Abstract
Background
In this work, chitosan (CH) was used to produce a novel coating for Ti6Al4V, the most widely used alloy in orthopedic implants, so as to improve the biological tissue response at the metallic surface.
Methods
The Ti6Al4V surface was sandblasted with alumina particles. CH was chemically modified, via carbodiimide chemistry, using lactobionic and 4-azidebenzoic acid to make it soluble at physiological pH and photocrosslinkable, respectively. The reaction was verified by FTIR, NMR and UV/vis spectroscopy. Ti6Al4V surfaces were coated with solutions of the modified CH and exposed to UV light, causing polymer crosslinking and formation of a hydrogel on the surface. The crosslinking reaction was monitored by FTIR at different exposure times. Coating morphology was observed by SEM. The coating's cytocompatibility was determined in vitro through the culture of rat bone marrow mesenchymal stem cells, using an MTT assay, with their morphology assessed by SEM.
Results
The developed coating behaved as a hydrogel on the Ti6Al4V and was stable on the surface. FTIR and NMR confirmed the crosslinking mechanism, based on an arile ring expansion, and subsequent reaction with the CH amine groups. Furthermore, the coating was able to support cell proliferation and osteogenic differentiation.
Conclusions
UV crosslinking of CH is easy to apply and has potential for future metallic implant surface modifications. Due to its nature as a hydrogel, the coating could be used for further studies in the encapsulation of bioactive molecules to improve osteogenic potential at the tissue–implant interface.
Introduction
Titanium and its alloys have been some of the most widely used materials in the fabrication of prosthesis and implants (1, 2). However, 2 limitations are encountered when using these materials: the first is the lack of bioactivity, and the second, the susceptibility of the surface oxides to bacterial colonization (3, 4). Approximately, 52.5% of dental implant failures are due to loosening and/or bacterial infections (5-6-7). Different strategies have therefore been developed to overcome these limitations.
Calcium phosphate coatings, especially hydroxyapatite, represent the most common surface modification technique (8-9-10-11-12); nevertheless, they do not offer an appropriate guard against postsurgical infections. The administration of presurgical antibiotic anaphylaxis can result in renal and hepatic complications, in addition to imminent propagation of antibiotic resistant microorganisms (2, 13).
Polymeric coatings with intrinsic antibacterial characteristics have taken on special interest. Especially, chitosan (CH) and its derivatives have been shown to have a great capacity to reduce the presence of some of the most common pathogens in bone surgeries (14-15-16), and support the proliferation of different cell phenotypes (17). Furthermore, due to the availability of different functional groups (amines and hydroxyls) in its chain, CH can be chemically modified to adjust its properties to different applications in the forms of 3D structures (17, 18).
Numerous CH-based coatings have been deposited via different techniques on top of metallic substrates, including ionic assembling and covalent unions. Some studies have assessed the use of hydrogel coatings over metallic surfaces. Ma et al developed CH–gelatin hydrogel coatings on titanium surfaces via electrophoretic deposition, which supported MC3T3-E1 cell attachment, proliferation and migration as well as significant new bone formation in vivo (19). Ding et al reported a potential hydrogel coating derived from functionalized CH with high antibacterial activity for both Escherichia coli and Staphylococcus aureus, the major pathogens encountered in orthopedic implants (20). However, CH coatings always require previous chemical treatment of metallic surfaces to ensure the attachment of the polymer, which limits their clinical implementation. Here we proposed a novel photocrosslinking coating method for Ti6Al4V implants without any pretreatment of its surface. This coating is based on physical interactions, and the mechanical anchoring produced by the photocrosslinking reaction of a functionalized CH with proven cytocompatibility (21). A short UV light exposition produces a hydrogel coating that possesses great potential for orthopedic applications. Moreover, to the best of our knowledge, this is the first study reporting a coating system that allows in situ deposition during surgery.
