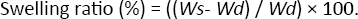Abstract
Purpose
This study reports on the synthesis and characterization of biodegradable dextran–allyl isocyanate–ethylamine (Dex-AE)/polyethylene glycol–diacrylate (PEGDA) hydrogels for the controlled release of gentamycin sulphate (GS) and in vitro inhibition of organisms.
Methods
The Dex-AE precursor was prepared through a 2-step chemical modification and characterized by Fourier transform infrared spectroscopy (FTIR).
Results
Scanning electron microscopy (SEM) results revealed that an increase in Dex-AE content led to an initial decrease in pore size of the Dex-AE/PEGDA hydrogels, but a further increase in Dex-AE content resulted in a slightly increase of pore size. The swelling data indicated that the swelling ratio depended on the precursor feed ratio. GS was incorporated into the hydrogels through 2 different methods, i.e., immersed and crosslinked. The crosslinked GS-Dex-AE/PEGDA hydorgels exhibited stronger antimicrobial activities against Escherichia coli, Staphylococcus aureus, and Pseudomonas aeruginosa. Finally, the viscoelastic properties of crosslinked GS-Dex-AE/PEGDA hydorgels were investigated.
Introduction
Burn infection constitutes a major worldwide threat to human health, which often causes systemic sepsis and severe septicemia (1, 2). It is reported that 75% of the morbidity in burn patients is related to infection (3). Pseudomonas aeruginosa and Staphylococcus aureus are of particular concern in burn infections (4). Currently, treatments of these infections include debridement, wound dressing and, if required, tissue-engineered skin (5). However, infected full-thickness burns are particularly difficult to repair. Thus, it is highly desirable to fabricate anti-infection tissue-engineered skin that can not only inhibit bacterial infection but also perform as a dermal regeneration template.
Hydrogel is defined as a polymeric network that swells to several times its original volume while remaining insoluble in aqueous solutions (6). Among the different tissue-engineered skins produced so far, hydrogels, which are structurally similar to the natural extracellular matrix (ECM), have the potential to direct cell migration, adhesion, and growth during tissue regeneration (7). Many kinds of hydrogel-based scaffolds have been designed and applied in wound healing. Beyond their utility as scaffolds, hydrogels have been used to deliver growth factors, antibiotics, and cells to induce complete skin regeneration (7-8-9-10-11).
Dextran, which consists of a-1,6-linked D-glucopyranose residues, is a bacterial-derived polysaccharide (12). Dextran is hydrophilic, biocompatible, and can be biodegraded by dextranase. From the structural point of view, only a fraction of hydroxyl groups have been substituted; more functional groups may be introduced onto the dextran backbone without altering its characteristics (13). Because of these properties, dextran-based hydrogels have been considered for cell immobilization and gene transfection and as drug carriers for a variety of pharmaceuticals. Dextran-based biomaterials have also been employed for skin tissue-engineering to promote tissue infiltration, neovascularization, and hydrogel degradation (7, 14-15-16). Result showed that customized dextran-based hydrogel alone, with no additional growth factors, cytokines, or cells, promoted remarkable neovascularization and skin regeneration. Although dextran-based hydrogels have been investigated for various purposes, their potential for in vitro anti-infection has not been fully explored.
Gentamycin sulphate (GS) has broad activity against both gram-negative and gram-positive bacteria, which has been used as a topical antibiotic in the treatment of superficial skin infections and sepsis (17). In our previous study, GS had been incorporated into SF scaffolds to treat infected full-thickness burns; the composite scaffolds inhibited infection and promoted dermal regeneration and re-epithelialization (18).
In this study, the PEG derivative (PEG–diacrylate, PEGDA) was chemically incorporated into a dextran derivative (dextran–allylisocyanate–ethylamine, Dex-AE) via photo-crosslinking to form biodegradable hydrogels. The chemical structure and morphology of the resulting hydrogels were examined. The swelling property of these hydrogels was studied as a function of the feed ratio of PEGDA to Dex-AE. GS was incorporated into these hydrogels and their in vitro antimicrobial activities against Escherichia coli, S. aureus and P. aeruginosa were evaluated. Finally, the viscoelastic properties of crosslinked GS-Dex-AE/PEGDA hydrogels were also investigated.
