Abstract
The design and development of novel materials for biomineralized tissues is an extremely attractive field of research where calcium phosphates (CaPs)–based materials for biomedical applications play a leading role. The biological performance of these compounds can be enhanced through functionalization with biologically active ions and molecules. This review reports on some important recent achievements in creating functionalized biomimetic CaP materials for applications in the musculoskeletal field. Particular attention is focused on the modifications of these inorganic compounds with bioactive ions, growth factors and drugs, as well as on recent trends in some important CaP applications as biomaterials – namely, as bone cements, coatings of metallic implants and scaffolds for regenerative medicine.
Introduction
Biomimetics mimics the strategies utilized by living organisms to fabricate biological materials for the development of new functional materials (1). In practice, biomineralized tissues often exhibit peculiar morphologies, structures and mechanical behavior, and provide useful models for the design of complex functional materials (2–4). Moreover, biogenic minerals are generally produced using low-cost, easily available raw materials, in mild conditions of temperature, pressure and pH. Thus, it is not surprising that biomimetics has developed and spread into many scientific areas – in particular, in the field of new biomaterials aimed to repair the skeletal system (5–8).
The success of a biomaterial for bone substitution requires the ability of the synthetic material to bond to the surrounding biological tissue (9). To this aim, similarity between the synthetic and the biological material, in composition, as well as in structure, morphology and functionality, plays a key role (10). Bone is a highly organized tissue assembled from nano to macro scales to give hierarchical networks (8, 11). During bone mineralization, the inorganic crystals are laid down on an organic matrix rich in collagen fibrils. Collagen is synthesized, secreted by the cell and then self-assembled in the extracellular space, where it acts as a scaffold for the mineral deposition. The organic matrix also contains acidic macromolecules, rich in negatively charged groups, which exert a further control on the modality of deposition, including nucleation, polymorphism, growth, orientation, shape and dimensions of the crystals (11–13). The mineral phase consists of a basic calcium phosphate (CaP), similar to synthetic hydroxyapatite (HA), Ca10(PO4)6(OH)2. Once mineralized, collagen fibrils are used to build up complex hierarchically assembled structures (14), which are designed to fulfill a wide variety of mechanical functions (15–18). A further peculiarity of bone tissue is its continuous remodeling, which is achieved through the resorptive activity of osteoclasts and the synthetic activity of osteoblasts (19, 20).
In spite of the complexity of the events involved in bone synthesis, which include hierarchical structuring, adaptive growth and constant remodeling, significant advances in understanding the biomineralization process have been made (16, 19, 21, 22). Nonetheless, the reproduction of the whole biomineral assembly mechanism by nonbiological methods cannot be regarded as an affordable goal. A more realistic approach is to develop bioinspired processes for obtaining tailored functions using cheap base materials.
Synthetic HA is the most widely utilized CaP for hard tissue applications due to its excellent biocompatibility and bioactivity, as well as its similarity with biological apatites. However, at variance with HA, bone apatite is poorly crystalline, consists of smaller crystals and contains a number of foreign ions, which account for its nonstoichiometry. On this basis, nanocrystalline biomimetic apatite, and further CaPs more resorbable than HA, such as dicalcium phosphate dihydrate (DCDP), octacalcium phosphate (OCP) and α- and β-tricalcium phosphate (α-TCP; β-TCP), most of which can easily hydrolyze into apatite, are gaining increasing attention (23–26). Some characteristic morphologies of CaPs are shown in Figure 1. Bone response to synthetic CaPs can be improved through modifications with biologically active ions and molecules. This paper reviews some relevant recent results achieved in the field of functionalized biomimetic CaP materials for bone tissue replacement and repair.

Transmission electron microscopy (TEM) image of hydroxyapatite crystals (A); scanning electron microscopy (SEM) images of octa-calcium phosphate (B) and β-tricalcium phosphate (C) crystals.
