Abstract
Background
Bioceramic granules are a widely studied material for regeneration of human tissues, and their biological assessment with in vitro cell cultures plays a fundamental role in the development of bioceramics. Design of samples for cell testing represents an important aspect of the biological evaluation, as it dictates how cells will interact with the biomaterial. The aim of this study was to develop samples for cell testing of bioceramic granules with a novel design that would enable direct physical contacts between cells and bioceramic and improved handling properties for efficient laboratory work. The goal was to produce a bilayered polycaprolactone-bioceramic composite with polycaprolactone serving as a bottom layer and support for a uniform and dense layer of bioceramic granules (upper layer), which would be only partly embedded and physically stabilized in the polymer with at least one face of granules still free of any polymer residues and available for direct attachment of cells.
Methods
A novel method for preparation of samples in six steps was developed. A bilayered design of samples with exposed bioceramic particles was accomplished by the application of a water-soluble alginate as a sacrificial polymer in the method protocol. Samples were analyzed with SEM/EDX and ToF-SIMS.
Results
Bioceramic granules had a uniform and dense morphology and were partly embedded in the polycaprolactone support. Detailed ToF-SIMS study showed that granules were clean and free of any polymer residues.
Conclusions
The developed samples enable direct exposure of bioceramic granules to cells and surrounding physiological solution during cell testing, and possess improved handling characteristics.
Introduction
Ceramic materials have been investigated for decades for biomedical applications, with reports dating back to the end of the 19th century, when the first report on the use of plaster of Paris (CaSO4 · H2O) to repair bone defects was published (1). The widespread interest in the clinical use of bioceramics, mainly for bone regeneration, began in the late 1960s with the work of different research groups (1–2–3–4), and since then the area has seen strong development, and numerous bioceramic products in different forms (granules, scaffolds, putties, coatings etc.) have been brought to clinics to aid regeneration of human tissues (2, 4). Ever since the early days of bioceramics research, material development and biological testing have worked in close association to develop high-performance implants which would meet biocompatibility requirements. While in the past mainly in vivo tests were used to assess the biological response to biomaterials, there has been an increasing demand to reduce the numbers of animals used in scientific experiments, following the “alternatives” concept or “3R concept,” which stands for
With regard to cell testing of bioceramics, the design of samples for cell testing has an important role and may have a critical effect on the cell response to a material. However, so far this area has been partly neglected, and no standard protocols for sample preparation have been proposed. As samples for cell testing represent the final biomaterial product which is going to be implanted in the human body, they should be designed to resemble as close as possible the biomaterial's properties, taking into account its physicochemical characteristics (composition, chemical and mechanical properties, surface roughness and modification) as well as the form in which the biomaterial is to be used in clinics. Bioceramic products used in clinics are used in different forms, such as granules, porous scaffolds, blocks, putties, coatings, cements etc. A widely used form of bioceramics (in which Lucideon Ltd. also has a strong interest) are granules of various size ranges, which are used alone (10–11–12–13–14) or applied in combination with a biopolymer to make a composite membrane (10, 15–16–17–18–19) or a malleable putty (13, 14, 20–21–22). In previous cell testing studies on bioceramic granules, cell tests were performed by culturing cells on (i) tissue culture plastic, together with a certain amount of bioceramic granules added in the wells of the cell plate (11, 23–24–25–26); (ii) by growing cells on bioceramic-polymer composites (membranes and scaffolds) (17–18–19, 27–28–29–30–31–32–33–34–35) and (iii) by incubating cells on tissue culture plastic in medium containing bioceramic extracts (36–37–38–39–40–41–42–43). Forms of bioceramic discs and sintered scaffolds are not considered here as they do not include granules. The fundamental drawback of these approaches is that during cell culture experiments there is a lack of direct contacts between cells and bioceramic granules, and the cells mainly grow on tissue culture plastic (i and iii) or on a polymer (ii), which can lead to misleading results.
The aim of this study was to develop samples for cell testing with a novel design, which would allow bioceramic granules to be exposed to the surrounding system and available to cells to establish direct contacts on the surface of bioceramics during cell testing and also enable improved handling characteristics for efficient laboratory work. Phosphate-based bioglass in the system P2O5–CaO–Na2O–MgO–SrO was used as a bioceramic material. Moreover, the goal was to combine polycaprolactone (PCL) and bioglass granules to design bilayered samples where PCL would serve as a support for a uniform and dense layer of bioglass granules. Furthermore, the goal was to employ electron microscopy and time-of-flight secondary ion mass spectrometry (ToF-SIMS) to characterize the samples at various length scales.
