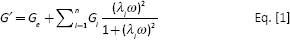Abstract
Background
The biochemical, mechanical and topographic properties of extracellular matrix are crucially involved in determining skeletal muscle cell morphogenesis, proliferation and differentiation. Human elastin-like polypeptides (HELPs) are recombinant biomimetic proteins designed to mimic some properties of the native matrix protein; when employed as myoblast adhesion substrates, they stimulate in vitro myogenesis. Given the influence that the biophysical properties of extracellular matrix have on skeletal muscle cells, the aim of this work was to investigate the effects of HELP hydrogels on myoblasts’ viability and functions.
Methods
We recently synthesized a novel polypeptide, HELPc, by fusing the elastin-like backbone to a 41aa sequence present in the α2 chain of type IV collagen, containing two arginyl-glycyl-aspartic acid (RGD) motifs. To obtain hydrogels, the enzymatic cross-linking of the HELPc was accomplished by transglutaminase. Here, we employed both non-cross-linked HELPc glass coatings and cross-linked HELPc hydrogels at different monomer densities, as adhesion substrates for C2C12 cells, used as a myoblast model.
Results
By comparing cell adhesion, proliferation and differentiation, we revealed several striking differences. Depending on support rigidity, adhesion to HELPc substrates dictated cell morphology, spreading, focal adhesion formation and cytoskeletal organization. Hydrogels greatly stimulated cell proliferation, particularly in low-serum medium, and partially inhibited myogenic differentiation.
Conclusions
On the whole, the results underline the potential of these genetically engineered polypeptides as a tool for dissecting crucial steps in myogenesis.
Introduction
Eukaryotic cells have the capacity of sensing and decoding not only the chemical composition of the extracellular matrix (ECM) but also its physical properties such as dimensionality, stiffness and architecture (1–2–3). Moreover, there is now much evidence that several pathologies, such as cardiomyopathies, atherosclerosis, muscular dystrophies and cancer, are associated with alterations in both the chemical and mechanical properties of the ECM (4). Accordingly, scaffolds and matrices are increasingly employed in soft tissue engineering due to their ability to support cell survival and activity, acting both as a mechanical support and as cell interactors and modulators (5). Compared with their natural counterparts, ECM proteins obtained by recombinant DNA technology have intrinsic advantages, including reproducible macromolecular composition, sequence and molecular mass, and overcoming the potential transmission of pathogens related to polymers of animal origin (6). Recombinant elastin-like polypeptides are modeled after the amino acid sequence of the native elastin, an abundant ECM protein, which plays a pivotal role in tissue biomechanical properties and modulates a variety of cellular responses (7, 8). In our laboratory, synthetic genes based on the repeated hexapeptidic motifs that characterize human tropoelastin have been cloned and expressed, and the resulting recombinant proteins, human elastin-like polypeptides (HELPs), have been described and characterized (9,10). Three of these polypeptides, employed as adhesion substrates for C2C12 myoblasts, stimulate in vitro myogenesis to a different extent, strictly dependent on their primary structure (11). Among the HELP family of polypeptides, both the prototype, HELP (12), and the novel, fusion peptide HELPc (11) contain a repetitive, hydrophilic sequence accommodating Gln and Lys residues which can be enzymatically cross-linked, giving rise to hydrogel matrices (13, 14). To exploit the potential advantages of HELP-based hydrogels on myoblast functions, we cultured C2C12 cells on HELPc hydrogels at different monomer concentration, with the aim of coupling the biochemical properties of HELPc polypeptide with the mechanical features of hydrogels. By comparing the responses of C2C12 cells seeded on HELPc hydrogels or on non-cross-linked HELPc glass coatings, we found some intriguing differences in cell adhesion, morphology, proliferation and differentiation, indicating that HELPc hydrogels can be employed as tools for testing the physiological consequences of cell–biomaterial interactions.
Methods
Reagents, antibodies and fluorochromes
Unless otherwise stated, reagents were purchased from Sigma-Aldrich (St. Louis, MO, USA). Bacterial transglutaminase (TGase) from Streptomyces mobaraensis was supplied by N-Zyme BioTec GmbH. Anti-vinculin antibody (mouse monoclonal, V9131; Sigma-Aldrich, St. Louis, MO, USA) was employed at 1:200 dilution. Antibody against myosin heavy chain (rabbit polyclonal antibody, H-300; Santa Cruz Biotechnology, Santa Cruz, CA, USA) was used at 1:30 dilution. Alexa Fluor 488-conjugated AffiniPure goat anti-mouse IgG (Jackson ImmunoResearch Laboratories, West Grove, PA, USA) was used at 1:100 dilution. Goat anti-rabbit fluorescein isothiocyanate (FITC)–conjugated affinity purified IgG (Jackson ImmunoResearch Laboratories, West Grove, PA, USA) was employed at 1:200 dilution. 4’,6-Diamidino-2-phenylindole (DAPI; Sigma-Aldrich) was employed at 100 ng/mL. Alexa Fluor 594 phalloidin (Molecular Probes, Eugene, OR, USA) was employed at 0.008 U/coverslip.