Functionalized CaP Crystals
Due to the many different ions found in the biological environment in close association with the mineral phase of calcified tissues, a great deal of attention has been given to ionic substitutions, especially in the structure of HA but also of other CaPs (23, 27, 28). Relevant biological ions include both bivalent, such as Sr2+, Mg2+ and Mn2+, and monovalent, such as Na+ and K+, cations, as well as anions, with CO32- and F” occupying a predominant role (Fig. 2) (29). Ionic substitutions, even to a limited extent, can greatly modify the physicochemical properties and bioactive behavior of CaPs. In particular, the amount of Si that can be incorporated into the HA structure is quite limited (0.1%–5%) (30), although this can be enough to provoke important improvements of the bioactivity of HA (31, 32). Silicon incorporation into the HA structure involves charge compensation, because of the replacement of a PO43- with a SiO44-, and provokes the creation of crystalline defects. Recently, it has been shown than nanocrystalline SiHA delays early osteoclast-like cell differentiation and decreases the resorptive activity of those cells (33).
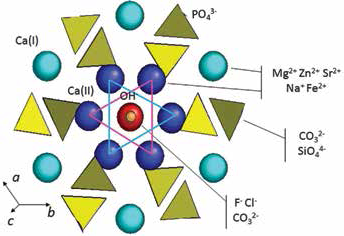
Perspective view along the
Strontium-enriched CaPs have recently been the object of increasing interest due to the success of strontium, administered as strontium ranelate, in the treatment of patients affected by osteoporosis (34, 35). Sr can replace Ca in the HA structure in the whole range of compositions (36). Sr substitution for Ca influences HA solubility, which increases with increasing Sr content (37, 38), thus affecting ionic release and finally biological response. In vitro studies carried out using normal and osteopenic osteoblasts and osteoclasts have suggested that SrHA promotes bone growth and inhibits excessive bone resorption in a dose-dependent manner (39, 40).
Although the structure of OCP is less favorable for ionic substitution than that of HA, Sr can replace Ca in the mono-clinic lattice of this CaP up to 7.4 at% (41). At variance with Sr, which has an ionic radius greater than that of Ca, smaller ions, such as Mg2+, Mn2+, Zn2+ and Co2+, usually destabilize HA, as well as OCP, and they prefer structures with available octahedral coordination sites, such as β-TCP (42–44).
Magnesium plays a key role in bone metabolism, and its reduction has been associated with decreased osteoblast activity, with consequent increase of fragility and loss of bone mass (45). The presence of Mg induces a decrease of crystallinity of HA (46–48), which may be coupled to increased surface hydration (49) and yields increased solubility and reduced thermal stability (50, 51).
As in the case of Mg2+, the HA structure does not sustain the incorporation of relatively high amounts of Zn2+ (29, 48). The presence of Zn in HA, as well as in β-TCP, inhibits in vitro osteoclast development and activity (52, 53).
Antiosteoclastic properties have been reported also for gallium (54), which has recently raised significant interest in view of the possibility of its being associated with CaPs to give antiresorptive materials (55).
Biomimetic strategies also involve functionalization of CaPs with organic molecules or macromolecules, as synthetic analogues of the acidic macromolecules present in biomineralized tissues, with the purpose of modulating CaP nucleation, growth, crystal structure, crystallinity and solubility. In particular, the interaction of CaPs with a variety of amino and polyamino acids has been investigated, to help clarify the mineralization process and to obtain materials able to provide an improved interfacial bonding with bone tissue (56–60).
The use of CaPs as potential delivery systems for osteogenic agents, such as growth factors and hormones, or other specific drugs, represents an even more difficult and attractive challenge. The continuously increased aging of the population in developed countries is the main cause of the observed serious increase in age-related musculoskeletal disorders, most of which are related to loss of bone mass, in conditions such as osteoporosis (61). CaPs functionalized with antiresorptive, antibacterial, antiinflammatory agents are promising systems to answer the increasing demand for materials for the repair and substitution of damaged tissues.