Materials and methods
Photocrosslinkable CH synthesis
Low-molecular-weight CH with 80% deacetylation and an average molecular weight of 4.3 x 105 Da, 4-azidobenzoic acid (AZ), N, N, N0, N0-tetramethylethylenediamine (TEMED), lactobionic acid (LA) and 1-ethyl-3-(3-dimethylaminopropyl)-carbodiimide (EDC) were obtained from Sigma-Aldrich. All reagents and solvents were of the highest purity commercially available.
Photocrosslinkable CH (LA-CH-AZ) was prepared based on a chemical modification proposed by Ono et al (21). Azide and lactose moieties were introduced to the CH molecules through condensation reactions (pH 5) via carbodiimide chemistry, using EDC, with the amine groups from CH. Introduction of lactose and azide yields a water-soluble CH at neutral pH and photocrosslinkable hydrogel, respectively. Using this procedure, it has been estimated that approximately 2.5% and 2.0% of the amino groups in the CH are substituted by the azide (as provided by AZ) and lactide (as provided by LA), respectively (21).
Characterization of LA-CH-AZ (FTIR, NMR, UV/vis)
The introduction of AZ and LA fractions in the CH molecular structure was corroborated by Fourier-transformed infrared spectroscopy (FTIR), using a FT-IR 4100 series spectrometer (Jasco Corp., Japan), and nuclear magnetic resonance spectroscopy (NMR) analyses of the lyophilized LA-CH-AZ. Samples for FTIR were prepared by taking 50 mg of the lyophilized product and mixing it with sufficient potassium bromide. NMR spectrums of LA-CH-AZ were obtained with a AL-400 NMR spectrometer (Jeol Ltd., Japan), using a proton NMR modality (H NMR), for which the LA-CH-AZ dissolution was carried out in deuterium at a concentration of 10 mg/mL. Additionally, a solution (1 mg/mL) of CH-AZ was exposed to UV light (254 nm, at 90° and 6 cm from the source) for 5 minutes to guarantee that the reaction took place in its entirety. The resulting hydrogel was analyzed in a UV/vis/NIR Jasco V-570 spectrophotometer at different wavelengths in a range of 200 nm to 500 nm, using water in the reference cell.
Gelation time of the LA-CH-AZ solution
The lyophilized LA-CH-AZ product was dissolved in Milli-Q water at a final concentration of 10 mg/mL. Thirty microliter aliquots were placed on a polystyrene plate, in a UV crosslinker (Hoefer, UVC500), at 2 cm from the UV light source, and were irradiated at 254 nm and 4 W for 30, 90 and 180 seconds. The gelation time was evaluated through FTIR analysis of the lyophilized products and of the UV light-exposed samples.
LA-CH-AZ hydrogel swelling
Aliquots (100 μL) of a LA-CH-AZ solution (10 mg/mL) were placed in Petri dishes, and irradiated for 90 seconds with UV light, using the aforementioned conditions. The formed hydrogels were dehydrated overnight under vacuum and later weighed. Subsequently, the dried samples were rehydrated in phosphate-buffered saline (PBS; pH 7.4) at 37°C, and weighed at different intervals. The hydrogel swelling ratio (SW) was then calculated using the following equation:
Where Wd and Ws are the weights of the dry and swollen samples, respectively.
Surface preparation of Ti6Al4V samples
Ti6Al4V samples were obtained through sectioning into discs the cylindrical portion of the femoral stem of a commercial hip prosthesis (Corail; Johnson & Johnson), using a diamond saw LECO VC-50. The discs measured 10 mm in diameter and 1 mm in width. The samples were surface treated through sandblasting using alumina particles as previously described (22), using the conditions summarized in Table I.
Parameters of the sandblasting process

After sandblasting, the discs were examined superficially by scanning electron microscopy (SEM; Jeol, JSM-639). Mean surface roughness of the samples, as well as that of the prosthesis outer surface, was measured through a surface roughness tester (Mitutoyo SJ-210).
Contact angle measurement
To assess the wettability of both the unmodified CH and LA-CH-AZ solutions of equal concentration (10 mg/mL) on the Ti6Al4V surface, contact angle was measured by the sessile drop method, using a contact angle analyzer (Phoenix 150; Surface Electro Optics). Five measurements were performed for each solution. The resulting value of each single measurement was the average value of left and right contact angles.
