Abstract
Background
The amniotic membrane is a favorable biomaterial to apply in the field of tissue engineering because of its unique biological properties. Human amniotic membranes consist of 2-layered sheets containing numerous growth factors, cytokines and other bioactive substances.
Methods
In this study, we explored the potential of amnion membrane extracts (AME) and amnion/chorion membrane extracts (A/CME) to promote osteogenic differentiation of osteoblast-like (MG-63) cells. MG-63 cells were cultured in osteogenic induction medium (OIM) with or without 100 µg/mL of AME or A/CME. To determine the early and late differentiation of osteogenesis, alkaline phosphatase (ALP) activity and calcium deposition were measured at 3, 7, 10 and 24 days. Expression of specific genes associated with osteogenic differentiation, including osteocalcin (OCN), osteopontin (OPN), runt domain-containing transcription factor (Runx2) and osterix (OSX) was also determined.
Results
In vitro experiments demonstrated that A/CME increased ALP activity, osteogenic gene expression and mineralization under osteogenic-inducing conditions. Notably, we found that A/CME contained growth factors related to osteogenesis, including fibroblast growth factors and transforming growth factors, which potentially promoted osteogenic differentiation of MG-63 cells to a greater extent than AME.
Conclusions
These results indicate that A/CME is capable of providing growth factors and other substrates for osteogenic differentiation, which significantly increased the efficacy of osteogenesis in MG-63 cells. Taken together, the results of this study suggest that human A/CME is a promising biomaterial with therapeutic potential in bone regeneration applications.
Introduction
The human amniotic membrane is composed of a 2-layered avascular sheet comprising amnion and chorion membranes that form an outer limit of sac involved in fetal protection. The amnion/chorion membrane (A/CM) is conjoined by several layers to form the fetal membrane. The amnion membrane (AM) and chorion membrane (CM) are loosely connected by a sponge layer, which can be easily separated.
The AM component of A/CM allografts has been used as a natural scaffolding material for tissue-engineering applications since the early 20th century (1). AM is a thin and flexible translucent membrane containing numerous growth factors and cytokines, making the structure an excellent candidate as a scaffold in tissue engineering. The unique functional properties of AM include antiscarring, antiinflammatory and antibacterial capacity as well as low immunogenicity (2-3-4). Therefore, AM has been applied in several clinical processes, notably skin injury (5-6-7-8) and ophthalmic surgery (9-10-11).
A/CM is a thicker and more opaque membrane compared with the single layer of AM. Similar to AM, A/CM allografts have also been used for wound healing since the early development of A/CM allografts (12). While some studies have described the efficacy of A/CM as a scaffold (13, 14), this allograft has limitations for broader clinical application because of the difficulties associated with its preservation and high immunogenicity. In a recent study, Koob et al showed that A/CM contains 4-5 folds more cytokines and growth factors compared with an equal surface area of a single layer of AM alone (15). Cytokines and growth factors are known to govern cellular function through intracellular signaling pathways that regulate biological processes such as cell migration, proliferation and differentiation. For example, epidermal growth factor (EGF) and transforming growth factor- β (TGF-β) are reported to influence epithelial cell migration and regeneration (9). Fibroblast growth factor 18 (FGF18), a newly identified FGF family member, has been implicated in cartilage regeneration and chondrogenesis in an injury-induced osteoarthritis (OA) rat model (16). In addition, it is widely known that growth factors including basic FGF (bFGF), TGF-β and insulin-like growth factor I (IGF-I) play important roles in new bone formation and bone repair (17-18-19). However, the role of growth factors in A/CM is not completely understood. Therefore, with the attractive chemical constituents of human amniotic membranes, we evaluated the effect of amnion/chorion membrane extracts (A/CME) on osteogenic differentiation of MG-63 cells.
Recent studies have shown that stem cells from human AM and acellular human AM matrix can induce osteogenic differentiation (20, 21). However, the preparation and application of these stem cells is technically challenging. In addition, decellularization of the scaffold from human AM is time consuming and could result in the loss of growth factors and cytokines during processing. Extracts from AM and A/CM allografts are conveniently prepared, easy to preserve and feasible to apply in combination with other treatments and approaches, including stem cell therapy (22). Moreover, the bioactive effects of A/CME on osteogenesis are currently unknown.
