Abstract
Background
Rough surface topography enhances the activation of Wnt canonical signaling, a pathway required for osteoblast differentiation. The present study investigated the effects of the modulation of prostaglandin E2 (PGE2) signaling on osteoblastic differentiation on titanium surfaces for endosseous implants with different topographies.
Methods
C2C12 cells were plated on polished or acid-etched/sand-blasted (SLA) titanium discs and stimulated with 1 μM PGE2 or 100 nM cyclooxygenase inhibitor indomethacin. Activation of Wnt canonical signaling was measured with a reporter system. Gene expression was measured in the same cell system by real-time polymerase chain reaction (RT-PCR). Osteoblastic MC3T3 cells were then plated on polished or SLA titanium discs with or without indomethacin, and their proliferation and the expression of osteoblast-specific genes was assessed by RT-PCR. Cell morphology was furthermore studied on SEM, and cell adhesion was assessed by fluorescent labeling of focal adhesion.
Results
PGE2 decreased Wnt signaling stimulation in cells growing on polished or SLA surfaces, while indomethacin increased the expression of Wnt target genes in C2C12 and MC3T3 cells, by reporter assay. Moreover, indomethacin increased the expression of early differentiation marker alkaline phosphatase in MC3T3 cells on polished discs and of late marker osteocalcin in cells on SLA titanium.
Conclusions
Prostaglandin signaling affects the activation of Wnt canonical pathway in osteoblastic and mesenchymal cells on microstructured surfaces.
Introduction
It is widely accepted that mesenchymal cells are capable of responding not only to the chemical stimuli to which they are exposed, but also to the mechanical forces applied on them and to the topographical features of the surface on which they grow (1-2-3-4-5-6). Several studies have demonstrated that rough titanium surfaces may induce specific cellular responses including control of cell proliferation and differentiation to the osteoblastic lineage (7). In spite of the surge of interest of the scientific community in this well-documented phenomenon, the underlying mechanisms are still poorly understood. Lack of such knowledge hinders the design of better and improved endosseous implant surfaces that can support bone formation at the interface between host tissues and the biomaterial, thereby achieving a more predictable osseointegration.
Some recent studies have provided compelling evidence that the autocrine and paracrine regulation of Wnt signaling plays a major role in bone cell responses to mechanical loading (8, 9). Wnt (wingless-type MMTV integration site family) growth factors include a large number of members that are required for bone formation during development, and bone regeneration during wound healing. Wnt signaling plays an essential role in osteoblast differentiation via a canonical pathway that involves release and nuclear translocation of bone formation at the bind, then T cell-specific transcription factor (TCF) in the nucleus, activating several target genes (10). Wnt proteins can also regulate cytoskeletal organization via a noncanonical, planar cell polarity (PCP) pathway, by activation of small GTPase RhoA (11, 12). Recent works have independently demonstrated that surface topography can regulate the activation of canonical and noncanonical signaling and thus contribute to osteoblast differentiation and activity through these pathways (13-14-15-16-17-18-19-20). It has also been reported that a key mechanism that mediates surface effects on cell differentiation is modulation of cyclooxygenase activity, and thus production and release of prostaglandins (21, 22). The present paper thus aimed at investigating whether prostaglandin E2 (PGE2) signaling could help control Wnt canonical pathways in osteoblastic cells growing on titanium surfaces with different topography.
Methods
Titanium surfaces
Polished and acid-etched, sand-blasted (SLA) samples were kindly provided by Straumann Institut AG (Basel, Switzerland). The samples were provided as sterile discs of 1-mm thickness and 16-mm diameter, and were used in 24-well plates (Euroclone, Pero, MI, Italy) for the biological assays.