LA-CH-AZ coating deposition on Ti6Al4V samples
Thirty microliter aliquots of LA-CH-AZ solutions (10 mg/mL and 5 mg/mL), previously determined to be a volume sufficient to cover the entire disc surface, were added on the sandblasted metallic discs, allowing polymer adsorption for 20 minutes. Subsequently, the surfaces were washed with Milli-Q water and irradiated with UV light, for 90 seconds, using the conditions previously established.
Coated surface characterization by SEM
LA-QT-AZ-coated Ti6Al4V samples were evaluated morphologically by SEM. Representative micrographs were taken at different immersion times in PBS at 37°C. In every case, samples were previously dried and sputter coated with gold for their visualization.
Cytocompatibility and cell proliferation using mesenchymal stem cells
Mesenchymal stem cells (MSC) were previously obtained from male Sprague Dawley rats of approximately 8 weeks of age, as reported by Alvarez-Barreto et al (23). Cells from the fourth passage were used. Prior to cell seeding, coated samples were prepared under sterile conditions in a laminar flow chamber, and placed in 24-well plates. Aliquots of cell suspension were placed on top of the samples at 2 x 104 cells/cm2. The wells were partially filled with Dulbecco's modified essential medium (DMEM; Life Technologies), just enough to cover the bottom surface so as to keep a humidified environment during the time allowed for cell adhesion, which was 6 hours at 37°C, 5% CO2 and 95% relative humidity. Wells were then filled with osteogenic medium (DMEM supplemented with 10% fetal bovine serum, 1% antibiotics-antimycotics, 10−8 dexamethasone, 0.05 mM ascorbic acid and 10 mM β-glycerophosphate) to cover the cells, and they were then incubated for 10 and 16 days with periodic changes of medium.
MSC viability and proliferation was evaluated with an MTT assay (MTT; Sigma) (24). Briefly, at each time point, the medium was discarded, then the samples were rinsed with PBS and incubated in the presence of an MTT solution in PBS (200 mg/mL) for 2 hours at 37°C, 5% CO2 and 95% relative humidity. The MTT solution was later discarded, and the formation of purple formazan crystals was carried out by adding 250 µL of dimethyl sulfoxide. Absorbance was measured using an ELISA plate reader (TECAN infinite M200) at a wavelength of 570 nm. The metabolic activity is directly proportional to the absorbance. Morphology of the cells on the surfaces was evaluated through SEM, and an X-ray diffusion scattering (XDS) analysis was performed on selected areas.
Statistical Analysis
Mean and standard deviation were calculated. A 2-way analysis of variance (ANOVA) was also applied, and multiple pairwise comparisons were carried out using the Tukey-HSD method with a 95% confidence level (p<0.05).
Results
Chemical modification of CH
The process of chemical modification of CH was initiated with the incorporation of LA in the presence of EDC and TEMED. The resulting product (CH-LA) was dissolved in distilled water, demonstrating its solubility at neutral pH and suggesting that the first stage of modification was successfully carried out. After that, AZ was introduced in a similar reaction, yielding LA-CH-AZ. Figure 1a shows a representative image of the contact angle measurements, indicating a decrease in θ due to the incorporation of LA and AZ in the CH structure. The values of θ were 98.3° ± 0.7° and 67.5° ± 0.9° before and after CH modification, respectively.