In this study, we examined whether AME and A/CME could induce osteogenic differentiation of osteoblast-like cells (MG-63 cells) when cultured with osteogenic induction medium (OIM) consisting of ascorbic acid, β-glycerophosphate and dexamethasone. We observed different growth factor compositions in AME and A/CME, which contained EGF, bFGF and TGF-β. Collectively, these factors might regulate the differentiation of MG-63 cells through differential activity of alkaline phosphatase (ALP), calcium deposition and osteogenic gene expression. Our study revealed that human A/CME is a more promising biomaterial than AME for stimulation of osteoblast function, suggesting a potential role for this extract as a novel therapeutic material for bone regeneration and bone fracture treatment.
Materials and Methods
Preparation of extracts from human amniotic membrane
To prepare AME and A/CME, human amniotic membrane matrices were collected from donors after caesarian sections with the approval of the Institutional Review Board (KUGH14239-002) of Korea University Guro Hospital (Seoul, Korea). Donors who had gestational diabetes, preeclampsia or infectious disease (specifically HIV 1-2, HBV, HCV) were excluded. Written informed consent was obtained from each donor after explanation of this study. AME and A/CME were prepared according to the method of He et al (23). Briefly, tissues were washed 3 times with phosphate-buffered saline (PBS) containing antibiotics (100 U/mL penicillin and 100 U/mL streptomycin) to remove blood clots, and then sliced into small pieces, frozen using liquid nitrogen and ground into fine particles using a mortar and pestle. Particles were mixed with PBS at a ratio of 1:1 weight (g) to volume (mL) and homogenized on ice for 1 hour, followed by centrifugation. After centrifugation, supernatants were filtered through a 0.22-µm-pore-size membrane. Protein concentration in the supernatants was determined by DC protein assay (Bio-Rad, Hercules, CA, USA) and stored at -80°C until needed. Two amniotic membranes were obtained from 2 different donors and then prepared for the experiments.
Histology
AM, CM and A/CM samples were fixed in 4% formalin overnight at 4°C. Samples were dehydrated and embedded in paraffin. Histological sections (5-µm thickness) were stained with hematoxylin and eosin (H&E).
Enzyme-linked immunosorbent assay
AME and A/CME samples were prepared, and the concentration of various growth factors, including EGF, FGF and TGF-β, was quantified using corresponding Quantikine® enzyme-linked immunosorbent assay (ELISA; Human EGF, bFGF, TGF-β) Immunoassays (R&D system, Minneapolis, MN, USA). Briefly, monoclonal antibodies specific for either human EGF, bFGF or TGF-β were precoated onto microplates. Standards and samples were added to the wells, and growth factors were bound by immobilized antibodies. After unbound substances were washed away, enzyme-linked polyclonal antibodies specific for either EGF, bFGF or TGF-β were added to the wells. After washing, a substrate solution was added for development of a color reaction. Optical density was measured at 450 nm using a microplate reader.
Cell culture and in vitro osteogenic differentiation
MG-63 cells were cultured in regular growth medium consisting of Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal bovine serum (Gibco BRL, Grand Island, NY) and 100 U/mL penicillin/streptomycin (Gibco BRL). Cells were maintained in a humidified atmosphere of 5% CO2 at 37°C. For in vitro osteogenic differentiation, cells were seeded in a 24-well plate (1 × 105 cells/well), and medium was changed to OIM when 90% confluence was reached. OIM contained 10 nM dexamethasone (Sigma, St. Louis, MO, USA), 0.2 mM ascorbic acid (Sigma) and 10 mM β-glycerophosphate (Sigma) in regular growth medium. Cells were provided with fresh OIM every 2-3 days.
Alkaline phosphatase activity
After 3, 7 and 10 days of culture in OIM with or without A/CME, MG-63 cells were lysed, and ALP activity was measured using a SensoLyte® pNPP Alkaline Phosphatase Assay Kit (colorimetric; ANASPEC, Fremont, CA, USA) according to the supplier's protocol. ALP activity was determined using a microplate reader at a wavelength of 405 nm.