Bisphosphonates (BPs) are the most popular antiresorptive drugs, utilized for treatment of metabolic diseases, such as osteoporosis and Paget's disease, and for the prevention and treatment of bone metastases (62, 63). BP-functionalized CaPs can be prepared through chemisorption from solution, as well through coprecipitation (64–70). In vitro results generally indicate that BPs maintain their inhibiting effect on osteo-clastogenesis and on osteoclast activity even if incorporated into CaPs (66, 67, 71–73). Both alendronate and zoledronate, two powerful amino BPs, can be incorporated into HA up to a maximum amount of about 7 wt%, through coprecipitation. In both cases, functionalization causes a reduction of the crystal dimensions, as shown in Figure 3 for zoledronate. The results of in vitro tests revealed a greater effect of zoledronate on osteoclast (67). Zoledronate-HA displays a better inhibitory action toward osteoclasts also compared with SrHA, which has a better stimulating action on osteoblasts. These results support the hypothesis that the cumulative effect of the two functionalized agents would be the optimal solution for therapeutic purposes (70). A recent in vivo study showed that zoledronate-HA nanocrystals injected intravenously into a rat model of postmenopausal osteoporosis displayed a greater effectiveness in promoting bone formation compared with pure zoledronate (74).

Transmission electron microscopy (TEM) images of hydroxyapatite nanocrystals with different zoledronate contents: 0 wt% (A), 4.0 wt% (B) and 7.4 wt% (C). Scale bars = 100 nm.
Loading CaPs with bone morphogenetic protein (BMP-2) and vascular endothelial growth factor (VEGF) was investigated with the aim of developing new materials with improved osteogenetic and angiogenetic properties, respectively (75, 76).
In addition, CaPs have been proposed as delivery systems for anticancer drugs, including cisplatin and doxorubicin (77, 78), and antibiotics, including gentamicin, vancomycin and β-lactams (72, 79).
Bone cements
CaP bone cements (CPCs) can be used to completely fill bone defects or cavities, as well as be molded in customized shapes (80, 81). The cementitious process implies the reaction between one or several CaPs and a liquid phase, which produces a workable paste that stiffens during the reaction, yielding in situ formation of a solid CaP (82). The reaction does not provoke an inflammatory response, since it is not exothermic, which represents a relevant advantage compared with polymethylmethacrylate (PMMA), the most widely employed material for implant fixation. Moreover, CPCs display excellent biocompatibility and are able to activate osteogenesis.
The first CPC consisted of tetracalcium phosphate (TTCP; Ca4(PO4)2O) and dicalcium phosphate anhydrous (DCPA; CaHPO4) (83), and it received the approval of the US Food and Drug Administration (FDA) for the treatment of non-load-bearing bone defects in 1996 (84). Since this first formulation, a lot of compositions have been tested, and a number of studies have been performed on the effects of several parameters, such as dimensions of cement powder, composition of the liquid phase and powder to liquid ratio, on the properties of the cements (82, 85, 86).
The possible final products for CPC hardening reactions can be brushite (CaHPO4·2H2O; DCPD), which, however, is a metastable phase and easily transforms into apatite, either as stoichiometric HA or as calcium-deficient HA (Ca9(HPO4) (PO4)5 OH; CDHA). Both cement types involve the dissolution of a less stable CaP phase to form a more stable one (87). At 37°C, brushite is the thermodynamically more stable phase at pH <4.2, whereas HA is the most stable at higher pH values. Brushite CPCs display shorter setting times than apatite CPCs, a fact that has been ascribed to the higher solubility of cement raw materials and to the faster crystal growth of DCPD (88).
