Abstract
Background
The aim of this study was to evaluate the antimicrobial efficacy and mechanical properties of dental resin composites containing different amounts of microparticulate bioactive glass (BAG).
Methods
Experimental resin composites were prepared by mixing resin matrix (70% BisGMA and 30% TEGDMA) and inorganic filler with various fractions of BAG to achieve final BAG concentrations of 5, 10 and 30 wt%. Antimicrobial efficacy was assessed in aqueous suspension against
Results
Under the dynamic contact condition, viable counts of
Conclusions
The results demonstrated the successful utilization of BAG as a promising biomaterial in resin composites to provide antimicrobial function.
Introduction
In recent decades, resin composites have achieved acceptance in aesthetic restorative dentistry due to their advantages, such as ease of handling and capability of adhesion to tooth using adhesive systems. However, resin composites tend to accumulate more bacteria on their surface during early plaque formation compared with other restorative materials and human enamel (1, 2). Bacterial accumulation on resin composites or at the tooth–restoration interface can lead to secondary caries. The breakdown of marginal areas between enamel and restorative material can provide potential pathways for bacterial reinfection and recurrence of caries (3). Moreover, the accumulation of bacteria may degrade the surface of the restorative materials and thus decrease the longevity of restorations (1). The lack of antibacterial activity of resin composites means that there is no inhibitory effect against plaque accumulation on the surface, and cariogenic bacteria such as
Resin composites comprise 2 major components: organic resin polymers and inorganic fillers in high loadings (6). Therefore, resin composites can be modified in 2 ways to obtain antimicrobial properties: alterations to the resin components or to the filler components (7). Resin components can easily be altered by the addition of soluble antimicrobials into the resin matrix (8). However, the resins containing soluble antimicrobials have shown a biphasic release profile, in which leaching of large amounts of the agent occurred within a few days followed by a dramatic decrease (9). The replacement of filler with one that has antimicrobial particles may be considered as the simplest way to alter the filler components. Several attempts have been made to replace filler components with antimicrobial particles to diminish bacterial adherence which is considered to be important in early plaque formation (8, 10–11–12). Many of these studies have employed silver to produce antibacterial activity. However, the antimicrobial effect of silver ions lasts for only a short period, and discoloration of the composite resin was found to be a significant problem (10, 11).
Bioactive glass (BAG), first introduced in the early 1970s by Hench and his coworkers, is capable of forming direct chemical bonds with both hard and soft tissues (13). BAG contains oxides of calcium, sodium, phosphorus and silicon in a proportion that provides the material with exceptional surface activity (14). Interest in potential dental applications of particulate BAG has expanded in recent years due to its biocompatible nature. The potential remineralizing effect of BAG, based on the release of calcium and phosphate ions, has also attracted much attention (15). Salehi et al (15) tested the in vitro cytocompatibility of resin composites containing BAG, using undifferentiated dental pulp cells, and reported that the cytotoxicity of composites with and without BAG must predominantly be attributed to the release of residual monomers, and not to the presence of the BAG. In a recent study conducted by Khvostenko et al (16), the flexural strength, fracture toughness and fatigue crack growth resistance of the experimental resin composites containing BAG were compared with commercial formulations. The authors reported that the mechanical properties of composites with BAG was as adequate and stable as those of successful commercial composites.
The wide-spectrum antimicrobial effect of BAG on different oral microorganisms has been reported (14, 17). To date, however, there is no published study on the antibacterial potential of BAG in resin composites. Accordingly, the aim of this study was to evaluate the antimicrobial activity of resin composite modified with the addition of microparticulate BAG. The incorporation of any additive into a composite matrix may considerably affect its mechanical characteristics, making it unsuitable as a restorative material. Therefore, in the present work, some basic mechanical properties of experimental composites, including surface roughness, compressive and flexural strengths, were also evaluated.
Materials and Methods
Preparation of resin composite specimens
The resin monomer mixture used as base material was produced by mixing bisphenol A glycidyl methacrylate (BisGMA; Sigma-Aldrich, St. Louis, MO, USA) and triethylene glycol dimethacrylate (TEGDMA; Sigma-Aldrich) at a weight ratio of 70:30. A 1 wt% photoinitiator was added to the mixture as well. The BAG granules (S53P4; BonAlive Biomaterials, Turku, Finland) were processed to a fine particle size using a planetary mill (Fritsch, Idar-Oberstein, Germany). Microparticulate BAG with an average particle size of 0.6 µm was incorporated into composite at 5, 10 and 30 wt% by replacing the same amount of silanized biostable glass filler (1-μm average particle size; Imicryl Dental, Konya, Turkey) with total filler load maintained at 70 wt%. All components were mixed in a speed mixer (FlackTek, Landrum, SC, USA) and inserted into PVC molds (8-mm diameter and 2-mm height) to produce disc-shaped specimens. Then, molds were sandwiched between 2 glass slides, and specimens were cured for 40 seconds from both sides using a light-curing unit (Valo LED, South Jordan, UT, USA). Disc-shaped specimens were used for surface roughness analysis and antimicrobial assays. Specially shaped specimens were also prepared with a similar curing regime to evaluate compressive and flexural strengths according to the ISO 9917 (18) and ISO 4049 (19), respectively. Composite specimens were designated as BAG0 (Control), BAG5, BAG10 and BAG30 according to their BAG contents.