Real-time reverse transcriptase polymerase chain reaction
Total RNA was isolated from MG-63 cells using an RNeasy® Mini Kit (Qiagen, Mississauga, ON, Canada) according to the manufacturer's instructions. One microgram of RNA was reverse-transcribed using a PrimeScript™ 1st strand cDNA Synthesis Kit (Takara Bio, Tokyo, Japan) and real-time polymerase chain reaction (PCR) was performed using an ABI Prism 7300 Detection System (Applied Biosystems, Foster City, CA, USA). Each reaction mixture contained 10 μL of 2X LightCycler 480 SYBR Green I Master (Roche, Penzberg, Germany), 1 μL of 5 pmol of both sense and antisense primers, 0.4 μL of 50X Roxy dye (Applied Biosystems, Foster City, CA, USA) and 1 μL of cDNA in a final volume of 20 μL. The amplification was carried out in 45 cycles of 15 seconds of denaturation at 95°C followed by 1 minute of annealing at 60°C. Relative mRNA expression was analyzed using the 2(-∆∆Ct) method and normalized to GAPDH expression. Specific primers sequences used for real-time PCR are listed in Table I.
Sequences of oligonucleotide primers, annealing temperatures and product sizes for quantitative RT-PCR

RT-PCR = reverse transcriptase polymerase chain reaction.
Calcium assay and alizarin red S staining
For the calcium assay, MG-63 cells were cultured in OIM with or without AME and A/CME for 24 days. At day 24, cells were washed with PBS, and 0.6-N HCl was added. After decalcification, the supernatant was used to measure calcium content using a QuantiChrom™ Calcium Assay Kit (DICA-500; BioAssay Systems, USA) according to the manufacturer's instructions. The amount of mineral deposition was determined using a microplate reader at a wavelength of 612 nm.
For alizarin red S staining, cells were cultured in the presence and absence of amniotic extracts with OIM for 9 days. Differentiated cells were washed with PBS and fixed in 4% paraformaldehyde for 15 minutes. After fixation, cells were washed twice with distilled water, and staining was performed by addition of an alizarin red solution (Millipore, Darmstadt, Germany) for 20 minutes at room temperature. Finally, cells were washed 3 times with 1 mL distilled water, and the color change was evaluated. The matrix mineralization was quantified following addition of 1 mL/well 100 mM cetylpyridinium chloride (Sigma).
Statistical Analysis
A 1-way ANOVA (Microsoft Excel; Microsoft, Redmond, WA, USA) was used to determine statistically significant differences at each time point or dose. Scheffe's F-test was used to correct for multiple comparisons when statistically significant differences were identified by ANOVA. A p value of >0.05 or >0.01, as indicated, was considered to be a statistically significant difference. All experiments were performed at least in triplicate, and data were expressed as means ± standard deviation (SD).
Results
Histology of AM and A/CM
We first performed a detailed examination of AM and A/CM structures. Histological staining of A/CM tissue sections clearly showed amnion and chorion layers (Fig. 1). As shown in Figure 1, the AM consists of a single layer of epithelial cells aligned on a thick basal membrane connecting to a CM through sponge layers. Under this intermediate (sponge) layer, the CM is composed of a basement membrane, reticular layer and trophoblast layer.
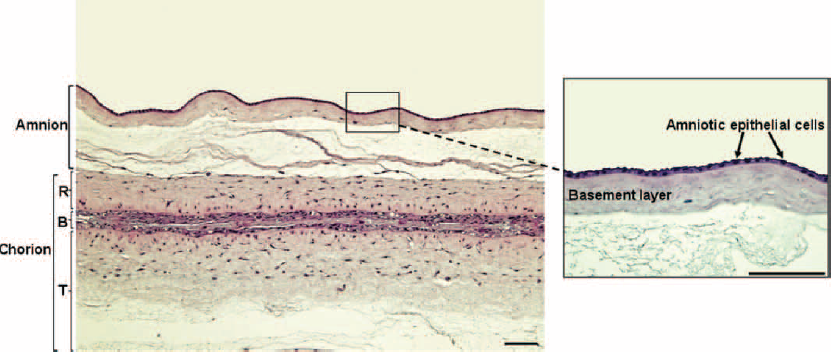
Histology of amnion and chorion membranes in human amniotic membranes. Representative image of an hematoxylin and eosin (H&E)-stained human amniotic membrane, which clearly shows the amnion and chorion layers (left panel). The right micrograph shows a magnified view of the amnion membrane, consisting of a flattened single layer of amniotic epithelial cells present on the basement membrane. The chorion membrane is composed of a reticular layer (R), basement layer (B) and trophoblast layer (T) under the amnion membrane. Scale bars represent 100 µm.
