Abstract
The insufficient osteogenesis of magnesium phosphate cements (MPCs) limits its further application. It is significant to develop a bioactive MPC with osteogenic properties. In this work, MPCs were reinforced by zinc oxide nanoparticles (ZnO-NPs). The composition, microstructure, setting time, compressive strength and degradation of ZnO-NPs/MPCs (ZNMPCs) were evaluated. The results showed that the setting times of MPCs were prolonged from 8.2 to 25.3 min (5.0ZNMPC). The exothermic temperatures were reduced from 45.8 ± 0.4℃ (MPCs) to 39.3 ± 0.5℃ (1.0ZNMPC). The compressive strength of ZNMPC composite cement with 1 wt. % ZnO-NPs (1.0ZNMPC) was the highest (42.9 MPa) among all the composite cements. Furthermore, the ZNMPCs were cultured with mouse bone marrow mesenchymal stem cells (mBMSCs). The results yielded that the ZNMPCs exhibited good cytocompatibility with enhanced differentiation, proliferation, and mineralization on mBMSCs, and it also pronouncedly elevated the expressions of genes and proteins involving osteogenesis. These findings suggested that ZNMPCs could drive the differentiation toward osteogenesis and mineralization of mBMSCs, providing a simple way to the MPC with enhanced osteogenesis for further orthopedic applications.
Introduction
On the condition of aging of population, incremental patients year by year need more bone grafts or bone graft substitutes for treatment of injury- or disease-elicited bone defects. The development of bone implant materials for clinical application has become one of the most important research fields. Seeking more ideal bone graft materials for bone defect repair, bone cement materials have been gained extensive attention during the past decades due to the unique properties of these materials, such as arbitrary shaping and self-setting.1–3 Clinically, the widespread investigative studies pertaining to the application of calcium phosphate cements (CPCs) or bone void fillers in bone defect treatment have been conducted. They are ideal substitutes benefiting from their capability of resembling the inorganic constituents of bone matrix after mineralization and simple operation with injectability. However, the unsatisfactory strength, acidic solidification or slow setting and biodegradation inevitably hinder their wider medical application. 1 In view of this, incremental implants with bioactivity, proper biodegradation and competent mechanical strength appear to be desirable for bone grafts, which is the emphasis of current research.
A look at recent interest in bone regeneration unveils that magnesium phosphate cements (MPCs) are attractive on account of its rapid setting, favorable initial strength, and medium-speed degradation relative to CPCs.4,5 Traditional MPCs are made of magnesium oxide (MgO) and monoammonium phosphate (NH4H2PO4) or diammonium phosphate ((NH4)2HPO4), with magnesium ammonium phosphate hexahydrate (NH4MgPO4.6H2O, known as struvite) as the primary reaction product. 6 Cytotoxicity probably brought about by ammonia release by the traditional MPCs in the setting reaction process in the physiological environment is an open question. Therefore, a work is focused on the replacements of acid ammonium phosphates. In addition, monopotassium phosphate (KH2PO4) serves as a substitute of NH4H2PO4 in civil engineering. 7 Compared to NH4H2PO4, KH2PO4 not only has lesser dissociation constant and lower solubility, resulted in controlling the reaction rate with lower exothermic temperature, but also does not produce unpleasant odor. 8 As a result, potassium MPC come into the spotlight to be a more proper biomedical material thanks to KH2PO4 as an excellent phosphorus source. Although potassium MPC has many merits, the insufficient osteogenesis of magnesium phosphate cements makes it difficult to use in practice. Wang et al. demonstrated that the citric acid modified MPC was featured of satisfactory setting time, good cytocompatibility and osteoinductivity, indicating its potential application for bone regeneration. 9 Kim et al. incorporated a novel indene compound, KR-34893, into magnesium phosphate (MgP) ceramic scaffold to enhance the osteoblast differentiation in vitro and bone regeneration in vivo. 10 Shi et al. modified MPC by incorporating different amounts of bioactive chondroitin sulfate (CS), and the novel CS-MPCs have improved physiochemical properties that enhance compatibility in vitro and bone regeneration in vivo, making them attractive materials for bone regeneration. 11 Hence, adding active ingredients to MPC to improve its osteogenesis is a research hotspot.
Zinc, a micronutrient pivotal for bone metabolism, interferes with enzyme activity and possesses abilities to promote bone formation via stimulating osteoblast (OB) differentiation and enhancing ECM mineralization.12,13 According to report, zinc enables bone regeneration via gearing up synthesis of collagens and deposition of minerals under the mechanism adopted by it to increment alkaline phosphatase (ALP), collagen type I (Coll-I), osteocalcin (OCN) and osteopontin (OPN) to impel osteogenic differentiation.14,15 Furthermore, zinc has also been reported to have an effect not only in the resorption activity of osteoclasts but also in the formation of osteoclasts from macrophages.16,17 Accordingly, interfusing zinc with bone grafts is constructive for bone regeneration.