Many CPC formulations involve the hydrolysis of α-TCP powder. The apatite obtained from the hardening reaction in an aqueous environment at 37°C is similar to bone apatite (89). The characteristic parameters able to promote fast kinetics and reduce the setting time of CPCs include small particle size, poor crystallinity, high setting temperature, low liquid to powder ratio (L/P ratio), as well as the presence of accelerating agents in the liquid or in the solid phase. Setting times must be long enough to allow workability during surgery, and fast enough to avoid delaying the operation. Efforts have been addressed to developing CPCs that mimic more closely the characteristic of hard tissues, and that enhance resorption and promote osteogenesis. To this aim, biomimetic strategies suggest the introduction of biopolymers, such as collagen and gelatin, in the cement composition (89–92). In particular, the addition of gelatin was found to accelerate cement hardening, improve mechanical properties, stimulate osteoblast proliferation and enhance the levels of osteoblast differentiation markers (87, 92, 93). As an exemplum, the compressive strength of an α-TCP-based cement enriched with gelatin was found to be 4 times that of the control cement (89).
CPCs are intrinsically microporous. Microporosity, which increases with increasing liquid to powder ratio (94), is essential for impregnation of biological fluids. On the other hand, the presence of interconnected macropores promotes bone ingrowth (95). Macroporosity can be created through porogen leaching and gas foaming (96). Recently, a new method based on mixing the cement CaP powder with a previously foamed surfactant-containing liquid has been proposed (97).
The presence of macro pores increases permeability and, as a consequence, degradation, but it also significantly worsens the mechanical performance of the cement. Poor mechanical properties is a minor issue in low- or non-load-bearing applications, whereas CPCs use in load-bearing applications requires a strength similar to or greater than that of the bone tissue being replaced (98). Solid phase additives, such as HA and DCPD, as well as the addition of accelerators and retardants to the liquid phase, have been proposed to increase cement strength (80, 86, 99–101). Increase of DCPD content up to 5 wt% in the composition of an α-TCP-gelatin cement was found to enhance the values of compressive strength and Young modulus from 16 to 35 MPa and from 480 to 1,600 MPa, respectively (86).
A further strategy for reinforcement involves the addition of fibers to the cement matrix: both resorbable and nonresorbable fibers have been tested to this aim, and the influence of several parameters, such as fiber length, amount and orientation, on cement properties has been investigated (90, 102–105). Enrichment of an α-TCP-based cement with electrospun fibers of poly(L-lactic acid) and poly(lactide-co-glycolide) was found to slow down the conversion of α-TCP to calcium-deficient HA, with a consequent reduction of crystal dimensions and crystallinity of the apatitic phase, which yielded fiber-reinforced cements able to maintain their mechanical properties in the long term (Fig. 4) (106). Incorporation of electrospun poly(D,L-lactide-

Scanning electron microscopy (SEM) micrographs of an α-TCP cement reinforced with electrospun polymeric fibers: starting powder (A) and after 40 days in physiological solution (B).
CPCs are also widely employed as drug delivery systems (108). Drugs can be combined with the solid or liquid phase, or can be introduced in a particulate carrier added to a cement formulation (61). Li et al (109) verified that CPC containing BMP-2 loaded gelatin microspheres yielded faster healing and greater mineralization of vertebral defects in osteoporotic goats compared with controls where BMP-2 was loaded directly on the cement.
Among the therapeutically important ions which can be utilized to functionalize CPCs, a great deal of attention has been given to Sr2+, due to its antiosteoporotic properties (110). The addition of Sr to the liquid phase was reported to have a modest impact on the mechanical properties of the cement (111), whereas its inclusion in the solid phase as SrCO3 provoked a significant increase of the compressive strength (112). Recently, a cement containing α-TCP, DCPA and HA, as well as SrCO3 as source of Sr, was implanted in a rat femur to fill the gap of a wedge-shaped critical size defect mimicking a typical osteoporotic fracture, leading to an increase in bone formation and enhanced remodeling (113).