Materials and methods
Materials
Dextran (Dex) of MW 7000, allyl isocyanate (AI), Dimethyl sulfoxide (DMSO), acryloyl chloride, dibutyltin dilaurate (DBTDL), 2-bromoethylamine hydrobromide (BEAHB), GS were purchased from Sigma Chemical Company (St Louis, MO, USA). Dextran was dried in a vacuum oven for 24 h at 50°C before use. Triethylamine and isopropanol were purchased from the Guangzhou Chemical Reagent Company (Guangzhou, China). Polyethylene glycol (PEG) of MW 4000 was purchased from Aladdin Chemical Reagent Company (Shanghai, China). E. coli (ATCC8099), S. aureus (ATCC6538) and P. aeruginosa (ATCC9027) were obtained from the Department of Biomedical Engineering of Jinan University and were maintained on solid agar medium at 4°C. All other reagents were analytical grade and used without further processing.
Synthesis of dextran–allyl isocyanate–ethylamine (Dex-AE)
The synthesis of Dex-AE included 2 steps: the incorporation of AI followed by the incorporation of ethylamine. Dextran-based precursor (Dex-AI) was synthesized and characterized according to a method reported previously (15, 16, 19). In brief, dry dextran (2 g) was dissolved in anhydrous DMSO (24 mL) under dry nitrogen gas. DBTDL catalyst (0.73 mL) was injected into the solution dropwise, and then AI (1.09 mL) was added dropwise. The reaction mixture was stirred at room temperature (25°C) for 6 h. The resulting polymer was precipitated in cold excess isopropanol. The product was further purified by dissolution and precipitation in DMSO and isopropanol, respectively. This resulting Dex-AI was dried under vacuum for a week at 25°C and stored in a cold dark place before further use.
To synthesize Dex-AE, Dex-AI further reacted with BEAHB in the presence of triethylamine. A total of 2.0 g dry Dex-AI was dissolved in 20 mL anhydrous DMSO under dry nitrogen gas at 50°C. Triethylamine (11.2 mL) was injected into this solution. BEAHB (7.5 g) was dissolved in DMSO (10 mL) and then added to the above solution dropwise. This reaction solution was stirred at 50°C for 5 h. The reaction mixture was then filtered to remove the precipitated Et3NH4Br. The resulting Dex-AE polymer was obtained by precipitating the filtered solution into excess cold isopropyl alcohol. The product was further purified at least 3 times by dissolution and precipitation with DMSO and cold isopropyl alcohol, respectively. The final product was dried at room temperature under vacuum overnight before further use.
Synthesis of polyethylene glycol–diacrylate (PEGDA)
Polyethylene glycol–diacrylate (PEGDA) was synthesized according to previously described method (15, 16, 19). Briefly, PEG reacted with acryloyl chloride in the presence of triethylamine as a catalyst. Predried PEG (12 g) was dissolved in anhydrous benzene (150 mL) under nitrogen atmosphere at 40°C and then cooled to room temperature. Triethylamine (1.78 mL) and acryloyl chloride (1.22 mL) were subsequently added. The reaction mixture was stirred for 3 h at 80°C. The resulting polymer solution was filtered and precipitated. It was further purified 3 times by dissolution and precipitation with benzene and hexane, respectively. The PEGDA was then dialyzed (MWCO: 1000 Da) against distilled water for 4 days and then lyophilized.
Preparation of Dex-AE/PEGDA hydrogel
The Dex-AE/PEGDA hydrogels were prepared as previously reported (15, 16, 19). Different ratios of Dex-AE/PEGDA were dissolved into phosphate-buffered saline (PBS) solution containing 0.1% (wt∕wt) 2-methyl-1-(4-(hydroxyethoxy)phenyl)-2-methyl-1-propanone (Irgacure 2959, Ciba Speciality Chemicals, Tarrytown, NY, USA). The mixture was pipetted into a sterile mold (80 μL volume per well) to obtain discs measuring 8 mm in diameter by 2 mm in thickness and photopolymerized (approximately 10 mW∕cm2 of UV light for 10 min) under UVP (Blak-Ray, Upland, CA, USA). The resulting hydrogels were washed in distilled water for 48 h to remove unreacted precursors before further characterization. Immersed GS-Dex-AE/PEGDA hydrogels were fabricated by immersed Dex-AE/PEGDA into GS solution. Crosslinked GS-Dex-AE/PEGDA were prepared by dissolved GS, Dex-AE/PEGDA into PBS solution containing Irgacure 2959 and then photopolymerized under UVP.