As known, on account of the nanoscale size impact, nanomaterials are prone to interactions with biomacromolecules. 18 Undoubtedly, control over cell behavior is a competence of nanomaterials, which fosters an environment guiding cell adhesion, localization, movement and condensation, and control over cell signaling pathways decisive for protein synthesis.19,20 Due to this key reason, nanoparticles (NPs) are widely used in the biomedical field. Among them, ZnO-NPs come into the spotlight thanks to their discrepant functions in the biomedical industry. 21 Although ZnO-based composites for bone tissue regeneration are scarcely reported, these reports confirmed that the introduction of ZnO enhanced cell compatibility, proliferation, mineralization and osteogenic differentiation.22–27 Therefore, we hypothesized that ZnO-NPs doped MPCs with the modified physicochemical properties may improve the ability to promote osteogenesis, which are beneficial for bone regeneration.
In this study, ZnO-NPs, as the zinc resource for bone regeneration, were introduced into MPC via simple physical blending with different contents. The microstructure and phase composition of hybrid cements were characterized via X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FT-IR) and scanning electron microscope (SEM). This paper was designed to delve into the functions of ZNMPCs in osteogenesis in vitro and conduct a mechanistic dissection for ZNMPCs in bone grafts.
Materials and methods
ZNMPCs preparation
ZNMPCs were prepared by KH2PO4 (purity: 99.5%, Jiudian Pharmaceutical Co., Ltd., Hunan, China) and MgO (purity: 98.5%, Xingtai Metallurgical Magnesium Industry Co., Ltd., Hebei, China) undergoing 5-h calcination at 1600°C at a mass ratio of 1: 0.9. Before experiment, 1-h grinding of the calcinated MgO and KH2PO4 was completed in a planetary ball mill (PM400, German Retsch) at 300 r/min, and the obtained substance was sieved through a 300-mesh sieve. The ZnO-NPs (AR, 30–80 nm, purity 99.0%, 21.50 m2/g, Jiangsu Xianfeng Nanomaterial Technology Co., Ltd., Jiangsu, China) was used without any treatment.
The MgO, KH2PO4 and ZnO-NPs were premixed and then added with the distilled water at a liquid-to-powder ratio of 0.4 (mL/g). The paste was then put into a cylindric mold to fabricate cylinders (diameter: 6 mm and height: 12 mm) for physical and chemical experiments, and it was placed into a circular mold (diameter: 6 mm and thickness: 3 mm) to fabricate samples for cell-based tests. According to the ZnO-NPs content (0, 0.5, 1.0, 3.0, and 5.0 wt. %), the composite bone cement was named MPC, 0.5ZNMPC, 1.0ZNMPC, 3.0ZNMPC, and 5.0ZNMPC, respectively.
Characterization
The structure and composition of ZNMPCs samples were examined by FT-IR (Nicolet 6700, Thermo Fisher Scientific, USA) with resolution of 4 cm−1 from 400 to 4000 cm−1 using KBr disks, XRD (D8 Advance, Bruker, Germany) at a scanning rate of 2°/min from 5° to 80° and field emission SEM (FEI Nova Nano 450, USA).
The setting time of the cement pastes was determined according to ISO 9597-2008 with Vicat apparatus (ZK-ISO, Beijing Hengqi Instrument Co., Ltd.). The setting temperature was measured by thermometers. Each assay was implemented thrice in each group (n = 3), and mean ± standard deviation (M ± SD) was adopted to reflect the results.
Measurements of the compressive strength of ZNMPCs (6 × 12 mm) were taken at indoor temperature on the MTS-858 Mini Bionix testing system (MTS Systems, USA) at a constant rate of 1 mm/min. The experiment was carried out six times in each group (n = 6), and the results were reflected by M ± SD.
The degradation was gauged. In outline, the ZNMPC specimens underwent 3-day solidification at room atmosphere, and M0 symbolized the precisely gauged initial weight. Later, the specimens were immersed in tri(hydroxymethyl) aminomethane hydrochloride buffer solution (Tri-HCl, 30 mL/g, pH = 7.40) for 28 days and were placed in an air bath shaker (THZ-92A, Shanghai Boxun Medical Biological Instrument Co., Ltd.) at 70 r/min and 37°C. Subsequent to discrepant lengths of soaking, the specimens were moved out for drying in a DZF-6050 Vacuum Drying Oven (Shanghai Yiheng Scientific Instrument Co., Ltd.) at 80°C until constant weight. Equation (1) was introduced for calculation of the weight loss rate of each specimen. Meanwhile, the suspensions were collected and the pH values were read by use of the PHS-3 C pH Meter (Shanghai INESA Scientific Instrument Co. Ltd., China) at each evaluation. Here, M0 refers the initial weight, Mi refers the weights of the dried samples after soaking at different periods.