CPCs have been successfully functionalized also with BPs (114). BPs have been loaded on the liquid phase or through chemisorptions on one of the components of the cement (115–117). Although just small relative amounts of BPs can be loaded into CPCs without provoking excessive lengthening of setting times, they were shown to be sufficient to stimulate positive in vitro and in vivo responses (14–17). In particular, in a recent study, an injectable CPC loaded with alendronate was implanted in osteopenic bone defects in the vertebrae of adult female sheep, showing a positive influence on the microarchitecture of the adjacent trabecular bone (118).
Coatings
Implants at high-load-bearing sites, such as in the case of hip and femur replacement devices, must have suitable mechanical properties and be resistant to wear and corrosion (119). Several metals, including titanium (Ti), Ti alloys and cobalt alloys, as well as stainless steels, provide proper mechanical performance, which is also associated to a high strength to weight ratio in the case of Ti and its alloys. However, bone implant failures, which are mainly ascribed to aseptic loosening or infection, occur frequently (120). CaP coating of the implants provides them with a highly bioactive interface for surrounding bone tissue.
Besides biomimetic deposition, the coating can be realized utilizing one of numerous physical and chemical methods, which include plasma spray (121, 122), pulsed laser deposition (123, 124), matrix-assisted pulsed laser evaporation (125–127), sputtering (128), electrodeposition (129) and sol-gel dipping (130).
Wet chemical techniques offer the best solutions when incorporation of organic or biological components is desired. Moreover, biomimetic methods offer further advantages, such as better control of the crystal structure, morphology and thickness (119). The biomimetic approach is based on deposition of a CaP thin layer from supersaturated solutions in mild conditions similar to those of biomineralization processes. As a consequence, it is a relatively easy and low-cost method, and allows coating of complex-shaped and porous materials (131, 132). Moreover, the composition of the supersaturated solution can be enriched with ions and biological molecules, as well as drugs, which are deposited together with the inorganic layer (133–135).
An interesting functionalization of biomimetic HA coatings on Ti alloys was developed by loading BPs. To this aim, different methods of loading BPs into CaP coatings have been proposed. The results of a comparative study indicated the coprecipitation method to be the best one to control the release profile of alendronate, which is incorporated in the inner layers of the coatings (136). Furthermore, BPs and antibiotics were incorporated simultaneously into the same coating, with the aim to provide a local release of the two different drugs. The results indicated that the presence of the BP, which was found to bind to the coating surface, does not affect the sustained release of antibiotics, which was suggested to provide improved stability of the implant and reduced risk of associated infections (137). Ti coatings of BP-containing CaPs have been obtained also using matrix-assisted pulsed laser evaporation which, in contrast to other physical techniques, allows the transferring of organic or biological materials (125, 127, 138). In vitro tests on HA coatings with increasing alendronate content showed a significant decrease of osteoclast proliferation and decrease of caspase-3. The analysis of phalloidin staining performed after 14 days of osteoclast culture confirmed the significant reduction of the cell number on the alendronate-containing coatings, as can be seen in Figure 5 (138).
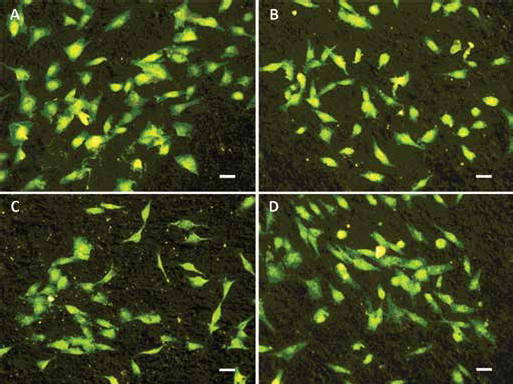
Phalloidin staining of osteoclast cultures after 24 hours from seeding on bare Ti (A) and hydroxyapatite coatings with different alendronate contents (B–D) (0, 3.9 and 7.1 wt%, respectively). Scale bars = 20 μm.