Composition of growth factors in AME and A/CME
Next, we quantified protein contents of 3 representative growth factors, EGF, bFGF and TGF-β in AME and A/CME preparations. The measured picograms of growth factors were normalized to the wet weight of the original tissue after washing (pg growth factor/mg tissue). ELISA demonstrated that bFGF and TGF-β were significantly increased in A/CME compared with AME and, there was a fourfold increase in EGF in AME compared with A/CME (Fig. 2). These results demonstrated a different composition of growth factors in AME and A/CME.
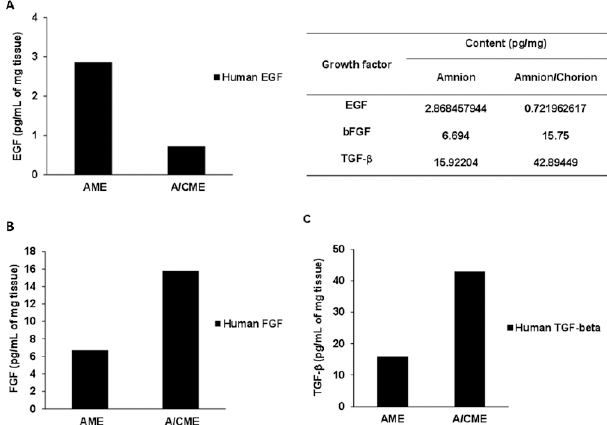
Contents of 3 representative growth factors in amnion and amnion/chorion membrane extracts (amnion membrane extracts [AME] vs. amnion/chorion membrane extracts [A/CME]). Relative amounts of growth factors in AME and A/CME extracts after tissue processing: (
ALP activity
To investigate early osteogenesis in MG-63 cells cultured in the presence or absence of AME and A/CME, ALP activity was determined at different time points (days 3, 7 and 10). Cells were cultured in OIM culture conditions with or without 100 µg/mL AME and A/CME for 10 days. Figure 3 shows ALP activity of cells cultured in OIM only (vehicle control), which gradually increased until day 10. At day 3, AME-treated cells showed slightly decreased ALP activity, but there were no statistically significant differences among vehicle-, AME- and A/CME-treated cells. Interestingly, ALP activity of A/CME-treated cells was significantly increased at day 7 (p<0.05) but decreased at day 10 compared with vehicle control cells. In contrast, AME-treated cells displayed lower ALP activity compared with both vehicle- and A/CME-treated cells during the culture period up to day 10, with a similar gradual increase in ALP activity compared with vehicle control cells (Fig. 3). These results showed clearly distinct patterns of ALP activity between vehicle- and A/CME-treated cells during early osteogenesis.
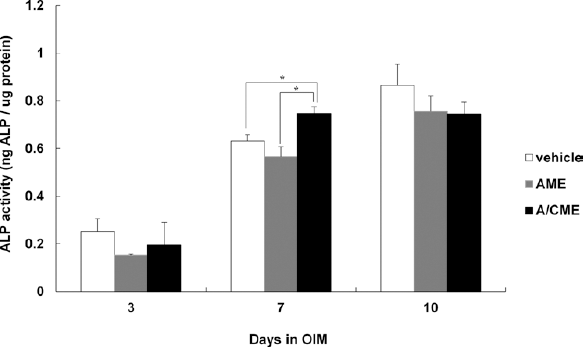
Effect of amnion membrane extracts (AME) and amnion/chorion membrane extracts (A/CME) on alkaline phosphatase (ALP) activity in MG-63 cells. ALP activity was measured in MG-63 cells cultured in osteogenic induction medium (OIM) with or without (vehicle) 100 µg/mL of AME and A/CME for 3, 7 and 10 days. The results showed different ALP activity in the presence of AME or A/CME, at day 7. The error bars indicate mean ± SD of multiple repeated experiments (*p<0.05).
Calcium content
For late osteogenic differentiation, MG-63 cells were cultured with or without 100 µg/mL AME and A/CME for 24 days under OIM culture conditions for osteogenic differentiation. After 24 days, calcium content was calculated per microgram of protein. As shown in Figure 4, A/CME-treated cells had significantly increased calcium (p<0.01) compared with untreated cells cultured with OIM. This result suggests that the 100 μg/mL of protein concentration in A/CME was sufficient to improve the mineralization of osteoblast-like cells. However, calcium deposition of AME-treated cells was still lower than vehicle control, consistent with the result from the ALP experiment (Fig. 3).