Cell cultures
The MC3T3-E1 cell line was obtained from the American Type Culture Collection (LGC Standards S.r.L., Sesto S. Giovanni, MI, Italy) and cultured in Dulbecco's modified Eagle'a medium (DMEM, PAA; GE Healthcare, Uppsala, Sweden), 10% fetal bovine serum (FBS; Gibco, ThermoFisher, Waltham, MA, USA), 1% penicillin and streptomycin (Penstrep; Sigma-Aldrich, St. Louis, MO, USA) and 1% glutamine (Sigma-Aldrich, St. Louis, MO, USA) as subculture medium. For gene expression assays, cells were plated on polished or SLA surfaces at the density of 30,000 cells/well in 24-well plates, in triplicate. After 24 hours, the subculture medium was replaced with subculture medium added with 250 μM ascorbic acid (Sigma Chemicals, St. Louis, MO, USA).
The C2C12 cell line was obtained from the European Catalog of Cell Cultures (Health Protection Agency Culture Collections, Salisbury, UK) and grown in subculture medium. For reporter assays, C2C12 cells were plated on titanium surfaces in OptiMEM (Invitrogen, San Giuliano Milanese, MI, Italy), 5% FBS, 1% Penstrep at the density of 125,000 cells/well, and the cells were assayed after 24 hours. C2C12 is a popular cell line to study Wnt canonical signaling because of the abundance of molecular machinery for this pathway.
Scanning electron microscopy
Cells were seeded on titanium surfaces at the density of 2 × 104 cells/cm2 in triplicate and were studied after 48 hours of culture. At each time point, they were fixed with 2.5% glutaraldehyde in 0.1 M sodium cacodylate buffer (pH 7.3) for 30 minutes at room temperature. They were then dehydrated through a series of alcohols, and then critical point dried with liquid carbon dioxide (CPD 030; Baltec, Wallruf, Germany). Specimens were then sputter-coated with a gold-palladium layer (Plano, Germany) using a SCD 040 coating device (Balzer Union, Wallruf, Germany). Samples were studied using scanning electron microscopy (SEM) with a Zeiss DSM 950 at an accelerating voltage of 10 KV (Zeiss, Jena, Germany).
Cytoskeleton and focal adhesion analysis
The intracellular cytoskeleton protein vinculin was used as a marker for focal adhesions (FAs). For FA analysis, a 48-hour time point was used. Longer experimental time points were avoided, because confluent cells overlapped and made the analysis difficult. Cells were fixed with 4% paraformaldehyde for 10 minutes. They were then permeabilized with 0.1% Triton X-100 for 5 minutes. Nonspecific binding sites were blocked with 1% bovine serum albumin in phosphate-buffered saline (PBS) for 20 minutes. Cells were then stained with an anti-vinculin antibody (MAB3574; Chemicon) for 1 hour. Nuclear counterstaining was performed by incubation with DAPI (D1306; Molecular Probes, Invitrogen) for 5 minutes. All of the steps were carried out inside the culture well at room temperature. The discs were then mounted using an antifade-mounting medium (P7481; Molecular Probes, Invitrogen) for photo bleaching reduction. Samples were examined using a Nikon Eclipse 90i microscope equipped for fluorescence analysis.
Constructs and reagents
A plasmid DNA vector containing a responsive element to the transcription factor TCF driving a firefly luciferase gene (TCF-Luc) was used for the experiments. A vector expressing Renilla Luciferase under the control of a constitutively active promoter was used to normalize. The constructs were purchased from SABioscience (Frederick, MD, USA). PGE2 and indomethacin were purchased from Sigma-Aldrich. Recombinant mouse Wnt3a was purchased from R&D Systems (Minneapolis, MN, USA).
Cell proliferation assay
To evaluate cell proliferation, MC3T3 cells were seeded at a density of 2 × 104 cells/500 µL per well in DMEM 10% FBS in 24-well plates (Falcon, Becton Dickinson Europe, Meylan, France) containing polished or SLA titanium discs, and after 2 hours (time 0), 500 µL of complete medium was added to each well. Cell growth was assessed 24 and 96 hours after seeding by MTT assay (Roche Applied Science, Penzberg, Germany) according to the manufacturer's recommendations. Briefly, we removed 500 μL of medium from the wells, added 50 µL MTT labeling reagent (final concentration 0.5 mg/mL), incubated for 4 hours and then added 500 mL of solubilization solution. After overnight incubation, we read absorbance at 570 nm with a Multiscan Ascent spectrophotometer (Titertek Instruments Inc., Huntsville, AL, USA).