Fourier transform infrared spectroscopy (FTIR)
The dextran, dextran-AE, PEG, and PEG-DA were all characterized for their chemical structure by FTIR. For FTIR characterization, all samples were dried in a vacuum oven for at least 24 h and then compressed into pellets with KBr powder (1/10, w/w) for FTIR characterization (Vertex 70; Bruker, Billerica, MA, USA). The IR spectra in the absorbance mode were recorded using a diamond crystal plate and obtained in the spectral region 400 cm−1 to 4000 cm−1 with a resolution of 4 cm−1 and 20 scans per sample.
Scanning electron microscopy (SEM)
The interior morphology of the Dex-AE/PEGDA hydrogels at room temperature was studied using a scanning electron microscope (SEM; LEO1530 VP, Philips, Amsterdam, The Netherlands). The pore sizes of the scaffolds were evaluated by measurement of 25 random pores in SEM images of the same sample using the ImageJ software (NIH, Bethesda, MD, USA).
Swelling study of Dex-AE/PEGDA hydrogels
The swelling property of the Dex-AE/PEGDA hydrogels was gravimetrically determined. Dry hydrogel specimens of known weight (Wd) were immersed in distilled water at room temperature (25°C). The swollen hydrogels were removed from water at predetermined intervals and weighed (Ws) after wiping off excess water on the surface with a wet filter paper. The swelling ratio was then calculated according to the formula below:
In vitro biocompatibility of Dex/PEGDA hydrogels
Cell proliferation was detected by using a Cell Counting Kit-8 (Beyotime, Jiangsu, China) according to the manufacturer's instructions. Briefly, NIH 3T3 fibroblasts (2000 per well), suspended in complete Dulbecco's modified Eagle's medium (DMEM) containing 10% bovine calf serum and 1% streptomycin and penicillin, were seeded (100 µL per well) on 96-well microculture plates and allowed to culture in the presence of macromer solution (10 μL/mL culture medium) for 1, 3, 5, and 7 days (conditions: 37°C; 5% CO2; 24 h). The growth medium was changed every 2 days. The optical density of the solutions was recorded using a microplate reader at a wavelength of 570 nm.
Antibacterial activity
The antimicrobial activity of the immersed and crosslinked GS-Dex-AE/PEGDA hydorgels were investigated using well-diffusion method against S. aureus (G+), E. coli (G-) and P. aeruginosa (G-) (20). The two kinds of hydrogels, which were sterilized under UV light in a laminar flow bench for 24 h, were placed in a Petri dish containing agar upon which S. aureus (G+), E. coli (G-), and P. aeruginosa (G-) had been cultured. After incubation for 24 h (37°C), the antimicrobial activity was evaluated by measuring the diameter of inhibition zone against test microorganisms.
Statistical Analysis
Data are expressed as the means of at least 3 replicates ± standard deviations (SD). Statistical comparisons were performed using ANOVA (t test). All statistical computations were performed using the SPSS software (ver. 16.0; SPSS, Chicago, IL, USA). Differences with a value of P<0.05 were considered statistically significant.
Results and discussion
Dex-AE/PEGDA hydrogel preparation
Figure 1 shows the FT-IR spectra of dextran, Dex-AE, PEG, and PEGDA. Compared with the spectrum of dextran (A), the FTIR spectra of Dex-AE (B) show the typical doublebond absorption bands at 1645 cm−1, and blown-up version of peak 1 clearly shows the C = C stretch vibration in Dex-AE. This confirmed that the C = C double bonds were successfully incorporated into the dextran. Both the C–H stretch and bending vibrations are weak and overlapped with Dex- AE (B) and PEGDA (C), thus they could not be used to identify the consumption of C = C bonds during gelation process. Amine groups from Dex-AE have –NH– bands showed that amide II at 1532 cm cm−1 and 3343 cm−1 due to the N–H stretching vibration of the amine group. The 1650 cm−1 band in the dextran spectrum (A) was due to the presence of trace amounts of adsorbed water that was difficult to remove in dextran (21).
