibited more spreading and more cells. The difference in morphology of osteoblast cell attachment between the as-deposited Zr and the oxidized Zr might lead to differences in terms of osteoblast cell growth.
LMD has also been used to coat titanium with tricalcium phosphate ceramics to improve bone cell–materials interactions (75). The effects were evaluated in vitro with human osteoblast cells (OPC1). The results indicated that there were more OPC1 cells attached to the surface of the tricalcium phosphate coating than to the uncoated Ti, and these had proliferated on the coating surface. The coating initiated cell differentiation, extracellular matrix formation and biomineralization.
Mechanical properties
The mechanical properties of the materials depend on their microstructures, which are strongly influenced by the AM processing conditions adopted, consequent to the repeated heating and cooling cycles that they undergo during the layer-by-layer production (34, 76). In particular, starting particles undergo rapid heating to melting temperature, absorbing energy from a laser or electron beam. After the energy source scanning, the melt pool rapidly solidifies, with the development of a fine-grained microstructure; in the processing of the next layers, the solid volume is exposed to heat again and again. This complex thermal cycle leads to metastable microstructures and compositional phases that can also vary in the part volume (34). The depth of the melt pool depends on the heat transfer, which generally differs among AM processes (76). In fact, in LMD, heat transfer involves conduction to the substrate, conduction the built material and convection to the shield gas. On the other hand, in SLM systems, the presence of a powder bed around the part limits heat conduction, and the lower flow rate of the gas reduces the convection contribution. In contrast, in EBM, the heat transfer occurs through conductive loss to the machine and radiative loss from the built surface, with the part heated at high temperatures (between 650 and 700°C (23)) until all layers are realized. The elevated temperatures involved in EBM lead to a different thermal history of the processed materials, which undergo a more limited thermal gradient after solidification. Indeed EBM produces Ti-6Al-4V parts with a coarser microstructure compared with those of SLM or LMD products, due to the lower cooling rates (77, 78). For the same reason, the EBM samples present an overall α + β dual microstructure (79, 80), with a fine lamellar α morphology and a small volume of retained β (34). On the other hand, in SLM and LMD, the very high cooling rate (greater than 104 K/s) and the higher thermal gradient lead to a higher degree of undercooling, with the consequent transformation from β to ultrafine, acicular martensitic α’ phase (34, 81, 82).
Moreover, a common feature for Ti-6Al-4V materials produced through the 3 AM processes is the presence of elongated columnar prior-β grains along the building direction of the parts. Their presence derives from the thermal history of the material (83-86). In particular, these columnar grains depend on the solidification phenomena and melt pool geometry, based on the heat flow, the thermal gradient and the liquid–solid interface velocity of the melt pool, which, for each process, can be strictly controlled through scan velocity, beam power and scan strategy (76).
The microstructural and compositional features above described can explain the different mechanical responses exhibited by final parts. First of all, according to the Hall-Petch relationship, finer microstructures of materials processed through AM lead to higher static strength compared with those fabricated with traditional methods (34).
In addition, the preheating possible in EBM leads to in situ tempering, hindering the formation of martensitic phase and stabilizing the α + β dual microstructure, thus obtaining a good compromise between ultimate tensile strength (UTS) and ε (23, 87). Higher stiffness and strength and lower ductility (E = 118 GPa, UTS = 915 MPa, yield strength [YS] = 830 MPa, ε = 13%) are generally recorded for as-built EBM Ti-6Al-4V materials compared with wrought and annealed ones (E = 104 GPa, UTS = 870 MPa, YS = 790 MPa, ε = 18) (88-91).
On the other hand, being made of α’ martensitic phase, SLM materials possess higher values of YS and UTS and lower ε (34, 89, 92, 93). As-built SLM Ti-6Al-4V alloys possess high YS and UTS values (990 and 1,095 MPa, respectively), but are characterized by a lower ductility (8.1%) compared with hot worked and annealed samples (18.1%) (94). By comparing Ti-6Al-4V specimens produced by both EBM and SLM machines, the different mechanical response is evident: EBM samples have a YS and UTS of 899 ± 4.7 and 978 ± 3.2 MPa, respectively, and an elongation at break value of 9.5% ± 1.2%; while SLM samples exhibit a YS of 1,195 ± 19 MPa, UTS of 1,269 ± 9 MPa and ε equal to 5% ± 0.5% (94).