Biomimetic CaP coatings have also been used as BMP delivery systems in successful spinal arthrodesis. The system provides a sustained, localized release of BMP into the fusion site (139).
The first proposed CaP supersaturated solution was the simulated body fluid (SBF) solution proposed by Kokubo et al (140), which had an ionic composition similar to that of human plasma. Since then, many studies have been performed with the aim of accelerating the deposition of a homogeneous CaP layer (141–144). Chemical or physical modification of the metal surface can accelerate deposition and promote adhesion of the coating. A multistep alkali treatment followed by heat treatment is the most common procedure to induce the formation of sodium titanate on a Ti surface, which results in the formation of a chemical bond between substrate and coating (145). The topography of the metallic surface is also very important and can be modified in a controlled way with different methods (141), including simple physical or chemical etching of the surface. Acid etching is very widely used because it is the standard surface-cleaning procedure for Ti substrates, and at the same time, it is able to generate rough surfaces. Lu et al (142) demonstrated that nitric acid treatment significantly increases Ti surface energy, and favors the CaP heterogeneous nucleation process on the metallic surfaces through the reduction of the net interfacial energy.
Recently, coating of Ti-based implants with an anodic TiO2 nanotube layer has been proposed to enhance the formation of apatite (146). Furthermore, thanks to their tubular nature, these coatings are good candidates for the “alternative immersion method,” which is based on preloading the coatings with synthetic HA. This method results in a significant promotion of apatite formation, and the resulting coatings may exhibit improved osteoconductivity while retaining a strong bond with the Ti metal substrate (146).
Variations in composition of the calcifying solution can be used to modify the chemical, structural and morphological characteristics of the coatings, as well as to accelerate the deposition process (147). A highly effective method to obtain a fast biomimetic deposition of nanocrystalline HA onto metallic substrates has been developed, using a slightly supersaturated CaP solution, with a simplified ionic composition compared with that of SBF. The simplified CaP solution (2.5 mM Ca2+, 25.5 mM Na+, 2.5 mM PO43-, 5.0 mM CI– and 18 mM HCO3–) yields the deposition of a uniform CaP coating on Ti alloy substrates in a few hours (130, 148). In contrast to other fast deposition solutions, which generally lead to deposition of an amorphous material, the coating obtained with the new formulation consists of nanocrystalline apatite. Cylindrical samples for in vivo tests (Fig. 6) display a homogeneous coating after 6 hours of exposure to the calcifying solution using specially designed supports. The results of implantation in rabbit cortical bone defects demonstrated that the presence of nanocrystalline apatite accelerated bone ingrowth around the implant (131).

Scanning electron microscopy (SEM) micrographs of the homogeneous coating obtained on Ti13Nb11Zr cylindrical specimens after just 6 hours of exposure to the slightly supersaturated Ca/P solution. The coating consists of nanocrystalline apatite.
Recently, an interesting study presented enzymatic decomposition of urea as a means to increase the pH of the calcifying solution during deposition. It was found that urease, as well as calcium and phosphate concentration, is a major factor in controlling nucleation and morphology of the coating, resulting in flake-like CaP crystals and nano-sized crystals (149).
Scaffolds
The advancement of regenerative medicine requires new complex materials with 3D porous structures, able to host living cells. Ideal scaffolds should be biodegradable and porous, with interconnected porosity to allow cell colonization, revascularization and a suitable supply of nutrients and oxygen (10). In other words, the scaffold must mimic the extracellular matrix (ECM) to promote cell attachment, proliferation, differentiation and normal bone growth during scaffold degradation (150). The ideal biomaterial must further exhibit suitable mechanical and degradation properties.