Determination of antimicrobial activity
The antibacterial activity of experimental composites was evaluated in both aqueous suspension and biofilm matrix. Both antimicrobial assays were performed in 3 replicates.
Antibacterial activity in aqueous suspension
Lyophilized cultures of
Antimicrobial activity was quantitatively assessed under dynamic contact conditions in accordance with the ASTM E2149-13a standard (20). In brief, 4 discs (≈1 g) for each composite types sterilized by UV treatment were added to screw-cap tubes containing 1 mL of working bacterial suspensions. Then tubes were placed on an orbital shaker (Biosan, Riga, Latvia) and shaken at 220 rpm for 90 minutes. The numbers of viable bacteria in suspensions before (time 0) and after exposure were determined by plate count technique. The suspensions and additional decimal dilutions up to 10-3 were plated on Nutrient Agar (NA, Lab M), and inoculated plates were incubated at 37°C for 48 hours. The resultant colonies were counted, and microorganism counts were calculated as cfu/mL. Antimicrobial activity of specimens expressed as percentage reduction (R) value was calculated using the following equation:

where B is the initial (time 0) enumeration result (cfu/mL), and A is the enumeration result (cfu/mL) after 90 minutes of contact time.
Antibacterial activity in biofilm
The test strain of
Six discs for each composite type were placed into wells (1 disc for each well) of a 24-well tissue culture plate (Corning, Tewksbury, MA, USA). Two milliliters of bacterial inoculum was added to each well, and the plate was incubated at 37°C. Every 12 hours during the incubation period, 1 mL of spent medium was pipetted from wells and replaced with an equal volume of fresh TSB. At the end of 48 hours of incubation, discs were transferred into the corresponding wells of a new plate and washed 3 times with phosphate-buffered saline (PBS) to remove nonadherent cells. Subsequently, discs were placed in glass tubes containing 2 mL of PBS, and tubes were sonicated for 1 minutes at 35 kHz (Bandelin GmbH, Berlin, Germany) 4 times to detach bacteria from the biofilm matrix and bring them into suspension. Then the resulting suspensions and additional decimal dilutions up to 10-3 were plated on NA plates. After incubation at 37°C for 48 hours, colonies grown on plates were counted, and mean microorganism counts of 6 discs were calculated as log cfu/disc for each composite type.
Determination of mechanical properties
Surface roughness
Surface roughness (Ra) was measured on 10 disc-shaped specimens for each composite type using a surface profilometer (Mitutoyo Corp., Kawasaki, Japan). Five measurements were performed in different directions on each disc, and the overall mean of Ra values was calculated for each composite type.
Compressive strength
Cylindrical-shaped specimens with a diameter of 4 mm and height of 6 mm were prepared in split PVC molds and stored in water at 37°C prior to test. Ten specimens from each composite type were tested after 24 hours using a universal testing machine (Instron, Canton, MA, USA) at a cross-head speed of 1 mm/min. Compressive strength was determined in megapascals (MPa) by dividing the failure load (N) with the specimen cross-section area (mm2).
Flexural strength
Bar-shaped test specimens were prepared in a rectangular stainless-steel mold with dimensions of 2 × 2 × 25 mm. After a 24-hour storage in distilled water at 37°C, 10 specimens for each composite type were subjected to a 3-point bending test using a universal testing machine at a cross-head speed of 0.5 mm/min. Flexural strength was calculated in MPa with the following equation:

where s is the flexural strength, F is the load at fracture (N), l is the specimen span (mm), b is the specimen width (mm) and h is the specimen height (mm).
Statistical analysis
Results were analyzed by 1-way analysis of variance (ANOVA) using statistical software (SPSS Inc., Chicago, IL, USA). Mean values were compared using the Duncan grouping test at p<0.05.
Results and discussion
Antimicrobial activity of experimental composites
Reductions in the levels of
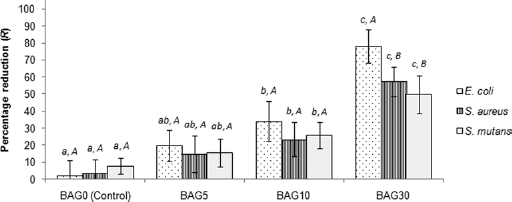
Antimicrobial activity of experimental composites in aqueous suspension (error bars denote standard deviations). Reductions with the same uppercase letter did not differ significantly (p>0.05) for the same composite type. Reductions with the same lowercase letter did not differ significantly (p>0.05) for the same microorganism. BAG = bioactive glass.