Considering the LMD process by contrast, Sterling et al (95) produced samples with 908 and 1,038 MPa of yield and UTS, respectively, and an elongation at break of 3.8%. Hence, compared with EBM and SLM results, LMD parts possess lower ductility and intermediate tensile strength values, probably due to the cooling rates involved (34). Table IV summarizes some of the findings in the literature on the mechanical properties and biocompatibility tests of the parts produced through SLM, EBM and LMD.
Mechanical properties and biocompatibility tests of TI-6AL-4V specimens
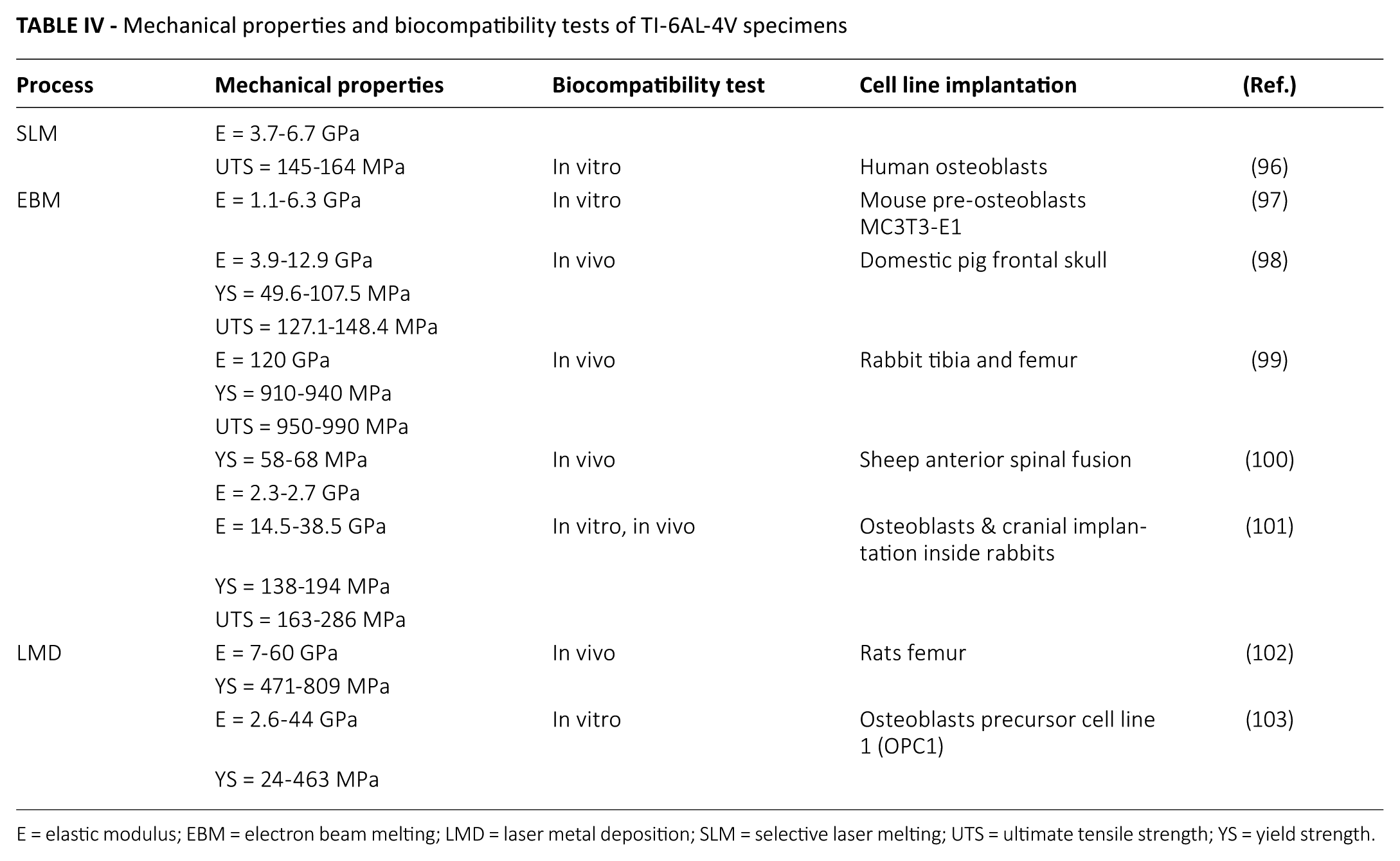
E = elastic modulus; EBM = electron beam melting; LMD = laser metal deposition; SLM = selective laser melting; UTS = ultimate tensile strength; YS = yield strength.