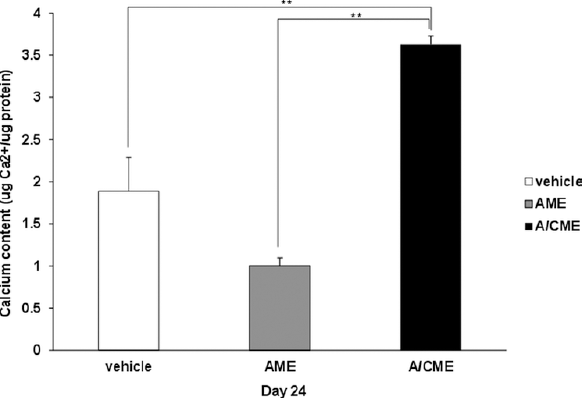
Effect of amnion membrane extracts (AME) and amnion/chorion membrane extracts (A/CME) on calcium deposition in MG-63 cells. Calcium contents were determined in MG-63 cells cultured in osteogenic induction medium (OIM) with or without 100 µg/mL of AME and A/CME for 24 days. The mineralization rate was measured by spectrophotometry using a phenol-sulphonephthalein dye as a substrate for free calcium. The relative amount of calcium was normalized to each microgram of protein. The error bars indicate mean ± SD of multiple repeated experiments (**p<0.01).
Osteogenic gene expression
Specific osteogenesis genes, including OCN, OPN, Runx2 and OSX, regulate the differentiation and function of osteoblasts (24). Therefore, we examined the mRNA expression levels of osteogenic genes in cells treated with or without AME and A/CME in OIM culture conditions.
Expression of OCN, OPN and OSX was evaluated by semiquantitative RT-PCR at day 24 after osteogenic induction. In the presence of A/CME (100 µg/mL), OCN and OPN expression was highly up-regulated (p<0.01) at the late differentiation stage of osteogenesis compared with the vehicle control. Moreover, AME-treated cells also increased OCN and OPN expression (p<0.01) after osteogenic induction for 24 days (Fig. 5A, B). Surprisingly, mRNA expression of another late osteogenic gene, OSX, was significantly down-regulated (p<0.05) in the presence of AME compared with vehicle- and A/CME-treated cells (Fig. 5C).
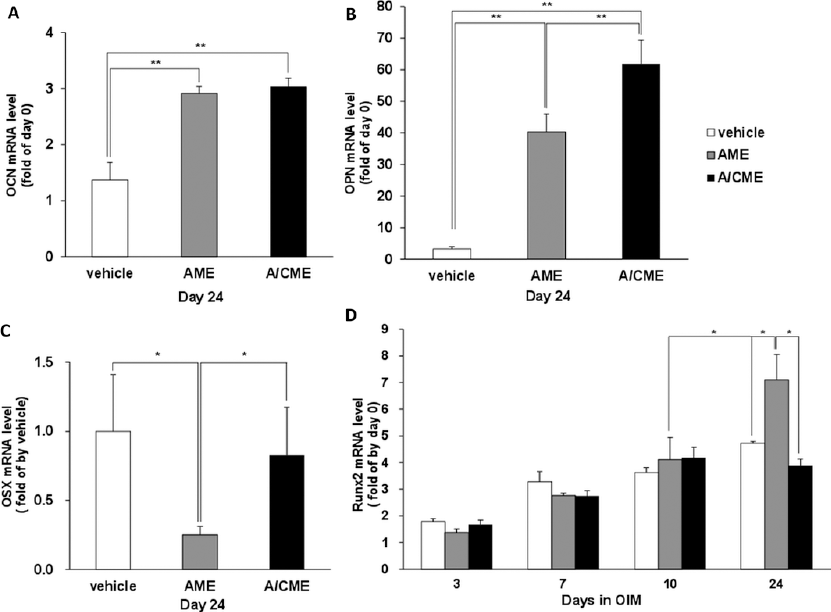
Effect of amnion membrane extracts (AME) and amnion/chorion membrane extracts (A/CME) on osteogenic marker genes expression in MG-63 cells. To determine relative expression levels of osteogenic genes, MG-63 cells were cultured in osteogenic induction medium (OIM) with or without (vehicle) 100 µg/mL of AME and A/CME. The mRNA levels of 3 genes for late differentiation of osteoblast-like cells: osteocalcin (OCN) (
Figure 5D shows the expression levels of Runx2 during osteogenesis of MG-63 cells. At 3, 7 and 10 days, no significant differences were observed among the 3 groups (vehicle-, AME- and A/CME-treated cells), and Runx2 expression gradually increased until day 10. Unexpectedly, Runx2 expression was not higher in A/CME-treated cells than in vehicle-treated cells at an early stage. However, Runx2 expression in vehicle- and A/CME-treated cells was similarly decreased at day 24, while expression in AME-treated cells was considerably increased compared with vehicle- and A/CME-treated cells (p<0.01). Taken together, these results clearly showed the different expression pattern of osteogenic genes by treatment with AME and A/CME during osteoblast differentiation.