MATERIALS and methods
Bioglass synthesis
Phosphate-based bioglass in the system P2O5–CaO–Na2O–MgO–SrO in the form of granules in the size range 90-250 µm was used as representative bioceramic material in the current study. Bioglass was prepared in-house via fusion.
Method for preparation of bioglass-PCL composite membrane
The aim was to prepare bilayered bioglass-PCL membrane where PCL would form the bottom part and serve as a support for a uniform layer of bioglass granules, which would be partly embedded in PCL but would still have at least one face free of a polymer and exposed to cells during testing (no. 6 in Fig. 1). Moreover, the aim was to prepare large composite bioglass-PCL membrane from which smaller specimens would be cut out for cell testing in order to fit specific cell plate sizes. Special attention was given to the preparation of membranes, with bioglass particles being exposed on the surface of the membrane and not being covered by a polymer. To achieve this, a novel method employing a sacrificial polymer was developed as described in Figure 1. First, a glass petri dish was coated with an alginate layer by casting 2 wt.% alginate solution (Sigma Aldrich, USA) in a dish and leaving it to dry overnight (no. 1 in Fig. 1). In the next step, an area around the alginate layer was masked, and a uniform and dense layer of bioglass particles (90-250 µm) was scattered on the unmasked part of the alginate (no. 2 in Fig. 1). Masking enabled the bioglass granules to be deposited only on a selected part of the alginate, thus allowing reproducible deposition of bioglass granules. The mass of the bioglass layer was 1.20 ± 0.05 g/36 cm2. A uniform and reproducible bioglass layer on alginate was successfully obtained by following the overall mass and distribution of deposited bioglass using a microbalance (XPE105 analytical balance; Mettler Toledo, Switzerland) and optical microscope (Leica Microsystems GmbH, Germany), respectively. In the next step, the bioglass-covered alginate layer was dampened with a gentle spray of deionized water, and afterwards samples were immediately dried with a hair-dryer (no. 3 in Fig. 1). The aim of this step was to soften the surface layer of alginate so it became sticky and to enable the bioglass granules to partly sink down in the alginate layer and allow them to be partly entrapped and stabilized in the alginate so that when turning around the petri dish, glass particles did not fall out from the dish. Afterwards, PCL/chloroform solution (10 wt%; molecular weight = 80,000; Sigma Aldrich, USA) was cast in the petri dish on top of the dried alginate/bioglass layer (no. 4 in Fig. 1). The dish was covered with a lid and left for 72 hours under a fume hood to allow for solvent evaporation. Using this procedure, a membrane with a “sandwich structure” was obtained, with glass particles partly entrapped both in alginate and PCL. The next step involved washing with distilled water (no. 5 in Fig. 1) to remove the alginate layer and obtain the final bioglass-PCL composite membrane with exposed bioglass on the surface (no. 6 in Fig. 1). For scanning electron microscopy (SEM) and ToF-SIMS analysis, circular samples with a diameter of 1 cm were cut out from the membrane using a punch.
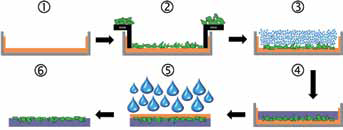
Schematic presentation of the method for preparation of bioglass–polycaprolactone (PCL) composite membranes with exposed bioglass granules on the surface. No. 1: Deposition of alginate (brown) layer in the petri dish (gray); No. 2: deposition of bioglass (green) layer on the alginate layer; No. 3: gentle dampening of the alginate to allow integration of bioglass into the alginate layer; No. 4: casting of PCL (violet) on top of the dry bioglass-alginate layer leading to a “sandwich-like” structure of the alginate-bioglass-PCL composite; No. 5: removal of alginate from the sandwich by washing with water; No. 6: final bioglass-PCL composite membrane.
Surface analysis of bioglass-PCL composite membranes
As explained in the above section, the aim was to prepare composite membranes with bioglass particles exposed on the surface. To check whether the surface of bioglass granules was free of any polymer residues, the prepared composite membranes were examined with SEM and with static ToF-SIMS imaging.
SEM/EDX analysis
Samples were characterized with SEM (JEOL JSM 6490LV, Japan) coupled with energy dispersive X-ray spectroscopy (EDX; INCA Energy Dispersive X-Ray analysis system, Oxford Instruments, UK). For SEM/EDX, samples were coated with a thin (∼10 nm) layer of gold prior to analysis.