As already stated, during AM, the rapid solidification and thermal cycles lead to the development of metastable microstructures and phases that can easily be modified to achieve better mechanical performances, through thermal postprocessing. In fact, the choice of treatment options can strongly influence grain size and orientation, precipitation and phase evolutions, densification phenomena, stress relief, etc.
For instance, Kasperovich et al (104) conduced different heat treatments and the hot isostatic pressing (HIP) process to evaluate the resultant mechanical and fatigue properties of Ti-6Al-4V. Postprocess heat treatments favored the nucleation of a ductile β phase, resulting in higher elongation at break values: the HIP process allowed improved ductility up to wrought part values (almost 19%) and increased the fatigue life of the as-built parts from 2.3 × 103-5.6 × 103 cycles to 1.5 × 105-3 × 105 cycles.
In the case of LMD materials, after thermal postprocessing, higher YS (between 827 and 965 MPa) and UTS values (between 896 and 1,000 MPa) are obtained (105). Regarding elongation at break, a particular effect is observed: After postprocessing, a 10% mean ductility is recorded for wrought samples, while values between 1% and 16% are found for the laser engineered net shaping (LENS) specimens. Such behavior has been observed in several other studies (84, 106-109) and can be related to the ultrafine microstructure possessed by LMD materials, but also to internal porosities, residual stresses and excessive surface roughness, which also favor embrittlement of the part.
Industrial scale-up
The reason AM processes have attracted so much interest in the medical field is based on the feasibility of the creation and production of patient-specific implants, precisely reproducing patient tissues and organs (1, 21, 110-112). AM permits the replication of the complex structure of bones, muscles, nerves and vasculature, and as a consequence, surgical interventions appear easier, the risk of implant rejection is reduced and injury healing is accelerated (21). Compared with traditional manufacturing techniques, the production of medical devices has become faster and cheaper: Reduction of tooling operations and of wasted material (by up to 75%) and superior mechanical strengths are some of the advantages offered by AM (113).
Cronskär et al (114) analyzed the production of hip stem prostheses with EBM and found a 35% cost reduction compared with conventional manufacturing. The Wohlers Report 2015 (16) stated that more than 100,000 acetabular implants have been produced with AM processes, and about 50,000 of them have been implanted in patients in recent years. Such data give a panoramic view of how the industrial sector’s interest and the practical applications of AM have grown (115). Furthermore, the possibility of processing different materials, with enhanced or comparable properties compared with those of traditional methods, in addition to new design approaches, such as those for the production of controlled porosity scaffolds, opens new horizons to industrial applications of AM (17, 113).
Conclusion
Titanium and its alloys, thanks to their excellent biocompatibility, mechanical properties and good corrosion resistance, are the most interesting metallic biomaterials for orthopedic and dental implants. Until a few years ago, titanium processing via AM technologies was given little consideration by the medical industry due to the relatively elevated costs. However, in recent years, AM metal technologies such as SLM, EBM and LMD are becoming increasingly popular in the biomedical field because of the ability to build metals with customized porous architectures which permit tailored cell morphologies enabling the cell proliferation and differentiation required for bone ingrowth. Although these AM processes share the same additive philosophy, each technology has its constraints and advantages in terms of useable materials, processing procedures and applicable situations. Based on the fact that titanium alloys can be successfully employed in the biomedical field, this review has discussed the potentialities of the main metal additive technologies that can be used, thus seeking to provide support for potential designers and material engineers. Only with an overview of the whole picture, can they understand which technology involves the best trade-offs for a specific application.
Footnotes
Abbreviations
AM Additive manufacturing
ASTM American Society for Testing and Materials
CAD Computer-aided design
CP Commercially pure
E Elastic modulus (or Young modulus)
EBM Electron beam melting
LMD Laser metal deposition
SLM Selective laser melting
UTS Ultimate tensile strength
YS Yield strength
Disclosures
Financial support: No grants or funding have been received for this study.
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