Additionally, the ions release was done as the followings. The specimens of 1.0ZNMPC were immersed in Tri-HCl solution (30 mL/g, 70 r/min, 37°C) in an air bath shaker for 1, 3, 5, and 7 days, and the liquid was refreshed at each time point. At 1, 3, 5, and 7 days, the concentrations of Zn, Mg, P, K ions were measured by inductively coupled plasma atomic emission spectroscopy (ICP-AES, PerkinElmer, MA, USA). The experiment was carried out three times in each group, and the results were reflected by M ± SD.
In vitro assays
Cell culture
Ultraviolet sterilization was done for 30 min ahead of schedule. The Kunming mouse bone marrow mesenchymal stem cells (mBMSCs) were objects for measurement of cell viability and morphology and OB differentiation. DMEM comprising 10% fetal bovine serum (FBS, Chengdu Hali Biological Technology Co., Ltd., China) and mBMSC osteogenic induction fluid (Cyagen Biotechnology Co., Ltd, Guangzhou, China) were employed for cell culture under an atmosphere with 100% humidity and 5% CO2 at 37°C, with medium refreshing 3 days/time.
Cell proliferation and viability
Subsequent to 24-h immersion in DMEM (Gibco, Thermo Fisher Scientific) at a weight/volume percentage concentration of 200 mg/mL at 37°C, centrifugation of aseptic ZNMPCs was fulfilled to harvest the supernatant. Next, the leaching liquid of samples were diluted to different concentrations with 10% FBS and 1% penicillin/streptomycin supplement. mBMSCs were seeded in the different extracts at 1 × 103 cells per 100 μL medium and placed in a CO2 incubator for incubation at 37°C. DMEM was utilized as the control. Subsequent to 1-, 4-, and 7-day cultivation, the cells were added with 10 μL of CCK-8 (CK04, Dongren Chemical Technology Co., Ltd., China) solution for 1-h incubation in the CO2 incubator. Measurements of the optical density (OD) at 450 nm of the culture medium were taken by use of a microplate reader (Multiskan GO, Thermo Scientific, USA). Calculation of the relative cell viability was completed according to equation (2).
In keeping with the protocol of the manufacturer, cell viability was further assessed by the Calcein-AM/PI Double Stain Kit provided by Shanghai Xiangsheng Chemical Co., Ltd. (China). Subsequent to seeding on 12-well plates at 2 × 104 cells/well, the cells received 5-days culture and both living and dead cells underwent dyeing by Calcein acetoxymethyl ester (AM) and propidium iodide (PI). Ultimately, a fluorescence microscope was introduced for picturing living cells (green) under 490 nm excitation wavelength and dead cells (red) under 545 nm excitation wavelength.
Cell morphology
Cell morphology examination was implemented. In brief, cell inoculation into 12-well plates at 2 × 104 cells/well was implemented after 3-days incubation. Next, cells were dyed via rhodamine-phalloidin and DAPI (Beyotime Biotechnology Co., Ltd., China) staining according to the guideline. Finally, subsequent to the addition of an anti-fluorescence quenching mounting agent, image observation and collection were implemented under a confocal laser scanning microscope.
ALP activity
Measurements of the ALP activity of mBMSCs were obtained by use of ALP assay kit (Nanjing Jiancheng Bioengineering Institute, China) at 4, 7, and 14 days following the manufacturer’s guidelines. 100 mL of serum reacting with matrix at 37℃ for 15 min to produce 1 mg phenol is defined per Gold Unit (GU). The ALP activity was calculated according to equation (3) and the optical density (OD) at 520 nm of each well was determined by a microplate analyzer (Multiskan MK3, Thermo Fisher Technology Co., Ltd.). Meanwhile, the ALP staining was observed under Nikon Eclipse E100 microscope (Nikon Instruments (Shanghai) Co., Ltd., China) after culturing for 4 days.
Alizarin red S (ARS) staining
Cells cultured with samples were evaluated with ARS (Wuhan Service Bio-Biotechnology Co., Ltd, China) with osteogenic medium after 21 days. Observation of calcium nodules receiving ECM mineralization was achieved under an optical microscope (Nikon Eclipse E100, Nikon Instruments Co., Ltd., Shanghai, China).
Quantitative real-time polymerase chain reaction (QRT-PCR)
In brief, mBMSCs were inoculated on 12-well plates at 2 × 104 cells/well and underwent 4-, 7-, and 14-day culture with ZNMPC samples, respectively. Afterward, OPN, ALP, Coll-I, OCN, and Runx-2 expressions were quantified. Subsequent to extraction, RNAs were measured. Primer sequences applied were displayed in Table 1.
Primer sequences.

Statistical analysis
T-test was introduced for statistical assessment of data, and M ± SD presented the results. A difference was deemed to be statistically significant in the case of a p-value below 0.05.