Effect of A/CME on stimulation of mineralization
We investigated the stimulatory effect of A/CME on mineralization of osteoblast-like cells, when MG-63 cells were treated with increased extract concentrations. At day 7 after osteogenesis induction, mineralization was determined by performing a calcium assay (Fig. 6A). Dose-dependent deposition of calcium was observed in the A/CME-treated group; in particular, a high concentration of A/CME (400 and 800 μg/mL) significantly increased mineralization. Interestingly, a high concentration of AME (800 μg/mL) also increased calcium content compared with vehicle control (p<0.05), suggesting that extracts from human AM matrix promote osteogenesis, consistent with previous reports on a human acellular AM matrix (20).
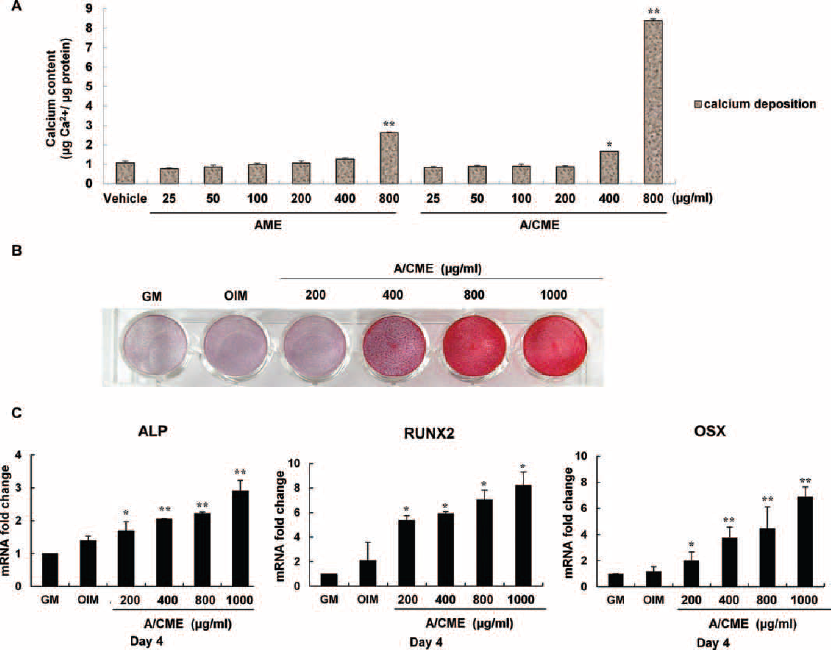
Stimulatory effect of amnion/chorion membrane extracts (A/CME) on osteogenesis in MG-63 cells. MG-63 cells were cultured in the presence or absence of amnion membrane extracts (AME) and A/CME under osteogenic induction medium (OIM). Indicated amount of AME and A/CME treated every 2-3 days with OIM and cultured for 7 or 9 days. The mineralization rate was confirmed by a calcium assay at day 7 (
As shown in Figure 6B, osteogenic differentiation of MG-63 cells was confirmed by alizarin red S staining at day 9 after osteogenic induction. The cell surface with a mineralized matrix clearly displayed a gradual color change in A/CME-treated cells from 400 to 1,000 μg/mL of concentration. In addition, the levels of ALP, Runx2 and OSX gene expression also gradually increased according to the concentration of A/CME, demonstrating the stimulatory effect of A/CME on osteogenesis of MG-63 cells (Fig. 6C). Unfortunately, a large amount of AME-treated cells subsequently changed morphology (data not shown) at day 7 after osteogenic differentiation and became detached from the bottom of the plate. From these findings, we found that exogenous A/CME promoted the differentiation and mineralization of MG-63 cells in a dose-dependent manner (Fig. 6).