HELPc synthesis and preparation of cross-linked HELPc hydrogels
The sequence of HELPc is reported in Figure 1A, and its synthesis has been previously described (9, 11). Briefly, the synthetic gene of the HELP polypeptide was fused with the 41aa coding sequence corresponding to the 854-895 region of the collagen IV α2 chain (NP_001837.2), exploiting the unique DraIII site in the expression vector that allows the in-frame insertion at the C terminus of the polypeptide (12). The final construct was verified by sequencing, and the recombinant product was expressed in the C3037 E. coli strain (New England Biolabs, Ipswich, MA, USA). Expression and purification were carried out as described previously (9). The recombinant polypeptide obtained was analyzed by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE), and the purified product was lyophilized for long-term storage. The lyophilized polypeptide was re-dissolved in water and sterilized by filtration (0.22 µm). Protein concentration was verified by the Bradford method and adjusted to the desired level by dilution.
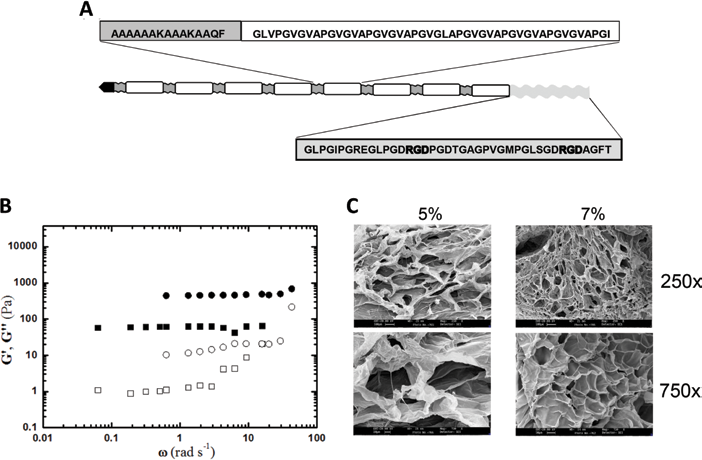
Structure and hydrogel characterization of the novel human elastin-like polypeptide (HELPc). (
Non-cross-liked HELPc coatings were obtained by depositing 0.1 mg/mL of aqueous protein solution onto borosilicate glass coverslips; 0.1 mL of coating solution was used per square centimeter surface area, thus resulting in 10 μg polypeptide/cm2. Coating solution was allowed to dry in a tissue culture hood, and cells were seeded directly on the polypeptide surface. Enzymatic cross-linking was performed in a tissue culture hood under sterile conditions. To obtain HELPc hydrogels, the polypeptide was cross-linked with bacterial TGase. The recombinant protein was dissolved in 10 mM Tris/HCl, pH 8, to a final concentration of 5% and 7% w/v. Solutions (100 µL each) were stored in an ice bath for 30 minutes to promote solubilization. Subsequently, 0.53 U of TGase (133 U/mL) was added, and the reaction mixture was quickly mixed. Then 15 µL of each polypeptide solution was dropped on round glass coverslips (13 mm) and incubated at 10°C for 2 hours. Cross-linking was completed after overnight incubation at 4°C. Hydrogels were bathed with 70% ethanol and then rinsed thoroughly with ultrapure water. Before cell seeding, hydrogels were washed extensively with phosphate-buffered saline (PBS).
Rheological analysis and determination of shear modulus
Rheological measurements were performed on 5% and 7% HELPc hydrogel specimens under oscillatory shear conditions to determine the extension of the linear viscoelasticity regime (stress sweep tests at 1 Hz) and the mechanical spectra (frequency sweep), respectively. The complex viscosity (η*), the storage (G’) and loss (G”) moduli of the hydrogels were recorded in the frequency range 0.01-20 Hz (stress = 4 Pa, within the linear regime). All of the tests were carried out with the controlled stress rheometer Rheostress Haake RS 150 operating at 25°C. A serrated plate PP20 Ti was employed for the mechanical spectra of the hydrogels to prevent slippage effects. In the case of the hydrogels, the contact between the serrated plates and the sample was verified through stress sweep tests performed at different gap values, and a 1.2-mm gap was finally selected.
SEM analysis
Samples of 5% and 7% (w/v) of HELPc hydrogel were prepared as described above (the section “HELPc synthesis and preparation of cross-linked HELPc hydrogels”) using cylindrical molds (4-mm diameter x 4-mm high). After hydrogel formation, samples were extensively washed in distilled water, frozen at -20°C and lyophilized. Slices were cut, mounted onto stubs using a double-sided adhesive and sputter coated with gold. Analysis was performed using a Leica Stereoscan 430i scanning electron microscope (SEM).