FTIR spectra of
Morphology and swelling
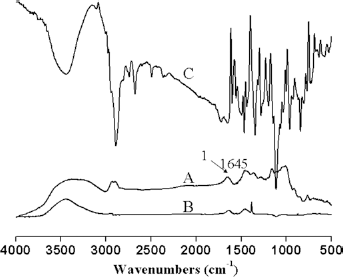
The interior morphology of these freeze-dried, swollen hydrogel samples as a function of the precursor feed ratio is shown in Figure 2. The swollen hydrogel network became denser and more compact as the Dex-AE content increased (from 10 to 30%). Evidenced by the increased pore size, however, a further increase in the Dex- AE content (i.e., 50 and 70%) led to a more open and looser network structure. Table I shows the change of average hydrogel pore size as a function of the Dex-AE content. Dex-AE content from 10% to 30%, the pore size decreased from around 23 μm to 18 μm, and the network become denser and more compact, which could be attributed to a higher level of crosslinking from the increasingly available crosslinkable unit (AI) for photo-crosslinking (19). As the Dex-AE content increased beyond 30% (by weight), the hydrogel network became more open, i.e., the pore size became bigger. This phenomenon could be attributed to the incomplete crosslinking in 50% and 70% Dex-AE content hydrogels. Similar results have been reported (19).
SEM micrographs of Dex-AE/PEGDA hydrogels. (
Pore size of Dex-AE/PEGDA hydrogels as a function of Dex-AE content

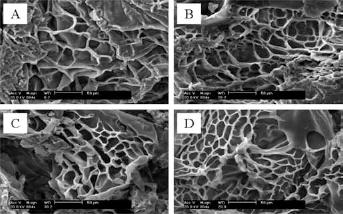
Figure 3 shows the effect of Dex-AE to PEGDA feed ratio on the swelling ratios of Dex-AE/PEGDA hydrogels at room temperature. The data show that an initial increase in Dex-AE content (from 0 to 10%wt) led to a reduced swelling of Dex-AE/PEGDA hydrogels. When the Dex-AE content increased from 10% to 30%, it led to a slight increase in swelling of Dex-AE/PEGDA hydrogels, but a minimum swelling between 10% and 30% Dex-AE content was reached. A further increase in Dex-AE content (from 30 to 50%), however, resulted in a decrease in swelling. Furthermore, when the content of Dex-AE increased to 70%, the swelling visibly increased. For instance, the swelling ratio increased 22% from around 900% to 1100% when the Dex-AE content increased from 10% to 30%, and then increased 44% (to 1300%) as the Dex- AE content reached 70%, which is believed to be attributed to presence of the distinctive, more open and large-pore network structure. The dependence of the swelling ratio of Dex-AE/PEGDA hydrogels on Dex-AE content is in good accordance with the morphological images (Fig. 2) and average pore size data (Tab. I).
Swelling ratio of Dex-AE/PEGDA hydrogels at room temperature.
In vitro biocompatibility
It is important to assess the biocompatibility of the modified dextran macromers for their future biomedical applications. As we know, the precursor molecules cannot completely react to form hydrogels during photocrosslinking. The unreacted macromers will be released during hydrogel swelling, which may result in cell toxicity. Therefore, we examined the biocompatibility by culture NIH 3T3 fibroblasts in growth medium containing macromer solution with the highest dextran content (i.e., a 70/30 Dex-AE/ PEGDA ratio). As demonstrated in Figure 4, the rate of cell proliferation in medium containing Dex- AE/PEGDA and PEGDA was indistinguishable from that in control mediums. The results were in accordance with previous research (15).
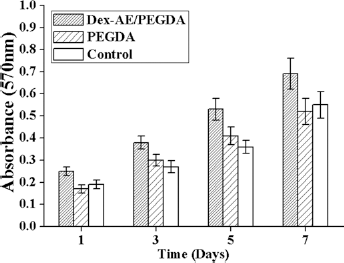
3T3 cell viability on different Dex-based hydrogels as a function of culture time.
Antimicrobial activity
The antimicrobial activity of immersed and crosslinked GS-Dex-AE/PEGDA hydorgels was tested using the well-diffusion method against S. aureus, E. coli and P. aeruginosa; the results are shown in Figure 5. The diameter of inhibition zones of 2 different methods of GS loaded hydrogels was found to be more or less similar, and hence, confirm the integrity of antimicrobial activity of GS in Dex-based hydrogels. However, the diameter of crosslinked GS-Dex-AE/PEGDA hydrogels was always larger than immersed GS-Dex-AE/PEGDA hydrogels, which indicated controlled and sustained antibiotic release profiles. It is known that GS has broad activity against both gram-negative and gram-positive bacteria, and has been used as a topical antibiotic in the treatment of superficial skin infections (17). In our previous study, GS had been incorporated into SF scaffolds to treat infected full-thickness burns, results showed that the composite scaffolds inhibited infection, and promoted dermal regeneration (18).