Figure 7 shows results of the alizarin red S staining experiment to compare the osteogenic effect between pure growth factors and A/CME in MG-63 cells. Growth factors such as bFGF, TGF-β and EGF were treated alone or in combination with each other from 5 ng/mL to 500 ng/mL, and we were able to examine the increment of mineralization at the 500 ng/mL concentration of bFGF and EGF. However, the treatment of A/CME showed significant effects to promote the mineralization of MG-63 cells compared with the application of pure growth factors Interestingly, the combination of 2 or 3 growth factors, + TGF-β and bFGF + TGF-β + EGF groups, showed an inhibitory effect on mineralization of MG-63 cells rather than a stimulatory effect (Fig. 7).
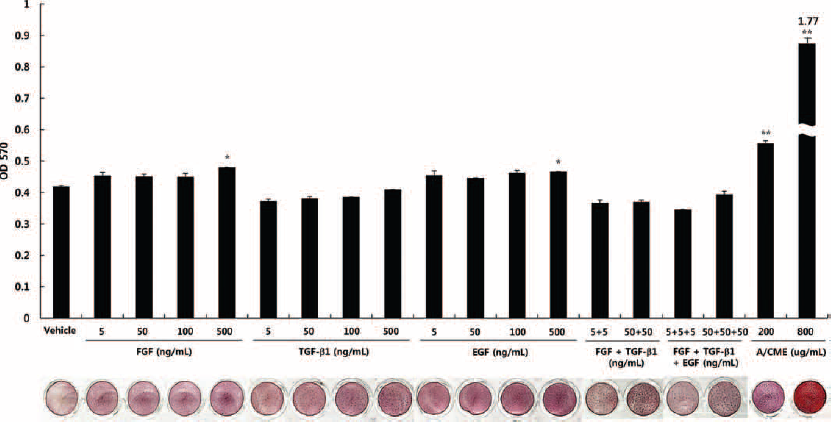
Comparison of mineralization between pure growth factors and amnion/chorion membrane extracts (A/CME) during osteogenesis in MG-63 cells. MG-63 cells were cultured in the presence or absence of bFGF, TGF-β, EGF or their combinations (FGF + TGF-β and FGF + TGF-β + EGF) and A/CME (200 and 800 µg/mL) under osteogenic induction medium (OIM). Indicated amount of growth factors and A/CME was treated every 2-3 days with OIM and cultured for 7 days. To examine the calcium deposition in extracellular matrix (ECM), alizarin red S staining was performed, and the absorbance value of solubilized alizarin red S measured at 570 nm. The error bars indicate mean ± SD of multiple repeated experiments (*p<0.05 vs. vehicle control; **p<0.01 vs. vehicle control). OD = optical density.
Discussion
The objective of our study was to apply human amniotic membranes for rapid bone regeneration more effectively than current approaches. Recently, Chen et al examined the scaffolding effect of a human acellular amniotic membrane matrix on osteogenic differentiation of human apical papillar cells (20). However, we focused on the potential role of numerous biochemical constituents of human amniotic membranes in osteoblastic differentiation, such as growth factors and cytokines, rather than the physical aspect of AM as a scaffold matrix. We demonstrated that A/CME displayed a significant capacity to accelerate the mineralization rate compared with AME, through an in vitro experiment using osteoblast-like MG-63 cells.
Figure 2 shows that AME and A/CME contained different amounts of growth factors, including EGF, bFGF and TGF-β, which influenced osteogenic differentiation (25-26-27). Osteogenesis is the process of bone tissue formation occurring by induction of osteoblast differentiation and function, which takes place in 2 major steps consisting of extracellular matrix (ECM) formation and calcification (mineralization). It is currently unclear how osteoblasts become differentiated, but the process is thought to involve growth factors and cytokines (28). Thus, we hypothesized that exogenous treatment of extracts derived from human amniotic membranes could provide signals that promote full osteogenic differentiation of MG-63 cells.
For ECM formation, ALP activity is known to increase following treatment with OIM until the activity peaks during 10-14 days and then subsequently decreases, which means that ECM mineralization has started to deposit calcium mineral in ECM. As shown by the ALP results, A/CME-treated cells had already reached early matrix maturation at day 7 after induction of osteogenic differentiation and subsequent initiation of the mineralization process. Indeed, it has been reported that the rapid expression of ALP drives ECM protein production followed by mineralization during bone development in vivo (29). Consequently, expression of osteogenic genes (OCN and OPN) and calcium deposition was also enhanced by treatment with A/CME at the late stage of differentiation, compared with vehicle- and AME-treated cells (Fig. 4), suggesting that 100 µg/mL of A/CME was sufficient to stimulate the differentiation and function of MG-63 cells. However, there was no significant increase in the mRNA levels of Runx2 and OSX within A/CME, suggesting that A/CME stimulates osteoblast differentiation via Runx2- and OSX-independent pathways. Conversely, treatment with 100 µg/mL AME was not sufficient for osteogenesis stimulation, as shown by decreasing ALP activity and calcium deposition (Figs. 3 and 4). Although expression of OCN and OPN increased with 100 µg/mL AME treatment after culturing with OIM for 24 days, increased mineralization was not observed (Figs. 4 and 5).
