Abstract
Background
Local infection near an implant may pose a serious problem for patients. Antibiotic delivery from acrylic (poly(methyl methacrylate)-based) cements is commonly used to prevent and treat infections in the proximity of, e.g., hip joint implants. However, at present, the drug release properties of PMMA cements are not optimal. An initial burst followed by very slow release means that an unnecessarily large amount of antibiotic needs to be added to the cement, increasing the risk of bacterial resistance. The main purpose of this study was to enhance drug delivery from PMMA cements without influencing the mechanical properties.
Methods
We incorporated strontium-doped calcium phosphate spheres (SCPS) into PMMA cement to enhance the antibiotic release and potentially improve the bone-cement integration. The release of strontium and vancomycin was investigated using inductively coupled plasma atomic emission spectroscopy and UV spectrophotometry, respectively.
Results
It was found that incorporating SCPS into PMMA could enhance the antibiotic release and deliver strontium ions to the surroundings. The incorporation of SCPS also increased the radiopacity as well as the working time of the cement. The compressive strength and Young's modulus were not affected.
Conclusions
Our results showed that SCPS/PMMA antibiotic-loaded cement had enhanced antibiotic release, delivered strontium ions and maintained mechanical properties, indicating that the SCPS additive could be a good alternative for controlling the drug-delivery properties of PMMA cement.
Introduction
Infections can follow surgery and be caused by direct contamination of the biomaterial or the surrounding tissue, or by blood-bone contamination or the spread of a superficial infection (1). In the case of joint replacement, infection is a devastating complication, which causes painful disability, prolonged hospital stay and increased medical expenses (2).
Antibiotic-impregnated poly(methyl methacrylate) (PMMA) cement is effective for providing sustained high concentration of antibiotics locally, in the treatment and prophylaxis of orthopedic infections (3-4-5). A retrospective cohort study of 501 shoulders showed that antibiotic-impregnated PMMA cement was effective in reducing deep infection for primary reverse total shoulder arthroplasty (6). The risk of infection after knee or hip revision arthroplasties is higher than primary knee or hip arthroplasties (7). Two-stage revision surgery using antibiotic cement to treat the infection associated with total hip or knee arthroplasty attained a higher rate of success (91%) than the same procedures without antibiotic-loaded cements (82%) (8). Accordingly, antibiotic-impregnated PMMA spacers are the first choice for patients with a chronic infection at the site of a total joint replacement (7). There are also indications that antibiotic-loaded cement could be useful in minimally invasive procedures, such as vertebroplasty and kyphoplasty (9, 10).
These above-mentioned studies have provided strong evidence that antibiotic-loaded bone cement can play a crucial role in the prevention and treatment of infection during and after surgery. However, large doses of antibiotics need to be loaded into PMMA cement to maintain high concentrations of antibiotics, which is a concern since excessive use of antibiotics can cause bacterial resistance (11). Therefore, the rate of drug release from cements needs to be improved to increase their antibacterial efficacy.
Calcium phosphate–based materials are biocompatible and can be used as a bone-filling material as well as a drug delivery vehicle (12). Compared with dense materials, hollow spheres are a promising system to safely transport and deliver a high dosage of antibiotics, with a controlled release profile. In this study, we used strontium-doped calcium phosphate hollow spheres (SCPS), which we have previously synthesized and characterized (13), to deliver vancomycin and strontium ions. Vancomycin is one of the most commonly used water-soluble antibiotics allowing a gradual release over time for a sustained bactericidal effect. Strontium ions can enhance bone repair and reduce bone resorption (14-15-16), and the addition of SCPS to the PMMA could potentially improve the bone–cement integration.
In this study, the antibiotic and strontium ion release properties of PMMA/SCPS cements were investigated as well as their mechanical properties, working time and radiopacity. Our aim was to show that incorporation of SCPS could improve the antibiotics release and deliver strontium ions while maintaining adequate handling properties of the PMMA cement.
Materials and methods
Materials
Vancomycin, strontium nitrate, sodium chloride, potassium chloride, sodium phosphate dibasic, potassium phosphate monobasic, calcium chloride and magnesium chloride were purchased from Sigma-Aldrich (St. Louis, MO, USA) and used without further purification. Commercially available PMMA bone cement for joint replacement fixation, Surgical Simplex P (Stryker Corporation, Kalamazoo, MI, USA) was used as the base cement.