Real-Time PCR
Total RNA was purified from cell cultures using a Cell-to-cDNA kit (Ambion, Monza, MI, Italy). TaqMan quantitative real-time polymerase chain reaction (RT-PCR) was performed as previously described using the following primer probe sets from Applied Biosystems (Foster City, CA, USA): Axin2 (Mm01265783_m1): alkaline phosphatase (Mm00475834_m1); Wisp-2 (Mm00497471_m1); beta-catenin (Mm00483039_m1); osterix (Mm00504574_m1); osteocalcin (forward 5’-GCTGCGCTCTGTCTCTCTGA-3’; reverse 5’-TGCTTGGACATGAAGGCTTTG-3’; probe 5’-FAM-AAGCCCAGCGGCC-NFQ-3’); housekeeping gene, mouse ribosomal protein S2, ChoB (forward 5’-CCCAGGATGGCGACGAT-3’; reverse 5’-CCGAATGCTGTAATGGCGTAT-3’; probe 5’-FAM-TCCAGAGCAGGATCC-NFQ-3’).
Reporter assays
The reporter assays were performed with a Dual-Luciferase Reporter assay system (Promega, Madison, WI, USA) according to the manufacturer's instructions. The samples were read with a Glomax 20/20 Luminometer (Promega) with double injectors.
Statistical Analysis
Data were analyzed using Prism 6 (GraphPad, La Jolla, CA, USA). All values are reported as the means ± standard deviation of 3 repeated experiments. Differences between group means were evaluated with the ANOVA statistical test, and differences were considered significant when the p value was <0.05.
Results
Titanium microtopography affects cell shape, adhesion and differentiation
Two titanium surfaces were considered for the present study, a smooth, polished surface (Fig. 1A) and a rough, sand-blasted/acid-etched (SLA) surface (Fig. 1B). SLA discs presented with an irregular profile, with sharp ridges surrounding pits and cavities. Osteoblastic MC3T3 cells attached and proliferated on both surfaces, although with a strikingly different shape. Cells were flat and large on polished discs and adhered tightly to the titanium substrate, whereas they often displayed a star-like appearance on SLA discs, with numerous cellular extroflections that anchored the cell body to the irregularities of the substrate (Fig. 1D). Consistently, when FAs were visualized by fluorescent labeling of the intracellular vinculin protein, focal contacts appeared distributed along the whole cell body in MC3T3 cells on polished discs (Fig. 2A). Interestingly, FA along the cell periphery appeared elongated, and oriented across the cell body. Cells on rough surfaces, however, presented with a stronger signal along the cell margins, and especially on the extroflections, sites that presumably are involved in cell anchoring on the substrate. Longer, dash FAs were present but less numerous (Fig. 2B).
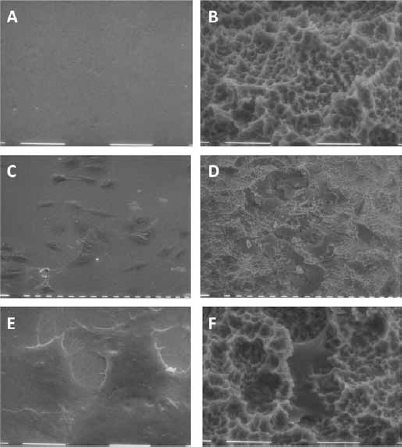
Scanning electron microscopy (SEM) microphotographs of polished (
When cultured on polished or SLA discs for up to 96 hours, MC3T3 cells showed an increase in mRNA levels for osteocalcin over time, and its expression was always significantly higher in cells growing on rough than on polished surfaces (Fig. 2C).