Atomic force microscopy analysis
Atomic force microscopy (AFM) was used to characterize HELPc dehydrated hydrogels’ thickness and morphology. AFM images were acquired using a commercially available microscope (Asylum Research MFP-3D™ Stand Alone AFM; Oxford Instruments, Santa Barbara, CA, USA) endowed with a 90 × 90 × 15 μm closed-loop metrological scanner. All AFM measurements were carried out in air at room temperature working in dynamic mode. HELPc hydrogels were carefully washed using Milli-Q water and gently dried in a nitrogen oven overnight at room temperature. Cantilevers used were characterized by a free resonance frequency of about 65 kHz and a force constant of about 0.6 nN/nm (HQ:NSC36/C; MikroMasch Co., Tallinn, Estonia). Images of 512 × 512 pixels were acquired at 0.5 lines/s scan speed to assess the morphology of dehydrated hydrogels. Thickness was calculated as height difference between the film upper surface and the glass surface underneath previously exposed by scratching away the gel with a scalpel. Heights were evaluated as mean value of 10 height profiles traced in different positions perpendicularly to a single scratch line. Gwyddion software (15) was used to analyze AFM images and to compute surface roughness (Ra), while all statistics and data processing were performed using Igor Pro software (16).
Cell culture and differentiation protocol
Mouse myogenic C2C12 cells (American Type Culture Collection; ATCC CRL-1772) were maintained as exponentially growing myoblasts in a growth medium consisting of Dulbecco's modified Eagle's medium (DMEM) supplemented with 20% heat-inactivated fetal calf serum, 4 mM L-glutamine, 100 IU/mL penicillin and 100 μg/mL streptomycin. Cells were cultured on 10-cm polystyrene Petri dishes at 37°C in a 5% CO2 incubator. Cell were serially passed at about 75% confluence every 2-3 days. For adhesion assays, cells were plated at a density of 104 cells/cm2, and allow to adhere for 5 hours. To obtain cell differentiation and myotube fusion, cells were plated at a density of 5 × 103 cells/cm2. Forty-eight hours after plating, cells were shifted to differentiation medium, consisting of DMEM supplemented with 2% heat-inactivated horse serum, 4 mM glutamine, 100 IU/mL penicillin and 100 μg/mL streptomycin.
Immunofluorescence
The general protocol for immunofluorescence experiments is described here. Coverslips were washed 3 times with PBS and then fixed with 4% paraformaldehyde for 30 minutes at 4°C. After fixation, samples were blocked with a solution containing 5% normal goat serum, 0.1% Triton X-100 in PBS for 10 minutes. Samples were incubated overnight at 4°C with a primary antibody at the appropriate dilution. After 3 washes of 10 minutes with PBS plus 0.1% Triton X-100, coverslips were incubated with the secondary antibody (or other fluorochromes) for 2 hours at 4°C. Samples, washed 3 times for 10 minutes with PBS plus 0.1% Triton X-100, were then mounted onto slides and visualized under a Leica DMLS fluorescence microscope (Leica Microsystems, Wetzlar, Germany). Cells were visualized under a Leica DMLS fluorescence microscope (Leica Microsystems, Wetzlar, Germany). Images were acquired with a Leica DC300F camera, coupled to Leica IM50 acquisition software. Image sizing, cropping and overlay were performed with Adobe Photoshop CC (Adobe Systems Inc., San Jose, CA, USA). Image analysis was performed with ImageJ (17).
Cell viability and proliferation
Cells were seeded at a density of 5 × 103 cells/cm2 on 13-mm glass coverslips which were either coated with HELPc or covered with HELPc hydrogels; coverslips where then placed in 24-well plate multiwells. Forty-eight hours after plating, cells were shifted to differentiation medium. Measurements were performed at 5, 24, 48 and 72 hours after plating. Cell proliferation was evaluated by counting nuclei. Samples were fixed with 4% paraformaldehyde for 30 minutes at 4°C and then processed for nuclei staining using DAPI at a final concentration of 100 ng/mL. Nuclei were counted with ImageJ software. Cell viability was evaluated with the (2-(4-iodophenyl)-3-(4-nitophenyl)-5-(2,4-disulfophenyl))-2H-tetrazoilium monosodium salt (WST-1), provided in a premix electrocoupling solution (Roche Applied Science, Mannheim, Germany) according to the manufacturer's instructions.
Differentiation assays
C2C12 cells were induced to differentiate and, at 5 days, processed for immunofluorescence against myosin heavy chain (MHC). A cell containing 3 or more nuclei was considered a myotube. The total number of nuclei and of MHC-positive myotubes was counted in 10 random fields (>400 total nuclei) for each condition. The fusion index was calculated as the ratio of the nuclei number in MHC-positive myotubes versus the total number of nuclei in the field. The average number of nuclei per myotube was also determined.