Results
Characterization
The FT-IR spectra of ZNMPCs were shown in Figure 1. The spectra and assignments for the pure MPC were identical to those reported by Banks et al. 28 The strong and sharp absorption band of 1015 cm−1 and 563 cm−1 were initiated by the PO43− in MPC. With the introduction of ZnO into MPCs, the FT-IR spectra of ZNMPCs were changed. The typical bands of PO43− at 658 and 578 cm−1 appeared in ZNMPCs. Meanwhile, ZNMPCs showed broader ν3 absorption bands and the peak of 1200–900 cm−1 was around 1075 cm−1. In addition, water bending (1626 cm−1) and water stretching (3742 cm−1 and a broad band at 3700–2750 cm−1) vibrations were detectable.
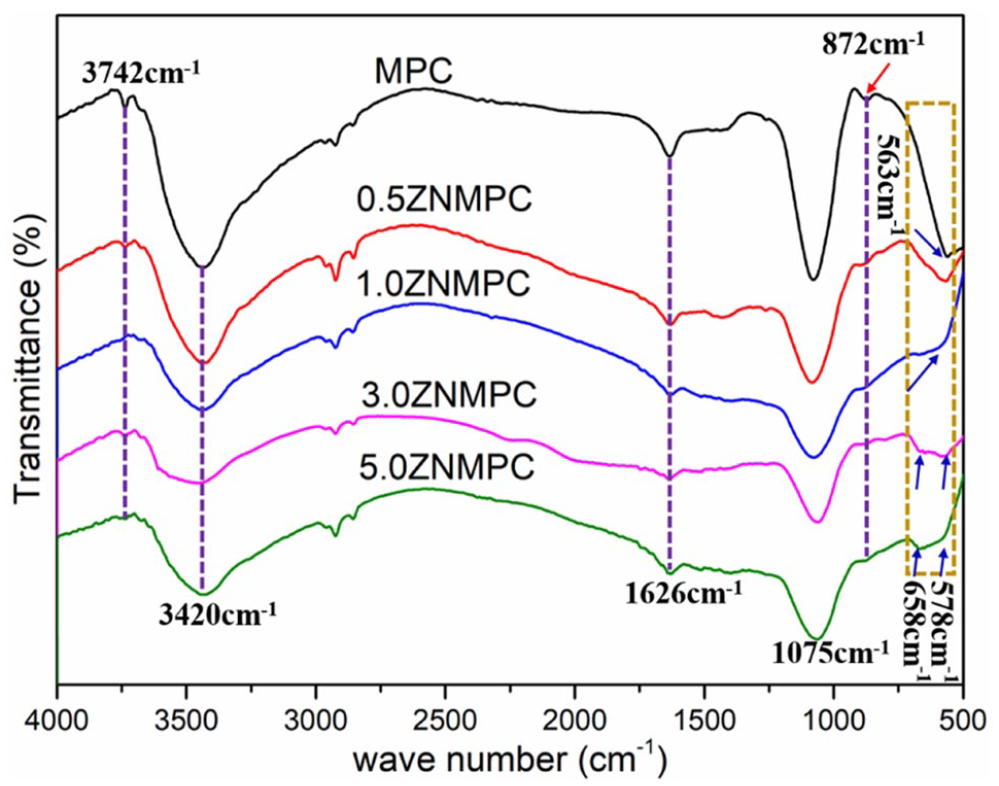
The FT-IR spectra of ZNMPCs.
In the XRD patterns for composite bone cement, except for the characteristic peaks of MgKPO4·6H2O (PDF#75-1076), the peaks appeared at 43.2° and 62.5° on the strength of the existence of MgO in excess (PDF#45-0946). As a reactant, KH2PO4 was not detectable as it dissolved rapidly. 29 As shown in Figure 2, the peaks obviously presented at 36.1° was attributed to the existence of ZnO in ZNMPCs. The diffraction peak intensity at 62.5° became stronger with the increasing content of ZnO-NPs. It may be due to the overlapped peak of ZnO-NPs at this position.

The XRD patterns of ZNMPCs.
The original mixed powders before adding water were shown in Figure 3(a). KH2PO4 was about 10 µm with smooth surface. But MgO was dispersed with a wide size distribution and smaller than 5 µm. The mixed powders were homogeneous by physical blending. These are important for the quality of the solidified products.

The original mixed powders before adding water (a) a1, a2: KH2PO4; b1, b2: MgO; c1, c2: physical blending of KH2PO4 and MgO; d1, d2: physical blending of KH2PO4, MgO and ZnO-NPs; (b) SEM graphs of ZnO-NPs and the cross-section of ZNMPCs (b).
In SEM micrographs (Figure 3(b)), ZnO was a regular spherical NP with about 80 nm. To MPCs, the main product was struvite (MgKPO4·6H2O), which was a closely spaced flat tabular crystal with wedge-shaped edges. 30 The findings yielded that the MPCs possessed a dense structure on account of the existence of MgKPO4 crystals and clay-like matters, and the two exhibits a close proximity. At the same time, partial unreacted ZnO-NPs were seen. The interior microstructure of the composite cements was changed as the content of ZnO-NPs rose. With the elevation of ZnO-NPs content, the fracture surface first became compact and then looser. When the content of ZnO-NPs in ZNMPCs was up to 3 wt. %, cracks were seen in the composite cements. This may be because that agglomeration caused by the over-dose of ZnO-NPs destroyed the integrity of the composite cements.