Synthesis of strontium-doped calcium phosphate hollow spheres
Strontium-doped calcium phosphate spheres (SCPS) were prepared based on our previously published method (13). In summary, strontium nitrate (0.6 mM) was added in as-prepared phosphate-buffered solutions with calcium and magnesium ions. The above solution was then sealed in a glass bottle, and kept in an oven at 100°C for 24 hours. The precipitate obtained was filtered, washed with ethanol and dried at 60°C for further use.
Preparation of PMMA and PMMA/SCPS specimens
PMMA/SCPS specimens were prepared according to the compositions shown in Table I. Unmodified PMMA (Simplex) was used as control. For preparation of Simplex-10SCPSV, Simplex-2.5V and Simplex-10SCPSV-2.5V, SCPS and/or vancomycin were first mixed with the PMMA powder component, into which the liquid component was added. The powder and the liquid components were mixed in a glass mortar for 30 seconds, according to the manufacturer's instructions.
Compositions of the powder components of the investigated PMMA and PMMA/SCPS cements loaded with vancomycin

PMMA = poly(methyl methacrylate); SCPS = strontium-doped calcium phosphate hollow spheres.
For Simplex-10SCPSV (see Tab. I), vancomycin was first loaded into the SCPS, which were then mixed with the cement powder. SCPS (570 mg) were soaked with 23 mL of vancomycin solution (10 mg/mL) and stirred for 24 hours. The amount of drug loaded into the SCPS (VSCPS) was calculated according to the equation: VSCPS = (VSol1-VSol2)/P × 100%, where VSol1 is the amount of drug in the solution before loading, VSol2 the amount of drug in the solution after loading, and P the mount of powder.
After centrifugation and drying, 10 wt% vancomycin-loaded SCPS were mixed with the PMMA powder component.
Twelve samples per group for Simplex-2.5V, Simplex-10SCPSV and Simplex-10SCPSV-2.5V were prepared for drug and ion releases. Eleven samples per group for Simplex and Simplex-10SCPS were prepared for mechanical testing and radiopacity measurements.
Antibiotic and ion release
To measure the release, cylindrical samples of 3.3 mm in height and 6 mm in diameter, were placed in 2 different phosphate-buffered saline (PBS) it's right solutions, with pH values of 4 and 7.4, respectively. Each group included 12 samples, where 6 samples were placed in pH 4 PBS and 6 samples in pH 7.4 PBS. Each sample was placed in a glass bottle containing 3 mL of PBS solution. During sampling, a 2-mL aliquot was extracted from each bottle, and 2 mL of PBS with the corresponding pH was added to maintain the same volume. Aliquots were extracted after 0.5, 3.5, 9.5, 21.5, 45.5, 93.5, 141.5 and 189.5 hours.
The antibiotic concentration was measured at a wavelength of 280 nm using an 1800 UV spectrophotometer (Shimadzu, Kyoto, Japan). The amount of strontium was quantified using inductively coupled plasma atomic emission spectroscopy (ICP-AES; Kleve, Germany). The release profiles for vancomycin and Sr ion were fit to the Higuchi model (17).
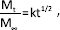
where
Scanning electron microscopy
The morphology of PMMA, SCPS and PMMA/SCPS was analyzed by field emission scanning electron microscopy (FESEM; LEO 1550; Zeiss, Oberkochen, Germany). The samples were sputtered with Au/Pd before analysis.
Working time
The powder and liquid components were thoroughly mixed in a mixer for 30 seconds, and then transferred to a BD 3-mL syringe. The syringe was then placed in a custom-made rig, and the cement was extruded using a universal materials testing machine (Shimadzu, Kyoto, Japan) at a displacement rate of 3 mm/min. The measurement was stopped when the load reached 200 N. The working time of each cement sample was analyzed taking the start of mixing as time point zero.
Uniaxial compression testing
Eight specimens (12 mm in height and 6 mm in diameter) were tested in compression, as stipulated in ASTM F451-08 standard 18 (18). The specimens were left to set in the moulds for 1 h and then taken out and stored at T = 21 ± 1 °C for 24 h. Finally, the specimens were tested using a universal materials testing machine (Shimadzu, Kyoto, Japan) at a displacement rate of 20 mm/min.