Statistical Analysis
Data were analyzed with OriginPro software (version 7.5; OriginLab Corp., Northampton, MA, USA). Statistical significance was determined using 1-way ANOVA after Levene's test for homogeneity of variances, and followed by the Scheffé test for multiple comparisons. Unless otherwise stated, results were expressed as means ± standard deviation (SD). Differences with a p value <0.05 were considered statistically significant.
Results
Properties of HELPc hydrogels
The sequence scheme of HELPc is reported in Figure 1A. The elastin-like backbone of the polypeptide is fused, at the C terminus, with a 41aa sequence from the collagen type IV α2 chain isoform, allocating two arginyl-glycyl-aspartic acid (RGD) motifs. The polypeptide was designed to genetically encode hydrophilic domains containing glutamine and lysine residues which can undergo enzymatic cross-linking by transglutaminase (12, 13). Accordingly, the Ala-rich, hydrophilic sequence of the elastin-like polypeptides containing the Lys and Gln residues is also called the cross-linking domain (18). Concentrations of 5% and 7% w/v were selected as optimal for the cross-linking reaction because lower concentrations (4%) failed to generate stable hydrogels, whereas concentrations higher than 7% w/v tended to give solubility problems, as previously described (13). Indeed, a peculiar and well-known property of elastins (both natural and recombinant) is coacervation (or “self assembly”); under appropriate conditions of concentration, ionic strength and temperature, the protein separates from the solution as a second phase (19).
Rheological measurements under oscillatory shear were performed to characterize HELPc hydrogels. Figure 1B reports the mechanical spectrum of the HELPc hydrogels at the concentrations of 5% and 7% (squares and circles in Fig. 1B, respectively). It can be noticed the elastic modulus of the 5% hydrogel is markedly lower (1 order of magnitude) than that of the 7% hydrogel. However, in both cases, G’ is higher than G” and shows little variation within the pulsation range explored. For the sake of comparison, a non-calcium-saturated 1.5% alginate hydrogel shows an elastic modulus which is approximately 1 order of magnitude higher than the one displayed by the 7% HELPc sample (20).
The frequency sweep test results were interpreted in terms of the generalized Maxwell model composed of a sequence of elements in parallel (spring and dashpot) to which an additional spring has been added (21). The storage and loss moduli can be modeled as a function of the pulsation ω according to the following equations:
where n is the number of Maxwell elements considered, Gi, ηi and λi represent the spring constant, the dashpot viscosity and the relaxation time of the ith Maxwell element, respectively. Ge is the spring constant of the last Maxwell element which is supposed to be purely elastic. The fitting of the experimental data was performed assuming that relaxation times are not independent of each other, but they are scaled by a factor 10. Hence, the parameters of the model are Ge, ηi and λ1. The number of the Maxwell elements was selected based on a statistical procedure, to minimize the product χ2*Np, where χ2 is the sum of the squared errors, while Np (=2+n) indicates the number of fitting parameters.
The use of a generalized Maxwell model allowed a determination of the shear modulus, G, with Equation [3]:
For the 7% HELPc hydrogel, G was calculated to be 512 ± 28 Pa, while it is reduced to 63 ± 12 Pa when the 5% HELPc hydrogel is considered. As a comparison, a calcium-saturated 1% alginate hydrogel showed a G of approximately 55 kPa (20).
The SEM analysis of cross-sectional interior structures of lyophilized HELPc hydrogels is shown in Figure 1C. Both 5% and 7% matrices exhibited a porous honeycomb-like structure with an irregular shape in which the 3-dimensional, interconnected macropores represent water-filled areas, while the pore size depends on the monomer concentration. Specifically, the 5% HELPc hydrogels were characterized by a mean pore diameter of 227 ± 36.6 µm (n = 50), while the 7% HELPc hydrogels exhibited a much denser structure with a mean pore size of 74 ± 32.5 µm (n = 56; p<0.001).
HELPc hydrogels appeared opaque and wax-like on visual inspection (Suppl Fig. 1A, available online as supplementary material at www.jab-fm.com). The different compactness of the 2 matrices was highlighted when hydrogels were cast in cylindrical molds. As shown in Supplemental Figure 1B (available online as supplementary material at www.jab-fm.com), after removing the excess water, the 7% hydrogel retained its shape, whereas the 5% tended to collapse. The hydrogel surfaces, observed under phase contrast microscope, appeared uniform, smooth and crossed by several folds (Suppl Fig. 1C, available online as supplementary material at www.jab-fm.com), presumably resulting from shrinkage during the cross-linking process.