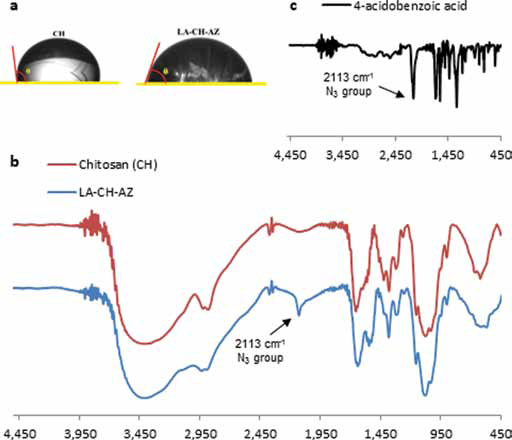
Figure 1b shows the FTIR spectra of CH, and the products of each of the modification stages: LA-CH and LA-CH-AZ. The CH FTIR spectrum shows the characteristic absorbance values of the polysaccharide, at 1,687 cm−1 (Amide I), 1,581 cm−1 (-NH2 bending) and 1,370 cm−1 (Amide III). Additionally, bands at 1,186 cm−1 (asymmetric stretching of the C-O-C bridge), 1,020 cm−1 and 1,010 cm−1 (skeletal vibration involving C-O stretching) are inherent to its saccharide structure (25).
Comparing the CH and LA-CH-AZ spectra, slight shifts were observed in the previously mentioned peaks due to intermolecular configurational changes that took place after the incorporation of LA and AZ through carbodiimide chemistry. Particularly, a strong absorption signal appeared at 2,113 cm−1 corresponding to the N3 group in the AZ covalently linked to CH, as indicated in Figure 1c.
The conjugation of the crosslinking agent (AZ) was confirmed through the H NMR spectrum displayed in Figure 2. The HAc signal (1.97 parts per million [ppm]) refers to protons of the acetamide group (CH3) of the acetylated monomeric residue. It is important to point out that CH is not completely deacetylated; the singlet observed at 2.92 ppm (H2D) corresponds to the D-glucosamine fraction; between 3.39 ppm and 4.01 ppm a triplet is seen, attributed to H2-H6 protons for both fractions D-glucosamine and N-acetylglucosamine. A doublet is evident between 4.50 ppm and 4.90 ppm due to the amine hydrogens. The mentioned signals correspond to the unmodified CH, while the signals at 7.77 ppm and 7.15 ppm represent hydrogens in the azidobenzamide group (26, 27), thereby confirming the modification via azide group incorporation into the CH molecule.
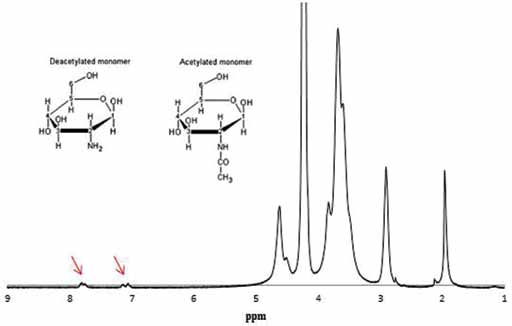
AZ-modified chitosan H-NMR spectrum. Arrows indicate signals originating from protons of azidobenzamide group. Top left corner: monomers conforming the CH molecule, both acetylated (Ac) and deacetylated (D), with their respective numerations that refer to the H NMR spectrum (28).
From the spectrum shown in Figure 2, it was possible to calculate the percentage of CH amine converted to 4-azidobenzamide by comparing an integrated area of the peaks at 7-8 ppm (4H, benzene) and those at 3-4 ppm (6H, CH). NMR spectra indicated that the absolute number of aryl azide groups in the system that can crosslink with other amine groups in CH was 1.5%. Moreover, the UV spectrum (Fig. 3) also confirmed the incorporation of azide in the CH structure, displaying an absorbance peak at 273 nm, typical of 4-azidobenzamide.
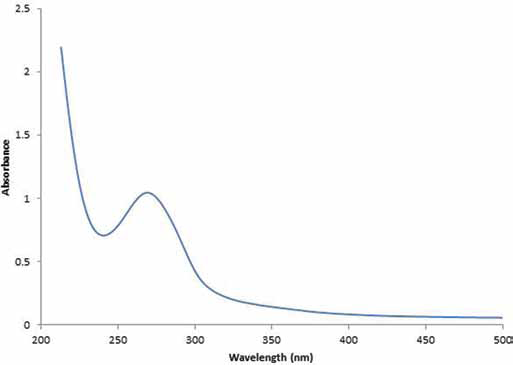
UV/vis absorbance spectrum of a photocrosslinked LA-CH-AZ hydrogel. ppm = parts per million.