Setting time, exothermic temperature, and compressive strength
Table 2 illustrated the setting time, exothermic temperature and compressive strength of MPCs and different ZNMPCs at 0.4 mL/g liquid-powder ratios. The setting time of ZNMPCs was longer than that of MPC. This was explained by that ZnO-NPs could lead the delay effect on the setting reaction. With the increasing content of ZnO-NPs, the retarding effect was more obvious. In the meantime, the exothermic temperature of ZNMPCs with 0.5 wt. % ZnO-NPs was 40.5 ± 0.3℃. This was significantly lower than that of pure MPC with 45.8 ± 0.4℃. But there was no significant difference in the change of exothermic temperature for different ZNMPCs with incremental content of ZnO-NPs. The compressive strength of 1.0ZNMPC topped among ZNMPCs in compressive strength (42.9 ± 1.5 MPa). This value of 1.0ZNMPC was about 230% times the compressive strength of pure MPC. When the content of ZnO-NPs was 3 wt. % in ZNMPC composite, the compressive strength was then decreased to 28.7 ± 1.6 MPa. These results were in agreement with microstructure analysis.
Setting time, exothermic temperature and compressive strength for different cements, Significant difference compared with MPC.

p < 0.01.
Degradation
As shown in Figure 4(a), the weight loss rates of ZNMPCs were inspected in Tri-HCl buffer solution for various time periods. As the degradation cycle extended, the weight loss rates of ZNMPCs exhibited a step-wise uptrend, and those of MPC, 0.5ZNMPC, 1.0ZNMPC, 3.0ZNMPC, and 5.0ZNMPC composites were 39.0, 43.8, 44.7, 46.1, and 47.5 wt.% after soaking for 28 days, respectively. It was observed that introduction of ZnO-NPs into MPC cement leaded to slight increase of weight loss rate.

(a) Weight loss rate of the ZNMPCs, (b) pH variation of the medium after soaking in Tris-HCl for 28 days, (c) SEM graphs of ZNMPCs before and after degradation for 7 days, and (d) the ion release of Zn, Mg, P, K for 7 days.
Figure 4(b) displayed the pH values of ZNMPCs degradation solution. The pH values were increased from initial 7.40 to 7.62 during the first day, and then dropped down to initial the pH value. After 7-day degradation, the pH values of degradation solution rose again. With the increase of ZnO-NPs, the pH values of degradation solution showed a slow gradual decline. On the whole, the pH values of ZNMPCs fluctuated around 7.50, which was not able to hurt the growth of cells and tissues.
The SEM graphs of ZNMPCs before and after degradation for 7 days were shown in Figure 4(c). The surfaces of ZNMPCs were also changed as the content of ZnO-NPs rose. With the increase of ZnO-NPs content, the surface first became compact and then looser. When the content of ZnO-NPs in ZNMPCs was more than 3 wt. %, more cracks and pores were seen in the composite cements. This result was consistent with the cross-section of ZNMPCs. After the samples were immersed in Tri-HCl for 7 days, the surfaces were changed dramatically. After degradation for 7 days, their surfaces became coarser. More porosities appeared due to the high degradation.
The variation of the Zn, Mg, P, K ions concentrations after the sample (1.0ZNMPC) soaked for 1, 3, 5, and 7 days were depicted in Figure 4(d). The release of Zn2+ in solution was lowest for 1.0ZNMPC and almost constant over the whole period of dissolution. In contrast, the release of Mg2+, PO43−, and K+ were very high, this was because MgO and MgKPO4·6H2O were the important main ingredients of the composite cement. The zinc exhibited a sustained release behaviors during 7 days in Tri-HCl solution.
In vitro assays
Cell proliferation and viability
The proliferation of mBMSCs of different concentrations of leaching liquid were illustrated in Figure 5(a). It was observable that the cell proliferation rate in all ZNMPC specimens at the same concentration firstly rose, and then descended as the content of ZnO-NPs rose at each test point in time. When the concentration of ZNMPC leaching liquid reached 50 mg/mL, the cell proliferation was lower than that in blank control groups. The high concentrations of leach liquor were toxic to cells. As unveiled in Figure 5(a), mBMSCs presented with sound viability with the content of ZnO-NPs at 0.5 and 1.0 wt. % in the case of the specimen concentration lower than 25 mg/mL. The cell viability in the ZNMPCs plummeted relative to that in control group with the content of ZnO-NPs above 3 wt. %. Comparatively speaking, 1.0ZNMPC exhibited the highest cell proliferation rate. By comprehensive consideration, 1.0ZNMPC sample was selected to carry an in-depth investigation.