AFM was employed to characterize some properties HELPc hydrogels. Preliminary attempts to determine by AFM the morphology and the mechanical properties of HELPc hydrogels were unseccessful given that the different probes employed for indentation tests tended to stick to the hydrogel surfaces (in particular to tips characterized by large contact surfaces, such as sphere-endowed cantilevers), thus preventing the measurement of hydrogel morphology and elastic modulus. To partially overcome this problem, obtaining in any case a preliminary characterization of the 2 HELPc-based matrices, surface texture, morphology and thickness were evaluated on dehydrated hydrogels (DHs). AFM images of DH surfaces (Suppl Fig. 1D, available online as supplementary material at www.jab-fm.com) reveal similar clouded morphology for the 2 samples, which appeared substantially different from the previously reported grainy and uneven distribution of non-cross-linked HELPc coatings (11). The roughness of the 5% HELPc hydrogels (Ra of about 60.8 nm) was slightly higher than that of the 7% sample (Ra of about 32.5 nm). DH height profile measurements over scratched hydrogels (see “Methods”) revealed that 7% samples were significantly thicker than 5% (3.702 ± 1.431 µm vs. 1.583 ± 0.446 µm, p<0.01). This is likely due to the higher cross-linking level of 7% DH.
Cell adhesion
Adhesion and spreading of C2C12 cells were evaluated 5 hours after seeding on 5% and 7% HELPc hydrogels. Adhesion properties were compared with those exhibited by the same cells adhering on HELPc coated coverslips (Hcc) obtained by adsorbing the polypeptide to glass coverslips (11). The stability of non-cross-linked HELP polypeptides adsorbed to plastic or to glass coverslips has been previously assessed by X-ray photoelectron spectroscopy (22) and by immunofluorescence and AFM (11). By immunostaining vinculin to visualize cell cytoplasm and focal adhesions, several morphological and functional differences could be observed (Fig. 2). Cells seeded on Hcc appeared flattened, well spread and displaying, in many cases, wide lamellipodia (Fig. 2A). On 5% hydrogels, most myoblasts appeared stellate due to protrusions expanding in all directions and often branching. Myoblasts adhering on 7% hydrogels mostly displayed an elongated and polarized morphology, characterized by a fan-like shape and a long trailing edge. The histogram distribution of cell aspect ratio (AR) and area accounts for the heterogeneity of cell populations, and highlights the differences observed in Figure 2A. Cell AR is an assessment of cell morphology, as the ratio between the length of the major axis and the minor axis of a single cell. Accordingly, the more elongated the cell, the higher the AR, whereas a polygonal cell exhibits an AR close to 1. Cells seeded on Hcc show the prevalence of a polygonal-circular shape and a considerable degree of spreading (area). Cells seeded on 5% hydrogels spread less on the substrate, and their AR values indicate a more elongated morphology. Conversely, cells seeded on 7% hydrogels show a broader distribution of both AR and cell area, with a considerable fraction of the cell population showing an increased length and a higher degree of spreading.
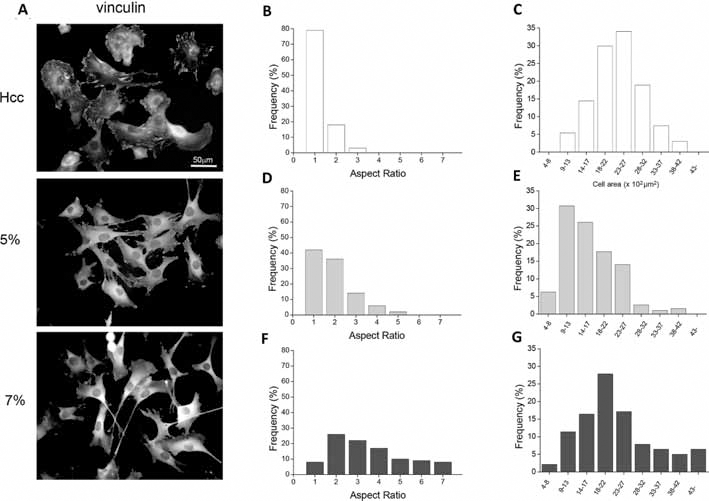
Immunofluorescence and morphometric analysis of cells. C2C12 cells were allowed to spread for 5 hours after seeding on the studied novel human elastin-like polypeptide (HELPc)–coated coverslips (Hcc) or on 5% and 7% HELPc gels. (
Given the role of cytoskeleton in shaping cell morphology and the part played by vinculin in coordinating the focal adhesion network and organizing the actin cytoskeleton dynamics (23), we compared f-actin organization and vinculin distribution 5 hours after seeding (Fig. 3). Cells seeded on Hcc where characterized by a highly organized actin meshwork with numerous stress fibers intersecting and ending at focal adhesions, suggesting a state of isometric contraction (24). Vinculin staining showed marked elongated spots, distributed not only at the cell periphery, but scattered throughout the cell soma, indicating an organized network of focal adhesions. Centrally located in the cell body, fibrillar adhesions were also detectable. In cells adhering to 5% hydrogels, the network of actin filaments appeared less well defined, with stress fibers running along the main cell axis and ending at the terminals of cell protrusions. Vinculin localized both in the cytosol, where it appeared diffusely distributed, and at the cell membrane, where dotted and dashed spots decorated the cell margins in correspondence to the ending of protrusions, forming focal contacts and small focal adhesions. Cells adhering on 7% hydrogels where characterized by a tightly organized actin meshwork with abundant stress fibers running along the whole cell soma. The thin trailing edge was also rich in filamentous actin. Vinculin staining showed a marked enrichment in elongated spots, distributed both at the cell periphery and throughout the cell soma, indicating a diffuse and organized network of focal adhesions.