Gelation time
The optimum UV light exposure time to produce photocrosslinking of the LA-CH-AZ solutions was determined through the analysis of the signal at 2,113 cm−1 (N3 of the AZ covalently linked to CH) in the FTIR spectrum. Figure 4 shows the tendency of this signal to diminish at longer exposure times, indicating that the reaction has completely taken place at 90 seconds, due to the signal suppression at this time.
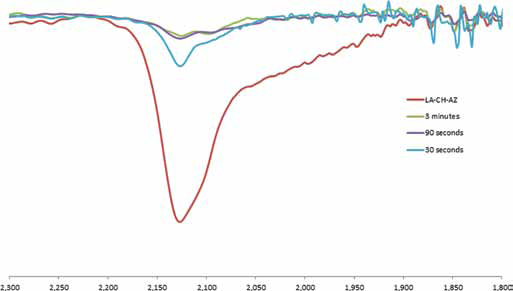
FTIR spectra of AZ-CH solution at different exposure times to UV light (0, 30, 90 seconds and 3 minutes), showing N3 signal decrease.
LA-CH-AZ hydrogel swelling behavior
Figure 5 describes the LA-CH-AZ hydrogel's water absorbing behavior as a function of the immersion time. The curve shows that, during the first 60 minutes, there was a tendency for rapid hydrogel swelling, reaching absorption levels of up to 10 times the hydrogel's dry weight. Equilibrium was achieved after 4 hours with a swelling ratio of approximately 8.47, which was maintained after 24 and 48 hours of immersion (results not shown).
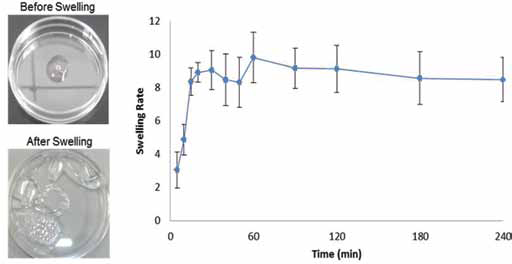
Swelling behavior of photocrosslinked LA-CH-AZ hydrogel in phosphate-buffered saline (PBS) as a function of the immersion time. Left: pictures of the hydrogel before and after swelling.
Metal surface treatment
Ti6Al4V samples treated superficially by the sandblasting technique reached a medium roughness that oscillated between 3.05 ± 0.03 µm and 3.97 ± 0.04 µm. On the other hand, the medium roughness of the commercial prosthesis was 4.1 ± 0.9 µm. The sandblasted discs were evaluated by SEM, and no alumina particles entrapped in metallic surfaces were found.
LA-CH-AZ deposition on Ti6Al4V samples
Morphological analysis of the crosslinked LA-CH-AZ coatings was performed through SEM. Micrographs of coated Ti6Al4V samples were taken before and after aqueous medium immersion. Valleys or cracks resulting from polymer retraction during photocrosslinking appeared as dark lines along the coating before aqueous immersion, as shown in Figure 6, suggesting nonhomogeneous coating thickness.

SEM micrographs before liquid immersion of coated Ti6Al4V by photocrosslinking from
Figure 7 shows SEM images at various magnifications of the samples incubated in PBS for 4 and 16 days. A change in the coating morphology was evident, with respect to the micrographs observed in Figure 6, where the samples had not been immersed in the aqueous medium. This new morphology is characteristic of the swelling process that hydrogels undergo when absorbing significant amounts of water. It was also observed that coating degradation took place at longer immersion times. At ×500 magnification, it was possible to capture the entire area, displaying LA-CH-AZ conglomerates in multiple simple zones after 4 days of immersion, while at 16 days; there was greater dissociation of the hydrogel (Fig. 6a and b, respectively). Thus, the area occupied by the conglomerates was smaller, and at that magnification, a complete coating coverage of the metal surface could not be observed. Instead, specific covered sites were irregularly distributed throughout the surface, with a distinct morphology for both naked substrate and coated samples before immersion.