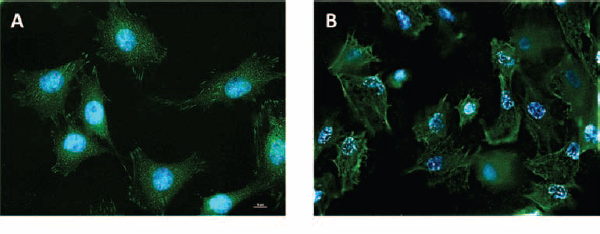
Transmission microscopy microphotographs of MC3T3 cells on (
PGE2 affects Wnt canonical signaling in cells growing on microstructured surfaces
Figure 3A shows the effect of PGE2 on Wnt/β-catenin signaling in cells growing on smooth and rough titanium surfaces; to this purpose, we transfected mesenchymal C2C12 cells with a reporter system for TCF, using a reporter construct that expressed firefly and Renilla luciferase and stimulated them with recombinant Wnt3a, an activator of Wnt canonical signaling. Interestingly, 1 μM PGE2 decreased the levels of luciferase activity in C2C12 on rough titanium surfaces and tended to decrease it on smooth surfaces, although not significantly. Consistently, inhibition of PGE2 synthesis by 100 nM indomethacin, a cyclooxygenase (Cox) inhibitor, increased the activation of Wnt canonical signaling in cells on titanium surfaces (Fig. 3B).
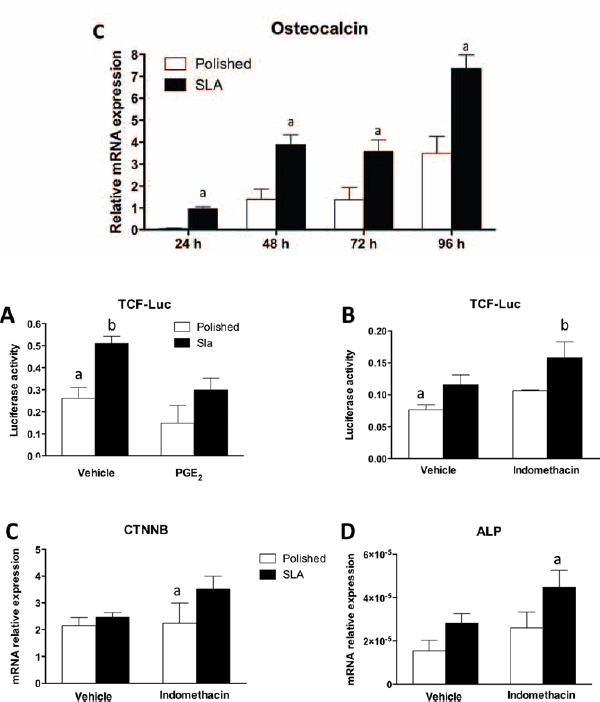
Reporter assay of C2C12 cells on polished and sand-blasted/acid-etched (SLA) titanium surfaces in presence of 1 µM PGE2 (
PGE2 modulates gene expression in cells growing on microstructured surfaces
We then investigated whether prostaglandin PGE2 played a role in modulating gene expression in C2C12 cells on smooth or rough surfaces. As shown in Figure 3C, D, the mRNA levels of Wnt target gene alkaline phosphatase (ALP) were increased in cells on SLA surfaces after inhibition of Cox with 100 nM indomethacin, in agreement with the reporter assay. Noteworthy is the fact that indomethacin increased the expression of β-catenin in C2C12 cells on rough surfaces, providing a possible explanation for the observed effects.
Indomethacin affects viability of MC3T3 cells growing on smooth and rough surfaces
We then moved on to an in vitro model of bone cells and tested whether the murine calvaria MC3T3 cell line was affected by PGE2 inhibition. First we tested cell viability using a well-established method, MTT, an assay that measures cell redox activity. When cultured on rough discs, MC3T3 cells appeared to grow at a slower pace than on polished substrates, as their viability level was significantly lower. However, when PGE2 synthesis was inhibited by indomethacin, no difference in viability between smooth or rough surfaces was detected after 24 or 96 hours of culture (Fig. 4A, B).