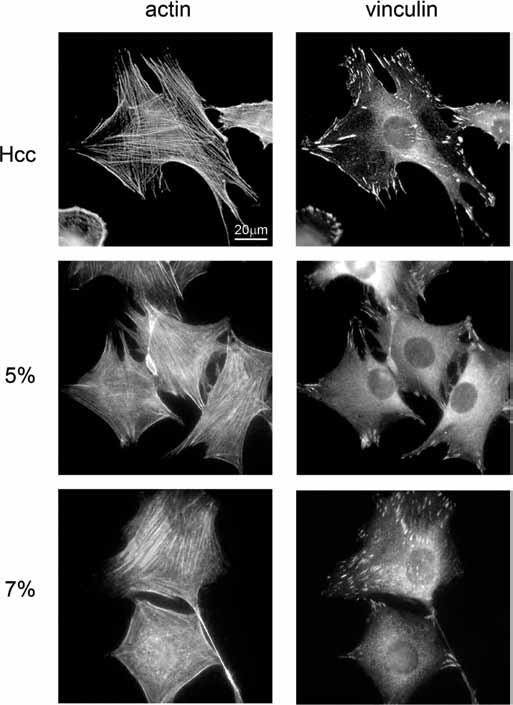
Immunofluorescence of cytoskeleton and focal adhesions. C2C12 cells were allowed to spread for 5 hours after seeding on the studied novel human elastin-like polypeptide (HELPc)–coated coverslips (Hcc) or on 5% and 7% HELPc gels. F-actin was labeled with Alexa Fluor 594 phalloidin, immunofluorescence-labeled vinculin. The results are representative of 3 independent experiments.
Cell viability and growth
The inhibition of cell proliferation is required for myogenesis to occur (25). Accordingly, we quantified cell viability and turnover before and after addition of differentiation medium. After the initial growth phase in growth medium, sampled at 5, 24 and 48 hours after cell seeding, cells were switched to differentiation medium and cultured for another day (72 hours after seeding). At these time points, in fact, cell-to-cell fusion has not yet started and polynucleated cells are not detectable. Cell turnover was evaluated by counting the cell nuclei stained with DAPI (Fig. 4A) and by measuring cells’ metabolic activity with WST-1 (Fig. 4C). Figure 4A shows the number of nuclei at the different time points for each culture condition. After 5 hours, the number of cells adhering to the 3 substrates was not significantly different, even though a tendency to higher adhesion was observed for the Hcc (Hcc 110 ± 31 cells; 5% hydrogels 90 ± 37 cells; 7% hydrogels 77 ± 23 cells). At 24 hours, the number of cells on either sample increased, indicating that proliferation started early after seeding. Moreover, the cell number present on both hydrogels was significantly higher than that present on Hcc. At 48 hours, proliferation continued maintaining the difference between Hcc and hydrogels. At this time the culture medium was shifted to differentiation medium, thus lowering by tenfold the concentration of growth factors. Seventy-two hours after seeding, the most striking differences were observed among the different culture conditions. A considerable increase in cell number was observed for cells grown on hydrogels, while the number on Hcc increased to a far lesser extent. To provide a more dynamic description of such variations, we calculated, for the 3 culture conditions, the proliferation rate, by normalizing the nuclei numbers to the initial density (Fig. 4B). Compared with cells seeded on Hcc, myoblasts cultured on both hydrogels showed a higher growth rate already at 24 and 48 hours. Strikingly, the shift to differentiation medium boosted proliferation of cells growing on hydrogels, but failed to affect the growth rate on Hcc. Similar results were obtained by measuring the cell metabolic activity with the WST-1 assay (Fig. 4C, D).