Time-of-flight secondary ion mass spectrometry
ToF-SIMS measurements were carried out using an ToF.SIMS 5 instrument (ION-TOF GmbH, Münster, Germany) equipped with a bismuth primary ion source and a pulsed electron flood source for charge compensation. Spectra and images were acquired using either the Bunch mode (for best mass resolution) or the Burst Alignment mode (for best spatial resolution using a Bi3+ spot size of <0.3 µm). Spectra and images were taken from 3 samples of bioglass-PCL composite membranes at several spots. Bioglass powder, PCL and alginate films were used as a reference. Characteristic masses (m/z) in the positive ion spectrum for each of the reference samples are listed below:
Bioglass: 23 (sodium), 24 (magnesium), 40 (calcium) and 88 (strontium). (Intense phosphate signals were detected in negative ion mode.)
PCL: 115 (Repeat Monomer Unit M, -C6H10O2-, +H) and 97 (Repeat Monomer Unit M, C6H10O2-, -OH).
Alginate: 59 (C3H6O.H), along with a range of high mass signals with the C3H6O repeat unit.
Results
Following the method protocol described in Figure 1, a bioglass-PCL composite membrane with the aimed-for bilayered design was successfully manufactured. The membrane was malleable and could easily be handled while maintaining full mechanical integrity. This property allowed further mechanical processing of the membrane – for example, cutting out smaller specimens from the membrane for the purpose of cell testing or analytical assessment.
SEM (Fig. 2) of prepared composite membranes showed that bioglass particles had a dense and homogeneous distribution and were partly embedded in the PCL polymer underneath, with a large portion of granules still free of polymer. Moreover, EDX analyses of exposed bioglass surfaces showed no signals for carbon, indicating that the majority of embedded bioglass particles were clean with no organic residue on the upper surface. However, since EDX cannot sample the surface at a very sensitive level, it was not possible to conclude that there was no polymer layer covering the bioglass particles. The polymer layer could have a thickness of a few hundred nanometers, and EDX still would not detect any polymer on the bioglass.

Scanning electron microscopy (SEM) analysis of bioglass–polycaprolactone (PCL) composite membrane.
To investigate the idea further and to get a more conclusive answer regarding the surface properties of the bioglass-PCL membranes, ToF-SIMS was employed. Results of the ToF-SIMS analysis are shown in the Figure 3. Images represent several spots on the sample surface and show the distribution of PCL (red) on the surface of the bioglass particles (green) using a thermographic scale where the bright regions correspond to high signal intensities and dark regions correspond to low signal intensities. From evaluation of each spectrum as a whole, no evidence for alginate was obtained in any of the bioglass-PCL areas analyzed, which means that it was all removed during the water-washing step (no. 5 in Fig. 1). Analysis showed that the bioglass surface was generally clean and free of any organic surface contaminants. However, in a few spots, PCL signals were detected on the surface of bioglass granules. The reason for this could be insufficient wetting of bioglass particles by the alginate gel (no. 3 in Fig. 1), leaving dry zones and cavities into which the PCL-chloroform solution could penetrate. Where PCL was detected on the glass, the underlying bioglass was also detected, indicating that the thickness of the PCL layer in these regions was very thin, probably only a monolayer, since ToF-SIMS in the static mode only samples the outermost few nanometers of a solid surface. This material appears to have been present as low molecular oligomers, as suggested by PCL signals in the mass range of ∼400-1,500 Da.
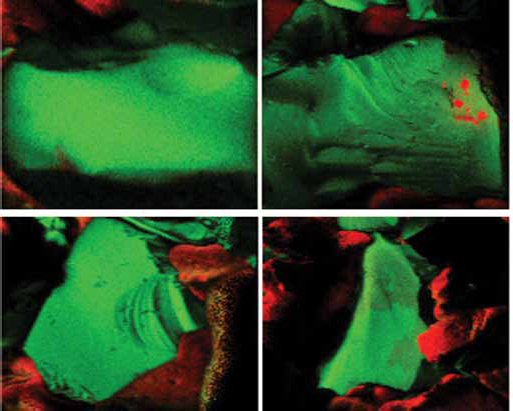
Time-of-flight secondary ion mass spectrometry (ToF-SIMS) surface analysis of bioglass–polycaprolactone (PCL) composite. Bioglass is shown in green and PCL in red.