While many synthetic polymers have been proposed as components of 3D scaffolds for bone repair (151, 152), a biomimetic scaffold should be close to the biological tissue also in terms of composition. CaPs offer a suitable interface, and have the advantage of unlimited availability (10). However, the excellent biocompatibility and osteoconductivity of CaPs are not matched by adequate mechanical properties. The mechanical performance of CaP scaffolding materials can be improved by addition of natural biological components – in particular, protein materials, such as collagen or fibrin, and polysaccharidic materials, such as glycosaminoglycans, chitosan and alginates (153, 154).
Porous scaffolds of gelatin with different inorganic phase (HA) contents were prepared using an optimized method based on foaming and freeze-drying (Fig. 7) (155). Micro-computed tomography reconstructed images indicated that the properties of the scaffolds can be tailored by varying the amount of inorganic phase from 0 to 50 wt%. In particular, compositions up to 30 wt% HA yield scaffolds characterized by very high interconnectivity and porosity, with a mean pore size in the range 100–300 μm, whereas higher inorganic phase content provoked a reduction of these parameters, as well as a significant increase of the compressive modulus and collapse strength. Three-dimensional scaffolds with graded composition were also developed to mimic the different zones of the osteochondral region. The scaffolds were obtained assembling layers containing different amounts of polymers and HA nanocrystals (156, 157).
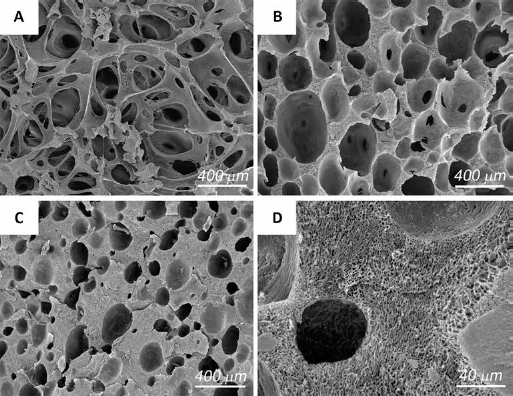
Scanning electron microscopy (SEM) micrographs of gelatin-HA 3D porous scaffolds with different HA contents: 0 wt% (A), 30 wt% (B) and 50 wt% (C). The higher magnification image of (C) shows the microporous structure of the cell walls (D).
A supercritical CO2 foaming method has recently been applied for preparing porous scaffolds by adding HA nanoparticles as filler to the polymeric matrix of polycaprolactone blends with different molecular weights (158). Foams with different structural and mechanical properties were obtained using polycaprolactone samples of different molecular weights and by varying the pressure or filler amount. Increasing the pressure provoked a decrease in pore size, even if porosity was increased. Adding apatitic nanoparticles to polymeric blends reinforced the mechanical properties, but reduced porosity and also pore size. Moreover, the molecular weight of polycaprolactone had an appreciable influence on the morphology of the scaffolds.
In an attempt to imitate the complex hierarchical structure of bone, as well as its composition, many studies have recently focused on the addition of CaP ceramics in fibrous composites formed using the electrospinning technique (159, 160). Thomas et al fabricated nanofibrous scaffolds containing up to 20% of inorganic phase through co-electrospinning of bio-composite fibers of collagen type I and nano HA (161). The nanocomposite fibrous matrices displayed improved mechanical properties and increased surface roughness with the increase in nano HA content. Since then, the electrospinning method has been improved to obtain electrospun polymeric nanofibers containing well-dispersed inorganic nanoparticles. A recent method encompasses the use of a special triaxial needle: mixing calcium and phosphate aqueous solutions in an intermediate needle yields CaP nanoparticles that immediately after precipitation are dragged by the outer polymeric solution and incorporated directly in the electrospinning jet, before nanofiber formation. Gelatin electrospun mats can contain different amounts of nanoparticles well dispersed throughout the fiber length (162).