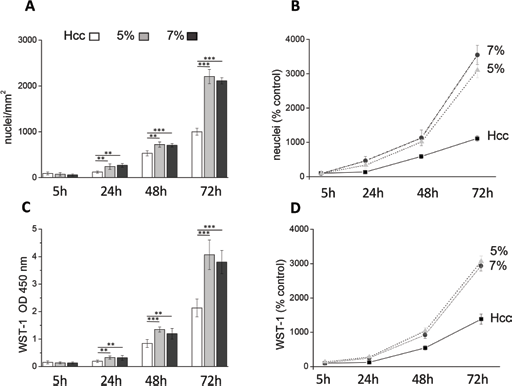
Cell viability and proliferation. C2C12 cells were cultured for 5, 24, 48 and 72 hours on the studied novel human elastin-like polypeptide (HELPc)–coated coverslips (Hcc) or on 5% and 7% HELPc gels. At 48 hours, growth medium was replaced with differentiation medium to start differentiation. (
Myotube differentiation
During differentiation, the C2C12 cells align and fuse together to form myotubes. To compare the effect of the 3 HELPc substrates on myogenic differentiation, C2C12 cells were shifted to a low serum medium (differentiation medium) to promote the myotube formation. Using phase contrast microscopy, morphological changes were compared at 5 days of differentiation (Fig. 5A). Although a diffuse network of elongated cells was present in all conditions, myotubes developed on Hcc appeared longer and wider with respect to those formed on both 5% and 7% hydrogels, and the first signs of branched structures could be observed. By comparing the morphology of myotubes formed on hydrogels, we noticed that longer and wider myotubes tended develop on 7% gels. After 5-6 days of differentiation, cells started to clump and detach from hydrogels, whereas myotubes developed on Hcc tended to peel off after 9 days, presumably due to spontaneous contractions.
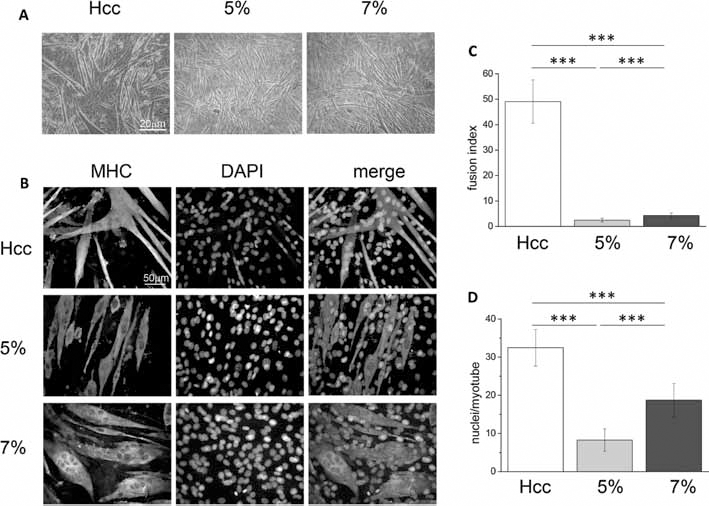
Myotube differentiation: C2C12 cells were plated on the studied novel human elastin-like polypeptide (HELPc)–coated coverslips (Hcc) or on 5% and 7% HELPc gels. After 48-hour proliferation in growth medium, the medium was changed to differentiation medium, and the cells were allowed to differentiate. (
During differentiation of C2C12 cells, the newly formed myotubes express several differentiation markers, among which the muscle-type MHC is considered one of the latest (26). Hence, we investigated the expression of MHC at 5 day of differentiation, performing a qualitative and quantitative analysis by indirect immunofluorescence (Fig. 5B). Myotubes formed on Hcc appeared long, multinucleated and often branched, due to multiple events of cell-to-cell fusion. Compared with cells adhering to 5% hydrogels, myotubes developed on 7% hydrogels appeared wider and more multinucleated. To quantitatively compare the differences observed in immunofluorescence images, we calculated the fusion index (Fig. 5C) and the average number of nuclei in MHC-positive myotubes (Fig. 5D). The fusion index represents the ratio of the nuclei number in MHC-positive myotubes (containing at least 3 nuclei) versus the total number of nuclei in the field. As shown in Figure 5C, the fusion index of myotubes grown on Hcc was considerably higher (49.0% ± 8.5%) than that of cells grown on either hydrogel. However, the comparison of fusion index for myotubes developed on hydrogels revealed that it was greater in cells grown on 7% hydrogels (7% hydrogels: 4.3% ± 1.0%; 5% hydrogels: 2.4% ± 0.7%). The number of nuclei per myotube (Fig. 5D) was higher for Hcc (32.0 ± 4.7 nuclei). However, as shown by Figure 5D, also the number of nuclei present on myotubes developed on 7% hydrogels was elevated (19.0 ± 4.0 nuclei) and significantly higher than that present on 5% hydrogels (8.0 ± 3.0 nuclei). The morphometric analysis of myotubes developed on the different culture conditions is reported in Figure 6. Myotubes developed on Hcc (Fig. 6A, B) were composed of 2 main populations: one consisting of elongated, thin cells, with many nuclei aligned along the main cell axis; the other comprising shorter and wider cells, highly branched, due to multidirectional cell-to-cell fusion. The size of myotubes developed on 5% hydrogels is reported in Figure 6C, D; the majority of them showed a length of between 100 µm and 200 µm and an average width of 18 µm. Myotube size significantly changed when cells differentiated on 7% hydrogels, with an increase in both length and width. As shown in Figure 6E, an average increase in myotube length (200-300 µm) and, most of all, an average 2.6-fold increase in width (47 µm; Fig. 6F) characterized these cells.