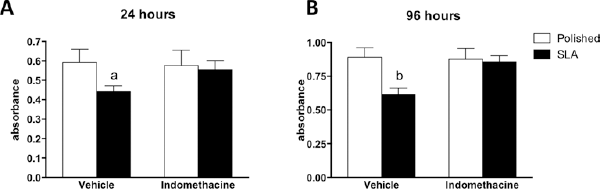
Viability of MC3T3 cells after 24 hours (
Indomethacin affects the expression of different Wnt pathway components in MC3T3 cells growing on smooth and rough surfaces
We then investigated the expression of Wnt target genes in MC3T3 cells after 96 hours of culture. We observed an increase in the mRNA levels of Wnt target genes Axin-2 and Wisp-2 in cells on SLA surfaces after addition of 100 nM indomethacin (Fig. 5). Moreover, Cox inhibition increased the expression of β-catenin in cells on both surfaces and significantly increased ALP mRNA on smooth surfaces.
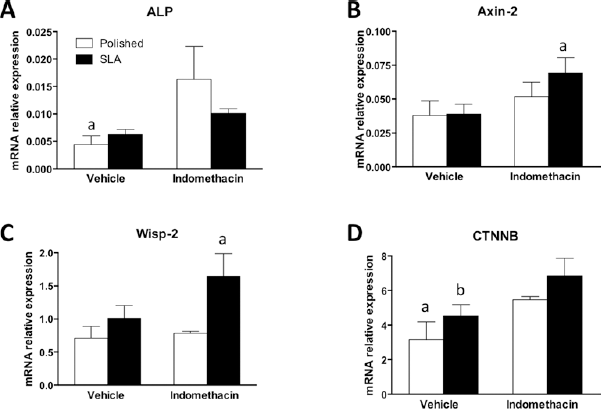
Real-time PCR analysis of mRNA for (
Indomethacin affects the expression of osteoblastic marker in MC3T3 cells growing on smooth and rough surfaces
We then measured the expression of osteoblast-specific genes osterix (Osx) and osteocalcin (OCN) (Fig. 6). Indomethacin significantly increased the levels of transcript for Osx and OCN in MC3T3 on SLA discs after 96 hours of culture.
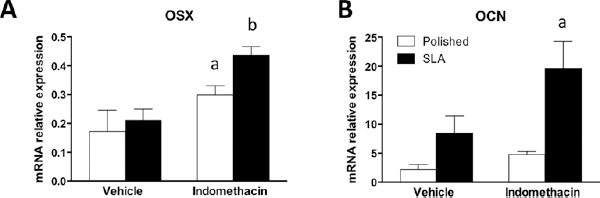
Real-time PCR analysis for mRNA for osterix (OSX) (
Discussion
Implant topography can profoundly affect the differentiation and activity of mesenchymal and osteoblastic cells, and this can have important repercussions on the clinical performance of medical devices. It is broadly accepted that rough topographies in the micron range can improve the clinical success of endosseous implants (23, 24), and a better understanding of the cellular mechanisms that control this response could further boost implant therapies in critical clinical situations. When confronted with implant surfaces with diverse topography, cells interact with very different microenvironments (Fig. 1). To adapt to such substrates, cells possess the capability to reorganize their shape and structure (Fig. 1), including their FAs, the molecular machineries responsible for guaranteeing an adequate adhesion on the substrate (Fig. 2A, B). What is most striking, however, is that rough surfaces promote a higher degree of differentiation in osteoblastic cells (1-2-3). Ever since this was discovered, a great deal of attention has been paid to the signaling mechanisms that could bring about such a change in cell behavior. The present study focused on the role of PGE2 inhibition on the regulation of Wnt canonical signaling in osteoblastic MC3T3 cells on microrough surfaces. Wnt canonical signaling is an important pathway for osteoblast differentiation (25), and it has been shown to be involved in osteoblastic responses to surface topography (13, 17). PGE2 is a cytokine with broad activity ranging from inflammation (26) to intercellular signaling (27-28-29), and more importantly to our purposes, it has been reported to be able to regulate osteoblastic differentiation on micropatterned titanium (21, 22). The clinical effects of the pharmacological regulation of PGE2, however, remain controversial (30, 31). Classic works by Boyan et al (21, 22, 32) have shown that indomethacin inhibits the effect of topography on osteocalcin expression and, to a lesser extent, ALP in MG63 human osteosarcoma cells. Recent reports 2006 and 2010 in the literature have pointed at examples where PGE2 may affect canonical Wnt signaling (33, 34), particularly in bone cells (8, 34). Somewhat surprisingly, our data show that PGE2 inhibits Wnt canonical signaling in the murine MC3T3-E1 cell line on rough SLA surfaces, as measured by reporter assay using C2C12 cells, a cell line that is very sensitive to Wnt regulation (Fig. 3A). This actually stands in agreement with findings of dual Wnt modulation by PGE2, with high cytokine doses, similar to what we used for this study, inhibiting Wnt canonical signaling in MC3T3-E1 cells (35). Consistently, we observed that cyclooxygenase inhibition by 100 nM indomethacin increased Wnt canonical signaling on SLA surfaces by