The relative proliferation rate (a) of ZNMPCs at different concentration culturing for 1, 4, 7 days, ⁕p < 0.05, ⁕⁕p < 0.01 versus MPC at each concentration; (b) Calcein-AM and PI staining of living or dead mBMSCs grown on ZNMPCs for 5 days. Scale bar 20 µm.
Furthermore, Calcein-AM/PI double staining was implemented for cell viability validation (Figure 5(b)). Calcein-AM, used to stain living cells, displayed viable cells on ZNMPC material surfaces. In contrast, the dead cells attached to the ZNFMPC material surface showed red nuclear PI staining. In comparison to blank and MPC controls, the fewer dead cells were seen on the ZNMPC material surface, suggesting that the environment could sustain mBMSCs growth and proliferation. Remarkably, the result of fewer dead cells seen on the ZNMPC material surface indicated that the introduction of ZnO-NPs was more beneficial for cell proliferation and viability.
Cell morphology
Cells co-cultured with ZNMPCs were stained with DAPI and rhodamine-phalloidin, which were flat and dilated in a spindle shape, presenting with a clear frame and striking antennas under a confocal microscope (Figure 6). Besides, the fluorescence images showed that cells spread out relatively better on 1.0ZNMPC surface as compared with those in other two groups. The results indicated that ZNMPCs could provide favorable cellular response for the cells after the introduction of ZnO-NPs.
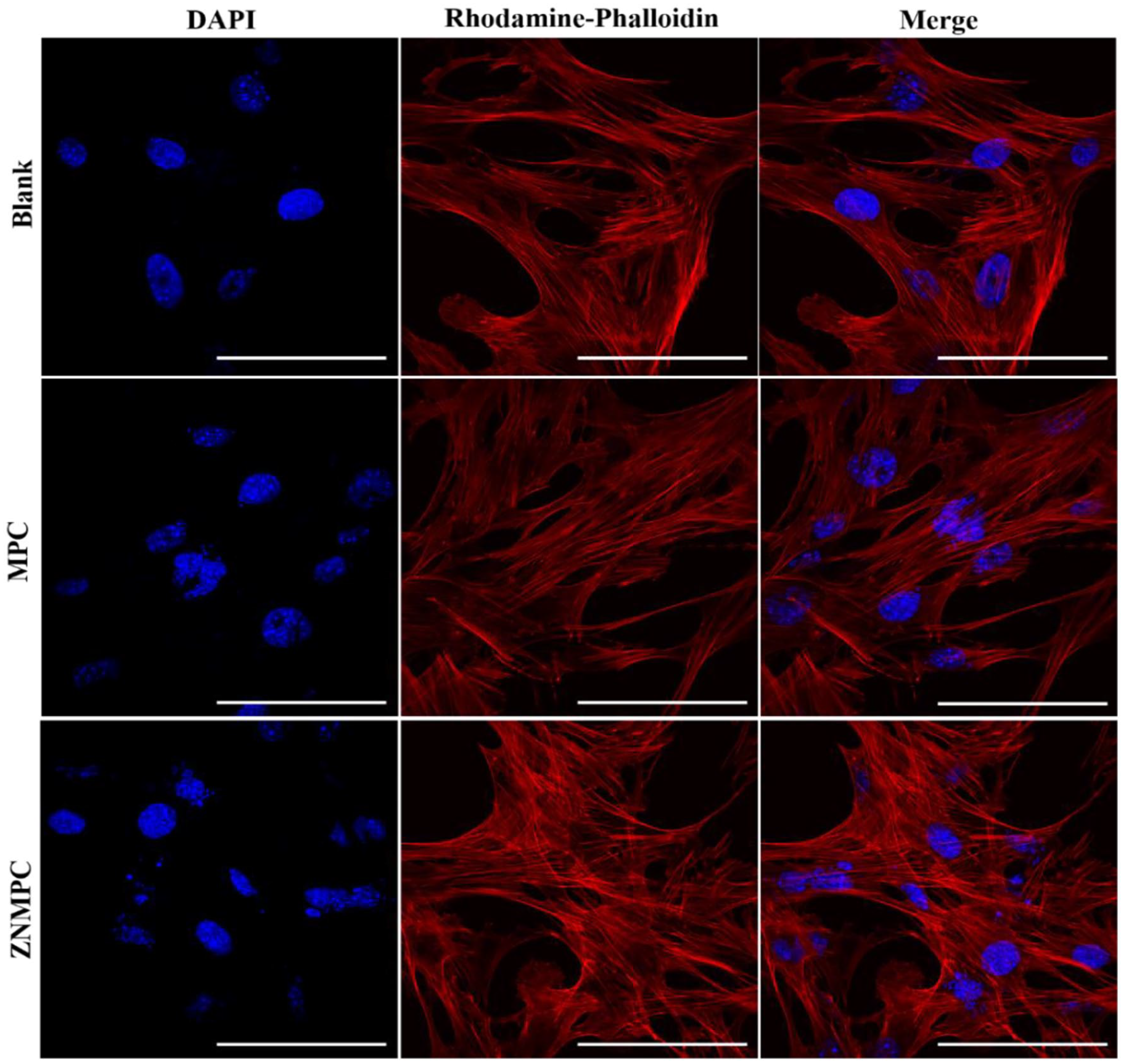
DAPI and Rhodamine-phalloidin double staining mBMSCs grown on ZNMPCs for 3 days. Scale bar 50 µm.