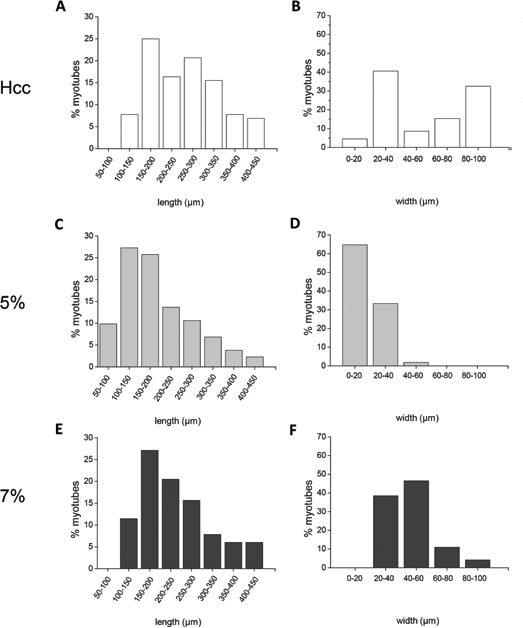
Myotube morphometric analysis: C2C12 cells were plated on the studied novel human elastin-like polypeptide (HELPc)–coated coverslips (Hcc) (
Discussion
Human elastin-like polypeptides, as non-cross-linked coatings, have been exploited to promote myoblast adhesion proliferation and differentiation (11, 22). Moreover, they have been successfully employed to improve the contractile activity of C2C12 cells in collagen/HELP composite scaffolds (27). In this work, the enzymatic cross-linking of a novel polypeptide of the HELP series allowed the generation of hydrogels at different monomer densities, capable of supporting myoblast adhesion, proliferation and, to a far lesser extent, differentiation.
The shear modulus of 5% and 7% HELPc hydrogels differed by an order of magnitude, indicating that even a moderate diversity in monomer concentration produced matrices with considerable differences in mechanical properties. Together with the qualitative observation of an evident discrepancy in hydrogels compactness (Suppl Fig. 1B), these data support the view that HELPc hydrogels provide myoblastic cells with substrates greatly different in their mechanical properties. Accordingly, the differences observed in cell morphology, vinculin clustering and cytoskeletal organization are likely related to the different compliance of the adhesion substrates. Actually, a more organized cytoskeleton and more stable adhesions characterize cells adhering to stiffer or rigid substrates, despite differences in ligand density and long-time elasticity (3, 28–29–30). Accordingly, the differences we observed in cell morphology, cytoskeleton organization and adhesion properties would likely depend on the different rigidity of the supporting material. On the whole, these results are in good agreement with previous studies testing C2C12 cell adhesion on soft and stiff substrates obtained from polyelectrolyte multilayers (31–32–33).
The evidence that adhesion to HELPc hydrogels stimulates cell proliferation, particularly in low-serum medium, is intriguing and potentially useful for tissue engineering strategies. Indeed, a major problem in muscle cell therapy is the need for a sufficient number of progenitor cells for successful transplantation; as such, in vitro expansion of muscle stem cells represents one of the strategies actively being pursued (34, 35). If the proliferative effect of HELPc hydrogels were observed on muscle precursor cells, it would reveal potentially promising applications for the future, particularly for an in vivo approach to muscle tissue engineering. This strategy requires that expansion is directly followed by transplantation, avoiding myogenic differentiation in vitro (36). The approach was successfully applied to muscle stem cells expanded on soft polyethylene glycol-based substrates: it increased cell viability and prevented their differentiation in vitro, but maintained their in vivo regenerative properties when engrafted in immunodeficient mice (37).
Myotube formation is partially inhibited in cells growing on HELPc hydrogels. Although the expression of MHC indicated that myogenesis started in either condition, the low values for fusion index and the reduced size of myotubes revealed that the process could not proceed beyond a certain stage (38) and that myotube development was abnormal. Intriguingly, the extent of the inhibition correlated with the substrate compliance – with softer 5% hydrogel being more effective in preventing cell-to-cell fusion and myotube formation. Similar results have been reported for C2C12 cells seeded on polyelectrolyte multilayers of different stiffnesses (29), suggesting that excessively compliant substrates could hamper the latest stages of myotube formation.
In conclusion, besides their potential application in tissue engineering, HELPc-based hydrogels could be employed in studies aimed at dissecting the molecular pathways responsible for the transition of proliferating myoblasts to quiescent, fusing myotubes.
Footnotes
Financial support: This work was supported in part by the European Regional Development Fund (Cross-border cooperation Program Italy-Slovenia 2007-2013, Trans2care strategic project) and by Beneficentia Stiftung (BEN 2014/125), Vaduz, Lichtenstein.
Conflict of interest: The authors declare they have no conflicts of interest.