The results of the present study suggest the potential broad-spectrum of antibacterial activity of BAG-containing resin composite. In accordance with these results, substantial reduction values were reported in previous studies dealing with the evaluation of antimicrobial efficacy of BAG both alone and in combination with composite matrices (14, 21–22–23). Particulate BAG was found to have a strong antibacterial effect against certain supragingival and subgingival bacteria (24). Yli-Urpo et al (17) demonstrated that glass-ionomer cement disks containing 30% BAG exerted antimicrobial effects on oral microorganisms.
The antimicrobial action of BAG in an aqueous environment has been explained by 2 mechanisms (14, 21, 25): The first mechanism is the increase in pH and the consequent change in the osmotic pressure due to releasing of ions such as sodium, calcium, phosphate and silicate. These changes can lead to cellular injury and inactivation of bacterial enzymes. The alkalinization of the medium may also hinder establishment of the proton motive force for adenosine triphosphate (ATP) synthesis. The second mechanism is that debris from BAG particles can react with the cell envelope, causing leakage of cell contents and eventually microbial lysis. The results of Hu et al (25) on
Enumeration results obtained after contact time at a BAG concentration of 30% indicated a significant difference between viabilities of the gram-negative (
The levels of viable cells in 2-day-old
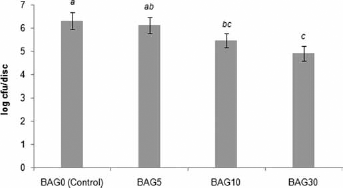
Levels of viable cells in
The possible antibacterial mechanism of BAG against biofilm cells has been postulated to be the result of the inactivation of bacteria upon coming into contact with its surface (29). Steinberg and Eyal (3) reported a positive correlation between the degree of bacterial adhesion to resin composite and viable counts of bacteria in the biofilm. In accordance with this finding, BAG has been proposed as a promising material for dental and medical applications due to its antiadhesion properties and growth-inhibitory effects on bacterial biofilms (30–31–32). Results of a study by Allan et al (30) in which the antibiofilm effects of BAG were evaluated against subgingivally modeled mixed species biofilms indicated that particulate BAG has the potential to reduce bacterial colonization of its surface.
Mechanical properties of experimental composites
Surface roughness promotes bacterial adhesion and plaque retention, which may influence the longevity of the restoration. Therefore, it is the most frequently used parameter in assessing the surface quality of restorative materials (33). The surface roughness (Ra) values of resin composites with different amounts of BAG are shown in Table I. These values indicated that there was no significant difference (p>0.05) between modified composites and BAG0 (control). In addition to the finishing and polishing treatments, surface roughness of the composites is mainly associated with the type, shape, size and distribution of the inorganic fillers (33, 34). Accordingly, the similarity observed in the Ra values can be attributed to the comparable average particle sizes of the filler (1 μm) and BAG (0.6 μm) used to modify resin composite.
Mechanical properties of experimental composites
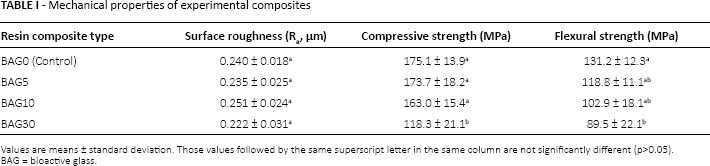
Values are means ± standard deviation. Those values followed by the same superscript letter in the same column are not significantly different (p>0.05).
BAG = bioactive glass.
Compressive strength of resin-based restorative materials has a particularly important role under the compressive forces of mastication. Resin composites usually replace a large bulk of tooth structure, and they should provide sufficient strength to resist intraoral compressive and tensile forces (35). A statistical comparison of compressive strength values, presented in Table I, showed that there was no significant difference between BAG0, BAG5 and BAG10 (p>0.05). However, an increase of BAG concentration to 30% resulted in a significant decrease in the compressive strength of the composite (p<0.05). Similar to our findings, it was previously reported that higher mass fractions of additive particles led to the formation of bundles and agglomerations leading to defects and flaws which in turn deteriorated mechanical properties of the composite (36, 37).
Flexural strength is used to evaluate the failure stress of the material in bending (38). It has been shown to be a more discriminating test and more sensitive to subtle changes in a material's substructure compared with the compressive strength test (39). The difference in flexural strength between BAG0, BAG5 and BAG10 was found to be statistically nonsignificant (p>0.05), whereas flexural strength of BAG30 was significantly lower compared with BAG0 (p<0.05) (Tab. I). Our results were in accordance with a recent study (16) that showed that flexural strength of composites modified with BAG was unaffected by increasing BAG content up to 15%.
Conclusions
The ability of bacteria to propagate and produce organic acids is an essential factor in the dental caries process. Cariogenic species such as
Footnotes
Financial support: No grants or funding have been received for this study.
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