ALP activity
The potential OB differentiation of mBMSCs cultured with ZNMPCs for 14 days was evaluated by the ALP activities in Figure 7. The ALP activity of mBMSCs cultured with ZNMPCs rose as the culture was prolonged, and turned out to be pronouncedly higher relative to that in the blank and MPC at different culture time. Meanwhile, ALP staining was conducted at the fourth day, and more bluish-purple sediments were produced in ZNMPC group. Such an improvement of ALP as the major bone-related markers could be ascribed to the incorporation of powder ZnO-NPs into the MPC matrix, strengthening the osteogenic differentiation competence of mBMSCs.

ALP activity and staining of mBMSCs cultured with MPC and ZNMPC for 4, 7,14 days and 4 days, respectively. Scale bar 100 μm. ⁕p < 0.05, ⁕⁕p < 0.01 versus MPC at each time point.
ARS staining
ARS was introduced for assessment of the mineralization of mBMSCs receiving co-culture with ZNMPCs after 21 days (Figure 8). A larger body of red-stained calcium nodules were detectable in ZNMPC group than that in blank and MPC groups, hinting that the introduction of ZnO-NPs into MPCs drove mBMSCs differentiation. It came to the identical consequence of the ALP activity

ARS staining analysis of mineralized mBMSCs for 21 days. Scale bar 100 μm.
QRT-PCR
Moreover, ALP, Coll-1, OCN, OPN and Runx-2 expressions were detected to delve into the impacts of ZNMPC composites on the osteogenic potential of mBMSCs (Figure 9). In the 1.0ZNMPC group, higher levels of osteogenesis-related genes ALP expression of mBMSCs cells than blank and MPC groups were discovered at 7 and 14 days. In comparison with blank and MPC groups, Col-1 expression levels in the ZNMPC group were enhanced at 7 days. But there was a slight increase in Col-1 expression among the three groups at 14 days. OCN and OPN gene expression levels in the ZNMPC group were enhanced at 14 days. It was worth noting that OPN gene expression level in the ZNMPC group was significantly increased at 14 days. Interestingly, there were a slight increase in Runx-2 expression among the three groups at 7 and 14 days. After induction for 4, 7, and 14 days, the mRNA expression levels of in the ZNMPCs were higher than those in the blank and MPC group on the same day unveiling that the introduction of ZnO-NPs exhibited an osteogenic induction impact on mBMSCs.

Typical gene expressions in MPC and ZNMPC groups subsequent to 4-, 7-, and 14-day culture with mBMSCs (★★p < 0.01, ★p < 0.05 vs blank group).
Discussion
The incorporation of nanomaterials into scaffolds successfully overcame the limitations, such as adhesion deficiency and unsatisfactory mechanical strength, and augmented the competence of scaffolds in treatments. The multi-functionalized ZnO-NPs exhibit promise for widespread use in the biomedical industry, 31 especially in tissue engineering.22–27 It is easy to see that ZnO-NPs-based composites exhibit excellent cell compatibility and elicit the proliferation, differentiation and mineralization of bone-related cells or stem cells without any exception.
In our present study, it was the first time that ZNMPCs with enhanced initial compressive strength had been successfully designed and fabricated. As the content of ZnO-NPs rose, the compressive strength of ZNMPCs climbed up and then declined. The compressive strength of 1.0ZNMPC (1 wt. % ZnO-NPs) reached around 42.9 MPa (Table 2), perfectly matching that of natural cancellous bone (1–10 MPa). 32 ZNMPCs showed improvement of mechanical properties after incorporating ZnO-NPs. This is explained by that the incorporated ZnO-NPs were not only acted as stress concentration points to absorb a large number of energy and promote shear strain of MPC matrix under the effect of external stress but also served as an effective barrier to dislocation movement and efficiently impeded the crack extension.33,34 However, with ZnO-NPs further increasing, the mechanical strength declined, which was closely related with the agglomeration behavior of nanoparticles. As a result, the compressive strength of ZNMPCs plummeted as ZnO content rose to 3 wt. %.