Radiopacity
Radiographs of 3 specimens (1 mm in height and 5 mm in diameter) per group were taken at 80 kV using microcomputed tomography (Skyscan 1172; Skyscan, Bruker, Kontich, Belgium). The radiopacity was calculated from a standard curve (amount of light vs. mm Al) created from an aluminium scale (0.5-4 mm) as a reference. The grayscale measurements were carried out using the adjustments tool in Adobe Photoshop CS4 (Adobe Systems Inc., San Jos, CA, USA).
Statistical Analysis
Statistical analysis was carried out in IBM SPSS v19.0 (IBM, Chicago, IL, USA), using a t-test and analysis of variance (ANOVA). The significance level was set to a<0.05.
Results
Drug loading and release
The loading of vancomycin in Simplex-10SCPSV was 2.8 wt%, which was slightly higher than in Simplex-2.5V (2.5 wt%) and Simplex-10SCPSV-2.5V (2.5 wt%). This could be due to the high drug loading of SCPS, which reached approximately 38 wt%. Figure 1 shows the cumulative release of vancomycin from Simplex-2.5V, Simplex-10SCPSV and Simplex-10SCPSV-2.5V at pH 4.0 and 7.4. In all groups a burst release followed by a sustained release was observed. The vancomycin release was faster in PBS pH 4.0 as compared with the release in PBS pH 7.4. Vancomycin release from Simplex-10SCPSV-2.5V was the fastest, followed by the release from Simplex-2.5V and Simplex-10SCPSV. The incorporation of SCPS in PMMA cement hence facilitated drug release, and the release became slower when Vancomycin was incorporated into the PMMA cement without SCPS. Figure 1 also shows that the vancomycin release in PBS at pH 4 was faster than that in PBS at pH 7.4. In all groups, there was a burst release, followed by a sustained release over the entire period of measurement.
Cumulative release of vancomycin in PBS pH 4
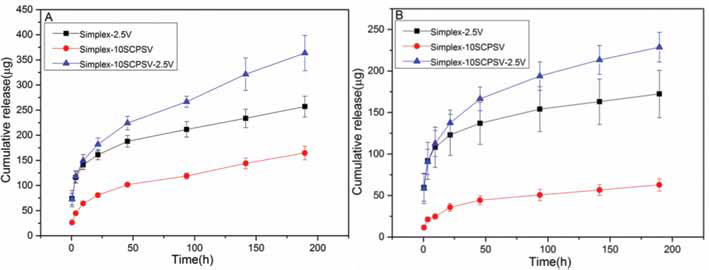
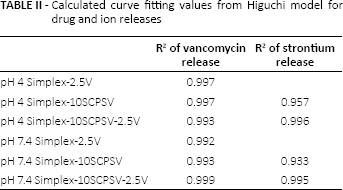
In Figure 2 and Table II, it is shown that the release behavior fits the Higuchi model, which means that the release of vancomycin from Simplex-2.5V, Simplex-10SCPSV and Simplex-10SCPSV-2.5V followed the Fickian diffusion model.
Higuchi analysis of vancomycin release in PBS pH 4
Calculated curve fitting values from Higuchi model for drug and ion releases
Ion release
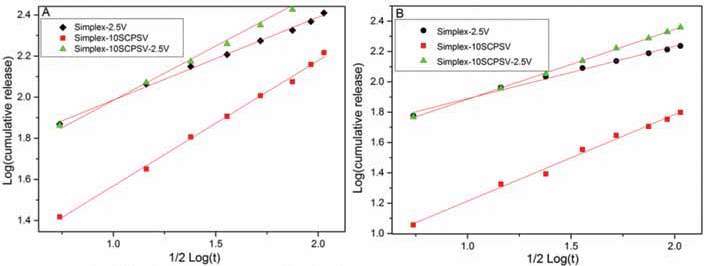
In PBS at pH 4.0, the strontium release from Simplex-10SCPSV-2.5V was faster compared with that from Simplex-10SCPSV (see Fig. 3). However, the opposite was observed at pH 7.4, with a lower strontium release from Simplex-10SCPSV-2.5V compared with that from Simplex-10SCPSV. The calculations based on the Higuchi model showed that the release occurred via diffusion (Fig. 4 and Tab. II).