SEM micrographs of Ti6Al4V coated with LA-CH-AZ solution (10 mg/mL) by photocrosslinking after liquid immersion for 4 days
Cell proliferation using MSCs
An MTT assay was carried out in both naked and coated surfaces to compare cell proliferation under different conditions. Figure 8 illustrates the absorbance due to the dissolved formazan crystals on samples after 4, 10 and 16 days of culture. At day 4, there were no significant differences among the different substrates. At day 10, naked samples had greater absorbance than coated samples. However, at day 16, the proliferation rates on LA-CH-AZ coatings were higher than on the naked Ti6Al4V.
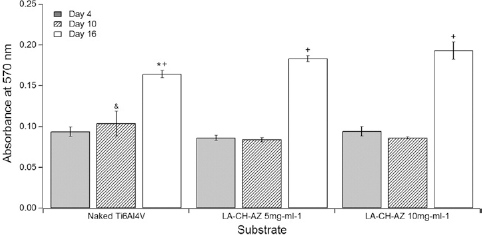
Cell proliferation by MTT on the naked substrate (naked Ti6Al4V) and coated metal surface (LA-CH-AZ 5 mg/mL and 10 mg/mL). *,& represent the lowest and highest absorbance value at a given time, respectively. +represents the day with the highest value at a given substrate. p<0.05.
Figure 9a shows the cell morphology on coated surfaces, with cells spread throughout the surface, and with numerous filopodia (arrows). It can also be observed that there are nodules at the extremes of some filopodia (square). When an XDS analysis was performed on the nodules, it was found that the main components of these nodules were calcium and phosphorous (Fig. 9B).

Discussion
The present work proposed the use of a photocrosslinkable polymeric solution to create a coating with intrinsic antibacterial properties, easy to apply and with the possibility of encapsulating growth and differentiation factors that could potentiate implants’ osseointegration.
The first stage of the study involved the incorporation of LA and AZ in the CH molecule – a carbodiimide condensation reaction that was corroborated by FTIR and NMR. Additionally, the success of the chemical modification was clear after the complete dissolution of the resulting product (LA-CH-AZ) in water and PBS and its gelation when irradiated with UV light.
Ono et al (21), who first developed the modification of CH with LA and AZ, explained that the photocrosslinking of the AZ-modified CH occurs through the formation of “azo” links (-N = N-) when the solution is exposed to UV light. Nonetheless, in the UV/vis spectrum of the modified CH solution, prior to irradiation (Fig. 2), there was no absorbance signal characteristic for the azo groups, which is manifested at wavelengths near 353 nm (29). The absence of a signal at 353 nm suggests that the mechanism proposed by Ono et al (21) and other authors (30, 31) may not have been the predominant one. The spectrum obtained in this study coincides with that found by Rickett et al (26), who suggested that the UV light produces the liberation of nitrogen and the photolytic conversion of the arile azide to a reactive nitrogen. Both nitrenes undergo a ring expansion and react with the nearby amines in the CH chain to form inter- and intra-molecular networks. In this work, it was considered that the mechanism governing the LA-CH-AZ crosslinking was the latter.
Under the conditions used to develop the coating's precursor solution, the reaction requires 90 seconds to be carried out completely. An insufficient exposure time to UV light would yield an incomplete AZ-mediated crosslinking reaction that could generate cytotoxic residues (21). On the other hand, the incorporation of LA and AZ in the CH backbone compared with unmodified CH was corroborated by contact angle measurements. A fundamental prerequisite for the elaboration of a coating starting from a solution is that the liquid must be able to wet the surface. The chemical modification of CH was translated into a decrease in contact angle, exhibiting greater spreading and probably stronger adhesion of the modified CH to the metallic surface (32, 33).
The physicochemical phenomena that occur at the solid–liquid interface during the coating process can be divided into 2 stages: prior and post exposure to UV light. The first stage follows the principles of immersion type coatings (dip coatings), in which the adsorption-desorption kinetics of the LA-CH-AZ solution during the initial minutes are important. The second stage, which yields the final coating, depends on the amount of polymer adsorbed during this stage.