Additionally, setting time is also a requisite player affecting the use of bone cements in clinics. In our research, the ZnO-NP content was altered for control over the setting time (Table 2). In the case of the ZnO-NP content exceeding 1.0 wt.%, the setting time of ZNMPCs was extended to above 15 min, in line with clinical operation standard. This extension is probably by reason of the fact that the MgO surface is coated with ZnO-NPs, retarding MgO dissolution and thereby cause reaction rate reduction. By this way, the ZNMPC composite cements conquered the fast concretion of magnesium phosphate cements with less than 3 min. 35
As an ideal bone substitute material, implants experience gradual degradation, which makes a space for new bone tissue and followed by replacement with new bones.36,37 It was unveiled by degradation that all ZNMPC specimens would experience gradual degradation in Tri-HCl solution, displaying discrepant degrading degrees under discrepant ZnO-NPs contents (Figure 4). The introduction of ZnO-NPs moderately increased degradation rate of MPC. The higher degradability of the ZNMPCs with higher ZnO-NPs content was a result from the less compact microstructure. It appears that new bones have a 3–6-month growth cycle. Such a degradation rate of the ZNMPCs will mildly match with the new bone growth rate and promote bone repair condition. Apparently, the degrading performance of a material is chemically and physically dependent. Accordingly, control over the degradation rate by changing compositions remains requisite. To confirm the release capabilities of Zn2+ ions, the short-term release of Zn2+ ions of 1.0ZNMPC were characterized (Figure 4(d)). During the degradation, the release of Zn2+ ions was almost constant. The release of Zn2+ ions play a pivotal role in osteogenic differentiation. Nevertheless, the in vivo degradation properties and mechanisms adopted by ZNMPCs obligate more investigative studies.
It is not uncommon to judge novel biomaterials on the strength of their actions with cells in vitro. As unveiled by cell proliferation and viability results, the relative proliferation rate of ZNMPCs were significantly higher and more mBMSCs grew on surfaces of these materials than that of MPC. All ZNMPC specimens at a proper concentration showed insignificant cytotoxicity. As uncovered by cell viability results (Figure 5), ZnO-NPs at 0.5 and 1.0 wt.% impelled cell growth and the extract concentrations were below 25 mg/mL. The mBMSCs seeded on surfaces of ZNMPC exhibited clearer morphology (Figure 6). These evidences proved that the introduction of ZnO-NPs into MPC possessed good cytocompatibility and provided favorable conditions for response and proliferation of cells.
Cell differentiation is also critical for ossification. Osteoblast lineage progenitor cells undergo continuous and indistinct phases.38,39 Pre-OBs undergo active proliferation and express collagen and osteopontin (OPN) during the first 10 to 12 days. Subsequently, cell proliferation is down, and immature OBs become mature OBs with collagen type 1 alpha 1 chain (Coll-I) secretion and alkaline phosphatase (ALP) expression in order to mature extracellular matrix during the immediate postproliferative period (from 12 to 18 days). Upon accomplishment of matrix maturation, matrix mineralization emerges via the expression of various osteoblastogenic markers, such as OPN, and osteocalcin (OCN), with on-going expression of ALP and Coll-I (days 16–20). Osteoblastogenesis is regulated by Runx2, acted as the master transcription factor leading to the expression of osteoblastogenic markers, such as ALP, OCN and OPN. And the Runx2 level peaks at the immature osteoblast stage(days 0–9), but decreases in the maturation stage.40–42 ZNMPC group presented with a strikingly higher ALP activity relative to blank and MPC groups at all time points (Figure 7). Meanwhile, ZNMPC group possessed more calcium nodes of ECM mineralization (Figure 8), indicating that ZNMPCs were propulsive for the mineralization of mBMSCs. Compared to blank and MPC groups, the expressions of Coll-I, OCN, OPN, and Runx-2 in ZNMPC group were higher (Figure 9). Hence, these results collectively illustrated that ZNMPCs were propulsive for mBMSCs differentiation toward osteogenesis on account of the doping of highly bioactive ZnO-NPs with osteogenic activity, in keeping with prior research on ZnO-based composites.23,25,27
Thus, the ZNMPCs have the capacity to stimulate bone formation, which is a promising multifunctional bone repair material. The introduction of ZnO-NPs into MPCs impelled the mBMSCs differentiation toward osteogenesis and mineralization in vitro. However, in vivo research should proceed to substantiate the propulsive impact of the composite cements on bone regeneration.
Conclusion
ZNMPCs with satisfactory compressive strength, appropriate setting time and adjustable biodegrading competence were successfully designed herein. Among them, 1.0ZNMPC topped among all ZNMPCs in the compressive strength (42.3 MPa). The exothermic temperature and setting time of the ZNMPCs were adjustable and adequate for clinical applications. Compared with pure MPCs, ZNMPCs potentiated cell proliferation and viability, reinforced ALP activity and mineralization, and elevated expressions of genes involving osteogenesis in mBMSCs. Therefore, this novel ZNMPC shows promise for bone repair as a bone substitute. Whilst the exact molecular mechanisms adopted by ZNMPCs in osteogenesis remain less-documented, in-depth explanations are requisite.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors wish to acknowledge the financial support of the doctoral research initiation funds provided by the China West Normal University (No. 20E055).