reporter assay (Fig. 3B) and increased the expression of mRNA encoding for Wnt target genes Axin-2 and Wisp-2 on rough discs (Fig. 5B, C) by RT-PCR. Noteworthy, our data show that the expression of βrcatenin was increased by indomethacin in C2C12 cells on SLA discs (Fig. 3C) and on both surfaces in MC3T3 cells (Fig. 5D). The Wnt canonical pathway relies on the cytoplasmic t genes Axin-2 and Wisp-2 on rough discs (Fig. 5B, of PGE bring a, which is normally sequestered and targeted for proteosomal degradation, upon Wnt activation, is released and translocates to the nucleus, where it initiates a complex transcription program (10). Our data thus offer a rationale as to why Wnt signaling may have been increased in the presence of indomethacin. Increased levels of nd targeted for prot, however, are insufficient to explain the stimulation profile of ALP and OCN expression by indomethacin in MC3T3 cells. ALP expression was higher in cells on polished surfaces in the presence of indomethacin (Fig. 5A), whereas PGE2 inhibition significantly increased OCN expression on rough surfaces, indicating a differential effect by surface topography, most likely through ancillary molecular mechanisms. Interestingly, ALP is considered a marker of an earlier differentiation stage than osteocalcin, whose expression is up-regulated in osteoblasts at a later stage (25). The expression of osterix, a Wnt-regulated transcription factor whose expression is increased at the preosteoblast stage (25) is somewhat consistent with this idea: its transcript levels are increased in MC3T3 cells on both surfaces (Fig. 6A), mediating the transition from an earlier differentiation stage to a later, more mature osteocalcin-producing stage. The discrepancies of our findings with those in the available literature could in part be explained by different responses from distinct cell types, and indeed it has been shown that indomethacin increases ALP expression in MC3T3 cells (36, 37) in plastic tissue culture wells and also that up to 1 lls an2 inhibits Runx2 signaling in periodontal ligament cells (38). Our findings are also at least partially supported by Dean et al, who reported that indomethacin increased ALP expression on micro rough discs in MG63 cells (31). Factors like PGE2 or indomethacin dose and timing of application in culture have also been proven to be relevant in determining their overall effect, thus indicating that the action of PGE2 is indeed complex and must be finely tuned (31, 35). Moreover, the discovery of isoforms of PGE2 receptors with distinct expression and effects must be taken into account when interpreting the results reported in the literature (39, 40).
Taken together, these results indicate complex interactions between PGE2 and Wnt canonical signaling, which demand caution in endorsing cellular models for the study of PGE2 effects in bone responses to titanium implants. Our data support the idea that further studies will have to specifically address components of the prostaglandin pathway to investigate whether it is possible to improve bone responses to endosseous implants, possibly relying on selective modulators of prostaglandin receptors (39, 40).
Footnotes
Acknowledgement
The authors would like to thank Straumann Institut AG (and in particular Dr. Appert), Basel, Switzerland, for kindly providing the titanium surfaces used in the present study. The authors are also grateful to Dr. Marco Vitale and Mr. Davide Dallatana, Department of Biomedical, Biotechnological and Translational Sciences, University of Parma for their precious advices and technical assistance. The authors would further like to thank Dr. Martina Lumetti.
Financial support: The study was funded by grant 839_2012 from the ITI Foundation (Basel, Switzerland) and FIL research grant from the University of Parma.
Conflict of interest: The authors have no conflicts of interest to disclose.