Currently, it is well known that titanium and its alloys, in contact with biological fluids, tend to be passivated, forming a thin oxide layer on the surface (3-6 nm) that is responsible for the elevated biocompatibility of different titanium implants (34). This layer presents electrostatic properties that can be used in the design and production of electroactive materials (35). TiO2 is intrinsically an n-type semiconductor that, when immersed in an electrolytic fluid, can regulate the concentrations of electric and ionic defects. When the hydration of the oxide layer takes place in coming into contact with the CH solution, hydroxide groups form on the surface. These undergo acid–base reactions with the medium and are responsible for electrostatic charges on the interphase. When in contact with fluids at pH 7, there is a slight negative charge due to the deprotonation of OH groups at acidic points. This superficial charge is responsible for the interaction with macromolecules such as proteins, lipids and polysaccharides (35). In this way, the CH solution (pH 7), displaying a polycationic feature and being applied on the metallic surface, can generate an electrostatic mechanism of interaction with the substrate–coating interface during the adsorption. This interaction may, moreover, have been favored by the previous sandblasting treatment of the Ti6Al4V surface (36).
Regarding the second coating stage, the metal–coating interaction can be explained through mechanic anchorage. When the LA-CH-AZ, immobilized on the Ti6Al4V, is irradiated with UV light, the polymer experiences a contraction, during photocrosslinking, which generates a mechanic anchorage favored by the surface roughness created through the sandblasting. Then, when the samples are exposed to aqueous medium, the coating behaves as a hydrogel while maintaining its interactions with the metallic surface.
Cell proliferation on uncoated and coated surfaces was studied by MTT assay. The ability of the cells to reduce MTT onto coated substrates is an indicator of mitochondrial integrity, and its functional activity can be interpreted as an indirect measure of the number of viable cells. According to the data obtained at day 4, it is possible that the surface had been saturated, and by day 10 the cells were still adapting to the surface, hence the maintenance in the level of absorbance. Although uncoated samples had higher absorbance than LA-CH-AZ coated samples at day 10 of cell culture, coated samples increased absorbance after 16 days (Fig. 8) suggesting greater cellular proliferation due to the coating on the metallic samples.
The appearance of filopodia from the cells on the surfaces is a sign of significant cell–surface interactions, essential in the process of osseointegration. Furthermore, the presence of nodules, rich in Ca and P, as revealed by XDS, showed that the cells were capable to secrete extracellular matrix that was later calcified. This is an indication of late osteogenic differentiation.
Conclusions
Ti6Al4V surfaces have been successfully functionalized with LA-CH-AZ solutions to produce a photocrosslinkable and cytocompatible coating, which can also support MSC osteogenic differentiation. Until now, LA-CH-AZ hydrogels had only been used as biological adhesives for soft tissue or injectable controlled drug delivery systems (21, 26, 30, 37-38-39-40); thus, important conclusions can be drawn from the present work. It is well known that the main attractive properties of CH as a biomaterial are constituted by its reactive groups; in this study, a chemical modification was carried out compromising only 1.5% of the amine groups for the reaction with AZ, suggesting that enough free amines remained to preserve the polymer's antibacterial and biocompatible features. Furthermore, the evolution of the LA-CH-AZ product into a hydrogel represents an alternative to encapsulate relevant biomolecules for their controlled delivery. In fact, in previous works, LA-CH-AZ hydrogels have been used to encapsulate cells and growth factors without affecting their activity (30, 38, 40).
This study proposes a photocrosslinked CH coating with its ease of application as the main advantage. The coating's deposition can be carried out in the operating room since it requires an exposure to UV light for only 90 seconds, using a portable lamp.
Footnotes
Financial support: This work was funded by Venezuela's National Development Fund (Fondo de Desarrollo Nacional, FONDEN).
Conflict of interest: The authors state that there are no conflicts of interests in the present work.