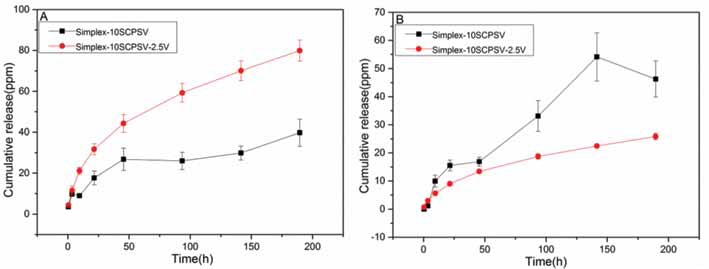
Cumulative releases of strontium from composites in PBS pH 4
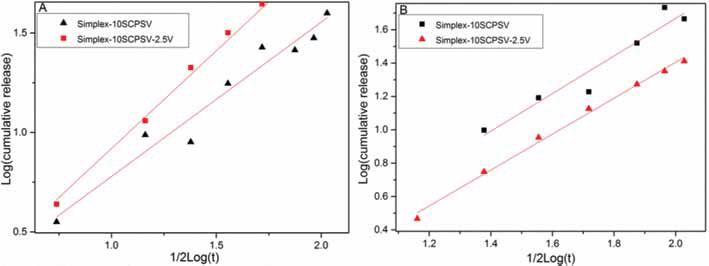
Higuchi analysis of strontium release in PBS pH 4
Working time
The working time of the cements is shown in Figure 5. The working time of the modified cement, containing 10 wt% SCPS in the powder, was approximately 50 seconds longer than that of regular PMMA.
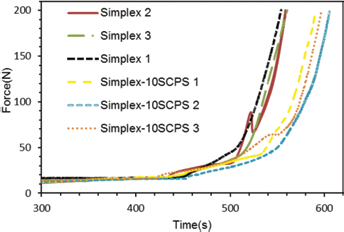
Working time of polymethylmethacrylate (PMMA) and PMMA /strontium-doped calcium phosphate hollow spheres (SCPS).
Mechanical properties
The mean values of the elastic modulus and compressive strength of Simplex and Simplex-10SCPS are shown in Figure 6. No significant differences were found in either compressive strength (p = 0.085) or elastic modulus (p = 0.446) between Simplex and Simplex-10SCPS, suggesting that the SCPS incorporation did not influence PMMA's mechanical properties.
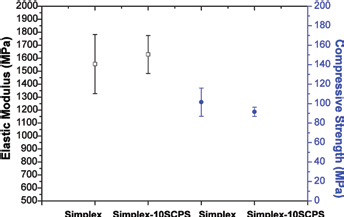
Elastic modulus and compressive strength of polymethylmethacrylate (PMMA) and PMMA/strontium-doped calcium phosphate hollow spheres (SCPS).
Radiopacity
The relative radiopacity of PMMA was 0.34 mm Al, while it increased to 0.67 mm Al after modification with SCPS, as shown in Figure 7. The relative radiopacity of PMMA/SCPS was significantly higher than that of unmodified PMMA (p = 0.002).
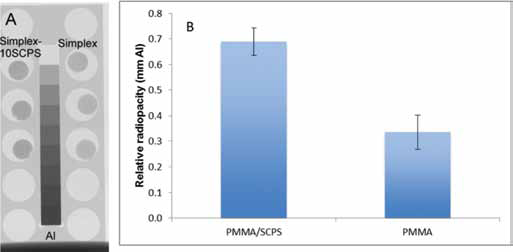
Radiograph of the of polymethylmethacrylate (PMMA) cement and PMMA/strontium-doped calcium phosphate hollow spheres (SCPS)
Cross-section analyses
Figure 8 shows the cross-section images of Simplex-2.5V, Simplex-10SCPSV and Simplex-10SCPSV-2.5V, before and after drug release, in PBS at pH 4. The spheres in Simplex-10SCPSV and Simplex-10SCPSV-2.5V were not homogeneously distributed in the PMMA matrix, but aggregated in some areas (see arrows in Fig. 8 B, C). On the other hand, the morphology of SCPS was retained after mixing with PMMA (Fig. 8D).