Additionally, we clearly observed different gene expression patterns of OSX and Runx2 at day 24 with 100 µg/mL of AME-treated cells in OIM. Runx2 is a transcription factor that is a key regulator of osteoblast differentiation and function. Runx2 regulates the expression of OCN, OPN, collagen type I alpha 1 (Col1 α1), bone sialoprotein (BSP) in osteoblasts (30). In a recent review, it was noted that Runx2 must be up-regulated at an early differentiation stage and then down-regulated at a late osteogenesis stage because it influences osteoblast differentiation from a positive to negative manner during osteoblastic differentiation (31). However, 100 µg/mL of AME induced high expression of Runx2 at a late osteogenesis stage in our experiment. OSX also plays an important role in bone formation and osteoblastic differentiation by up-regulation of BMP2. It has been known that OSX is controlled via a Runx2-dependent or Runx2-independent manner during osteogenesis (32). In this study, Runx2 expression was significantly induced at a late differentiation stage in the presence of AME (100 µg/mL), while OSX expression was decreased (Fig. 5). These results indicate that 100 µg/mL of AME induced high expression of Runx2, whereas expression of OSX was negatively regulated via a Runx2-independent mechanism at a late differentiation stage; this could result in suppression of mineralization in MG-63 cells under osteogenic-induced culture conditions. However, Figure 6 shows that a high concentration of AME (800 µg/mL) was capable of increasing calcium deposition in MG-63 cells. Therefore, these findings suggest that osteoblast differentiation could be promoted by AME concentration, but A/CME-treated cells showed significantly higher mineralization than did the same amount of AME-treated cells.
In Figure 7, our results show that a high amount of growth factors such as bFGF and EGF had small effect on stimulation of mineralization. Consistent with our findings, a previous report described the potentiating effects of growth factors induced transcription factors on osteogenic differentiation (28), supporting our results here. However, A/CME was more effective in stimulating the mineralization of MG-63 cells compared with the treatment of pure growth factor alone or in combination. These results might imply that the composition of growth factors or cytokines in A/CME provided an optimal environment for osteogenic differentiation of MG-63 cells.
López-Valladares et al reported that human amniotic membranes from donors of increased age and gestational age showed relatively lower levels of growth factors such as bFGF, HGF and TGF-β, indicating that the biochemical composition of human amniotic membranes is altered by age (33). More recently, Russo et al reported that the different preservation and storage processes of human amniotic membranes affects total protein and growth factor content (34). These 2 studies showed that variations in human amniotic membranes influence clinical results, as growth factors and cytokines have crucial roles in the regulation of cell fate. Therefore, further study of osteogenic differentiation using A/CME is needed to determine the exact conditions that promote mineralization, including TGF-β and bFGF content in extracts. In addition, further study is required to explore other possible biological constituents of human amniotic membranes that potentially control osteogenic differentiation. Future studies will assist in clarifying the conditions needed to establish an accurate standard amount of A/CME for in vivo application.
In conclusion, we demonstrated that A/CME promotes the differentiation of osteoblast-like cells. A/CME treatment in vitro resulted in rapid osteogenic differentiation. The regulatory mechanism associated with A/CME stimulation of osteoblast differentiation and mineralization remains to be elucidated. However, our current findings clearly show the potential of A/CME to stimulate osteogenesis. Further examination of gene expression profiles in the presence of A/CME during osteogenic differentiation will provide a greater understanding of the regulatory mechanisms underlying A/CME function in osteoblast differentiation. Additionally, 2 studies have reported scaffold surface modifications using physical and coating methods to improve adhesion strength and biocompatibility of MG-63 cells for bone tissue engineering (35, 36). Based on these studies, A/CME might be used as a novel therapeutic material with scaffold fabrication method for bone regeneration.
Footnotes
Financial support: This work was supported by a National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIP) (No. 2012M2A2A6035679).
Conflict of interest: None.