Discussion
Cell testing of bioceramic granules has been used frequently to assess materials’ biological properties. Usually experiments are performed (i) by culturing cells on tissue culture plastic, together with a certain amount of bioceramic granules added in the wells of the cell plate (11, 23–24–25–26); (ii) by growing cells on bioceramic-polymer composite membranes (17–18–19, 27–28–29–30–31–32–33–34–35); and (iii) by incubating cells on tissue culture plastic in culture medium containing bioceramic extracts (36–37–38–39–40–41–42–43). In the case of (i), which is the most frequently applied approach, normally a specific amount of bioceramic powder is randomly scattered in the well of a cell plate and tissue culture plastic takes up the large majority of the surface area available to cells. Consequently, cells are mainly growing on tissue culture plastic, which is a stiff surface-treated substrate specially developed to support cell growth. The final cell results will therefore be the result of the contribution of several factors, such as the tissue culture plastic, the bioceramic surface and leached species (ions) from the bioceramics. Moreover, loose bioceramic granules may easily be moved around when handling and changing the cell medium, which represents a shock to cells that may significantly affect the cell response. Furthermore, the inability to reproduce the way bioceramic powder is scattered around the well may also lead to additional deviations. In the case of (ii), composite membranes are usually prepared by the solvent-leaching method, which includes casting of a uniform suspension of bioceramic granules and a polymer solution and then allowing the polymer solvent to evaporate, leaving behind the composite membranes with bioceramic granules homogenously distributed across the whole polymer matrix (15–16–17–18–19, 29–30–31). Due to the wetting between ceramic granules and polymer solution, even the bioceramic granules which stay on the surface of the membrane (these represent a small portion of the total bioceramic mass) will be covered by the polymer in the final composite membranes. For example, in our experiments, ToF-SIMS of bioglass-PCL composite samples revealed that even though special attention had been given to protecting the granules and keeping their surfaces free of polymer residues, a thin layer covered the bioglass granules in a few spots. Therefore, it can be expected that when membranes are prepared by casting bioceramic-polymer suspensions, polymer will fully surround the bioceramic granules and will also be much thicker. As a consequence of polymer covering the ceramic granules, cells will establish direct physical contact only with a polymer instead of with the desired bioceramics during cell culturing. In the case of (iii), cells grow only on tissue culture plastic with no possibility for them to establish any physical interactions with bioceramics, and the bioceramic extracts in the culture medium represent the only trace of bioceramics in the experiment.
An unavoidable consequence of the culturing methods (i, ii, iii) reviewed above is that during the whole period of the cell culture experiment, cells mainly grow on tissue culture plastic (i and iii) or on a polymer (ii), and there is a lack of direct contact between cells and bioceramics, which can result in misleading results. Including direct cell–material interactions in the biological assessment becomes especially important when the desired property of the material is a stimulation of cell activity on its surface (proliferation, differentiation) and guiding of tissue regeneration, such as in the case of osteoconductive and osteoinductive bone implants. In such cases, including direct cell–material interactions in the biological assessment of the material provides more conclusive data about the regeneration capacity of the biomaterial.
The method developed in the current work allows preparation of samples with a design that enables cells to adhere directly onto the surface of bioceramics during cell testing. To the best of our knowledge, this is the first report on the preparation of such samples with exposed bioceramic granules. Further benefits of prepared samples are that granules stay in place and do not move when handling cell plates or changing the culture medium during the cell testing experiments. Moreover, the versatility of the method makes it possible to use bioceramics with very different properties. For example, due to the short period of wetting the bioglass-alginate layer in step 3 of the method protocol (no. 3 in Fig. 1), it is possible to employ even highly water-soluble materials, such as glasses in the ternary P2O5–CaO–Na2O system. PCL was chosen as a “support” polymer due to its relative stability, inertness and biocompatibility (44, 45). However, any other polymer with relatively low solubility in water could be used for preparation of samples. Another advantage of the method developed is that it is carried out at room temperature, making it compatible with temperature-sensitive biological molecules for incorporation in the samples. Finally, applications of the method go beyond the preparation of samples for cell testing, on which this paper is focused. For example, the bilayered structure of the prepared membranes represents a functionally graded material with bioactive and bioinert “faces” which may be a desirable property for various applications. For example, this property combined with flexibility of membranes makes such membranes a good candidate for the manufacture of barrier membranes for guided bone regeneration, where the bioactive face would point toward the dental implant and bony side, while the bioinert part would be toward the soft tissue.
Conclusion
In this work, a new method for preparation of samples for cell testing of bioceramics in a granular form is presented. The design of the samples enables direct exposure of bioceramic granules to cells and surrounding physiological solution during cell testing, thus allowing cells to adhere directly onto the surface of bioceramics. SEM and ToF-SIMS analyses of the sample surface confirmed that bioglass granules were free of any polymer residues and had a uniform distribution. Another benefit of the method is that it is robust and versatile and can be adapted to different sample sizes and shapes and to different materials for cell testing.
Footnotes
Financial support: The authors would like to acknowledge European Commission funding under the 7th Framework Programme (Marie Curie Initial Training Networks; grant number: 289958, Bioceramics for Bone Repair).
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