To produce custom-made bone substitutes, a promising strategy is a rapid-prototyping technique through which complex physical models can be fabricated starting from 3D clinical images. Several processes and types of equipment are nowadays available to produce such complex 3D shapes, the main one being the additive fabrication method, usually based on a layer-by-layer construction (163). Three-dimensional printing has been revealed to be a cheaper technique compared with manufacturing individual prototypes by subtractive processes. The possibility of using a low-cost 3D printer to produce a bone substitute in a polylactic acid/nano-HA composite material has recently been explored (164). The proposed technology ensured a high degree of repeatability in terms of quality and properties of the polymer composite filament and in terms of the 3D-printed final parts. The material processing allowed a good dispersion of the filler in the matrix, and the composite material was successfully printed, showing a combination of good mechanical properties and ease of creation.
A different approach, which exploits the CPC chemistry, has been proposed to synthesize scaffolds with tailored architectures at room temperature using 3D printing (163). The setting reaction of the CaP cement, such as that between α/β-TCP powder and phosphoric acid which yields brushite, causes hardening of the samples during printing, which results in complex 3D structures with a dimensional accuracy of ± 200 μm (165, 166).
Deville et al (167) proposed a HA porous scaffold with a layered arrangement that resembles nacre. These authors used freezing as a tool to develop a variety of complex porous and layered hybrid materials (168). In particular, directional freezing of an aqueous HA suspension, followed by ice sublimation and sintering, results in a multilayered structure characterized by well-defined pore connectivity, as well as open and unidirectional porosity. This approach has recently been implemented in the development of a bidirectional freeze-casting technique to fabricate large-scale lamellar structures (169). The two different temperature gradients provide a graded nucleation and propagation of ice crystals, which gives rise to the assembling of HA particles into aligned lamellar structures. The resulting porous scaffolds exhibit a nacre-like structure with a long-range order at the centimeter scale.
Scaffolds can be functionalized through loading of active substances on their surface or within the bulk structure (61, 170). In particular, BPs have been loaded on CaP based scaffolds through adsorption from solution, within the bulk structure as scaffold components, and through decoration of liposomes which were then incorporated into the scaffolds (171–173) (Fig. 8). Bone regeneration was also reported to be effectively enhanced when scaffolds were functionalized with growth factors or their encoding genes (174).

Surface morphology of polycaprolactone-coated 3D-printed tricalcium phosphate scaffolds. This scaffold was engineered for adsorption of bisphosphonate and controlled drug release applications (171). Copyright 2014 American Chemical Society (available under the terms of the ACS AuthorChoice license: http://pubs.acs.org/doi/full/10.1021/am501048n).
Even better results can be achieved through multifunctionalization, which is aimed at providing local delivery of active agents to promote a variety of functions, including integration, osteoconduction and angiogenesis (175). Vancomycin-loaded gelatin and Si-doped HA 3D porous scaffolds fabricated by rapid prototyping were recently shown to severely inhibit bacterial growth around the implant (176). Sun et al (177) investigated cell responses to growth factors released from a 3D biodegradable porous CaP scaffold and found that angiogenesis and osteogenesis were promoted by a combination of a gradient of 3 different growth factors released from the multilayered scaffold, more readily than by single growth factors. Moreover, a study of codelivering anabolic and anticatabolic agents – namely, BMP-2 and zoledronate – using a biomimetic collagen-HA scaffold, confirmed the ability of the BP to enhance bone formation promoted by the growth factor (178).
Concluding Remarks
Innovative approaches based on biomimetic strategies lead to significant improvements in the performance of CaP biomaterials for hard tissue repair. Further exciting progress has been achieved thanks to the relevant and continuously increasing amount of research on CaPs functionalized with ions, molecules, macromolecules, drugs and growth factors. Modified CaPs can be utilized as-prepared or as raw materials for the preparation of bone cements and coatings, as well as scaffolds for regenerative medicine. Moreover, CaP-based biomaterials are gaining increasing relevance as potential delivery systems for local administration of therapeutic agents.
Footnotes
Financial support: This research was carried out with the financial support of the University of Bologna (FARB 2012).
Conflict of Interest: Both authors declare they have no conflicts of interest.