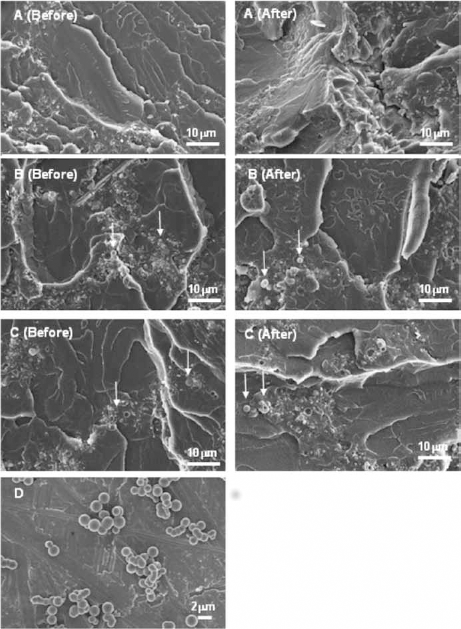
Cross-section images of polymethylmethacrylate (PMMA) cement before and after drug release Simplex-2.5V
Discussion
In the present study, the release of antibiotics from unmodified PMMA was similar to what has been observed in previous studies (18, 19). There was an initial burst release of antibiotic in the first few hours, followed by a steady decline of antibiotic release. In our study, the addition of calcium phosphate spheres was found to improve the vancomycin release, possibly since the incorporation of spheres increased the void volume fraction and induced the formation of additional defects within the PMMA cement, hence leaving more room for vancomycin to elute (20). When vancomycin was loaded into the spheres, the release rate decreased. This was probably due to the release of vancomycin taking place in 2 steps: firstly, penetrating the shell of the sphere and secondly, diffusing through the channels of the cement.
The pH value of body fluid is 7.4, while the body fluid surrounding infected tissues is around 4 (21). Acidic medium may increase the degradation of SCPS, which would result in a quicker release of drugs and ions from the cement. The strontium release from Simplex-10SCPSV-2.5V showed a similar trend to that of previous reports on bone cement – i.e., a burst release during the first few hours followed by a slow release (22). Interestingly, strontium ions release from Simplex-10SCPSV showed a different behavior. The loaded vancomycin may influence the strontium ions’ release from the cement. In this study, the results suggest that both an acidic environment and drug loading can improve the strontium release rate from the cement.
The working time – i.e., how long the cement can be injected – is an important aspect for clinical applications. The working time, when the force of injection was lower than 150 N, was around 9 minutes, which is in accordance with previous studies (23). Adding SCPS to PMMA prolongs the working time, which could be advantageous. However, the increased working time may have a negative effect on the risk for leakage, which remains to be investigated. No significant changes in compressive strength and elastic modulus were found between the unmodified PMMA and SCPS modified cement.
The incorporation of SCPS into PMMA increased the radiopacity since SCPS contain strontium. The effect of the content of strontium on radiopacity of cements has been investigated previously. Strontium halides have been evaluated as radiopacifiers in injectable calcium phosphate cements (24), and the radiopacity of glass ionomer cements has been found to increase as strontium content increases (25). An increased radiopacity could be especially useful in minimally invasive procedures.
Conclusions
The incorporation of strontium calcium phosphate spheres can facilitate vancomycin release from PMMA cement when SCPS and vancomycin are mixed with PMMA. However, the release rate decreased when the vancomycin was preloaded into the SCPS. As the loaded drug was released, strontium ions were also released from the cement, which could have positive biological effects. The incorporation of SCPS did not change the mechanical properties of PMMA under compression. The working time was slightly increased for the PMMA/SCPS cement, as was the radiopacity. The results of this study show that PMMA/SCPS cement could be an alternative to currently used PMMA cements, where enhanced drug delivery is desired.
Footnotes
Financial support: The Swedish Research council (VR, Grant no. 2013-5419), VINNOVA (VINNMER 2010-02073) and China Scholarship Council provided financial support for this project.
Conflict of interest: None.