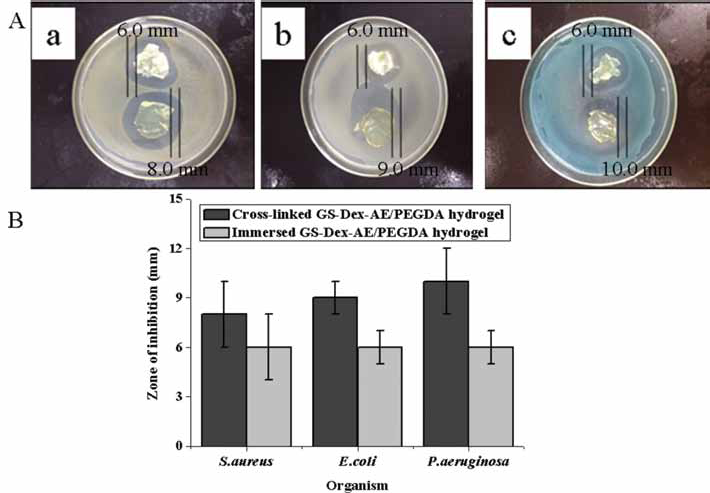
Antimicrobial activity (
Mechanical properties of crosslinked GS-Dex-AE/PEGDA hydrogels
The viscoelastic parameters were measured as a function of time at a constant frequency of 1.0 rad/s. To ensure the rheological measurements within linear viscoelastic regions, strain sweep tests were carried out prior to the time as well as frequency sweep analyses. Figure 6A gives typical strain amplitude sweep curves, in which the changes of storage modulus (G’) and loss modulus (G″) with strain amplitude were recorded. As seen, the G’ and G″ remained almost constant and then suddenly changed when the strain increased from 0.1% to 100%. At the very beginning, with G’ lower than G″, the system behaved like a viscous fluid. As gelation continued, G’ elevated quickly due to the crosslinking. A sudden decrease of the G’ and a sudden increase of the G″ indicated the bond breakage within the hydrogel network and a transition from linear to nonlinear behavior (22, 23). In both moduli, the build-up rate of G0 was much higher than that of G00. In this case, the strain of 1.0% was determined for linear viscoelastic measurements. To understand the properties of the crosslinked GS-Dex-AE/PEGDA hydorgels, frequency sweep analyses were conducted at 25°C. The frequency applied to the hydrogel sample increased from 0.1 Hz to 100 Hz and the results are shown in Figure 6B. The G’ value was larger than the G″ value, indicating that the system was elastic. From 0.1 Hz to 10 Hz, the changes in G’ and G″ were slight, which indicated that the crosslinked GS-Dex-AE/PEGDA hydrogels were stable.
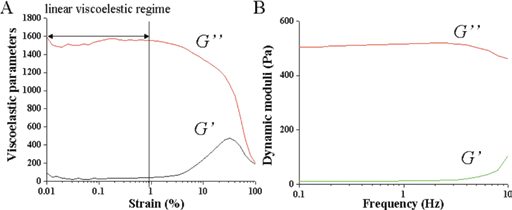
Typical strain amplitude sweep curves (
Conclusions
Biodegradable dextran–allyl isocyanate–ethylamine (Dex-AE)/polyethylene glycol–diacrylate (PEGDA) hydrogels for the controlled release of gentamycin sulphate (GS) were prepared. The crosslinked GS-Dex-AE/PEGDA hydorgels exhibited stronger antimicrobial activities against E. coli, S. aureus, and P. aeruginosa. The mechanical properties of the crosslinked GS-Dex-AE/PEGDA hydrogels were stable. These results indicate the potential for using such composite hydrogels for the controlled release of antibiotics for drug delivery or for dressing material for quick healing of wounds and burns, and other related applications.
Footnotes
Financial support: This study was supported financially by the Natural Science Foundation of China (51303064), Natural Science Foundation of Guangdong (S2012040008003), the Key Project of DEGP (cxzd1109), the Ph.D. Programs Foundation of the Ministry of Education of China (20124401120015), and the Fundamental Research Funds of the Central Universities (21612327).
Conflict of interest: The authors declare that there is no conflict of interest.