Abstract
Background
Cardiovascular heart disease is one of the leading health issues in the present era and requires considerable health care resources to prevent it. The present study was focused on the development of a new coronary stent based on novel auxetic geometry which enables the stent to exhibit a negative Poisson's ratio. Commercially available coronary stents have isotropic properties, whereas the vascular system of the body shows anisotropic characteristics. This results in a mismatch between anisotropic–isotropic properties of the stent and arterial wall, and this in turn is not favorable for mechanical adhesion of the commercially available coronary stents with the arterial wall. It is believed that an auxetic coronary stent with inherent anisotropic mechanical properties and negative Poisson's ratio will have good mechanical adhesion with the arterial wall.
Methods
The auxetic design was obtained via laser cutting, and surface treatment was performed with acid pickling and electropolishing, followed by an annealing process. In vitro mechanical analysis was performed to analyze the mechanical performance of the auxetic coronary stent. Scanning electronic microscopy (SEM) was used to determine the effects of fabrication processes on the topography of the auxetic stent.
Results and Conclusions
The elastic recoil (3.3%) of the in vitro mechanical analysis showed that the auxetic stent design effectively maintained the luminal patency of the coronary artery. Also, the auxetic coronary stent showed no foreshortening, therefore it avoids the problem of stent migration, by expanding in both the radial and longitudinal directions. By virtue of its synclastic behavior, the auxetic stent bulges outward when it is radially expanded through an inflated balloon.
Introduction
Myocardial infarction and angina pectoris are the leading complications of coronary heart diseases. These disorders are mainly due to atherosclerosis, a disease caused by the accumulation of plaque, fats or cholesterol below the endothelium layer of the coronary artery of the heart which lead toward stenosis (1). This build-up of plaque or fats inside the coronary artery causes resistance in the flow of oxygen-rich blood to the walls of the heart. This disturbs the luminal patency of coronary artery and narrows artery walls, which causes complications. These complications may be fatal, and it has been estimated that the number of annual worldwide heart disease–related deaths by the year 2030 will be about 23.3 million (2, 3).
Worldwide each year more than 1 million coronary stents are implanted inside the coronary arteries (4). A stent is a small medical device comprising an expandable mesh-like tubular structure that when implanted acts as a scaffold (4) and maintains the luminal patency of a stenosed artery by opening the narrow coronary artery (5). This in turn allows the oxygen-rich blood to flow smoothly without any obstruction. The chances of myocardial infarction or heart attack are greatly reduced by the intervention of stents. Stents which are corrosion resistant, such as those fabricated with 316L stainless steel, cobalt-chromium alloy and titanium, are clinically used to restore luminal patency and tissue structure inside stenosed coronary arteries (6). The intervention technique deployed to treat the stenosed artery is called percutaneous transluminal coronary angioplasty (PTCA). PTCA is an implantation technique in which a stent is deployed to dilate the stenosed artery (7). Many metallic coronary stent designs have been developed, with different structural and mechanical properties (5).
The 316L type stainless steel stent provides a good platform for clinical applications (8). Previous studies have revealed that stent design has a seconday influence on neointimal proliferation (9) as well as on late lumen loss (10). This ultimately affects postprocedural intervention requirements and the rates of restenosis. Thrombogenesis and platelet activation are also considered to be affected by the design of a stent (11). However, issues such as thrombosis, restenosis, collapses and migrations are still prevalent (12, 13).
Auxetic structures that exhibit a negative Poisson's ratio (NPR) (14) are an emerging development in the field of new biomedical implants and devices. An NPR is a property of a system, which when stretched becomes wider and when compressed becomes narrower; this phenomenon is the opposite of a positive Poisson's ratio (14). Devices with an NPR have the capability of expanding radially when they are stretched, while in the transverse direction under uniaxial compression they contract. Developing biomedical implants with auxetic geometry will not only improve the physical and mechanical properties of the device but will also minimize tissue adhesion damage when deployed into the injured tissue site. In geometry-based models, auxetic behavior is scale-independent just like in auxetic materials, so with deformation mechanisms, the auxetic characteristics can be maneuvered along with the same geometry combination at any stage. Similarly, deformation in the geometric features of the device is due to the effect of the NPR when subjected to uniaxial loads. Material geometry that displays auxetic behavior can be achieved in macro-, micro- and nano-scale levels.
This research presents the fabrication of a new coronary stent based on novel auxetic geometry that averts the localized constriction of blood flow in the coronary artery of the heart. The new coronary stent design complements the anisotropic properties of the body's vascular system. This is a novel structural characteristic that was missing in previously commercialized coronary stents. Based on previous scientific evidence, we can hypothesize that the new auxetic design for a coronary stent will allow good anchorage with arterial walls. The special feature of this design allows the stent to aid in maintaining luminal patency by expanding in 2 directions simultaneously when expanded through an inflated balloon.
Design Considerations of Auxetic Coronary Stent
The design of auxetic geometry of the present coronary stent is built with a plurality of unit cells shown in Figure 1a, interconnected with the help of horizontal and vertical edges to form struts and hinges. The unit design of the stent formed by 4 angulated squircles along with a hollow diamond as shown in Figure 1b. An array of unit cells in the radial and longitudinal directions is fixed through horizontal edges (HA, HB, HC and HD) and vertical edges (VA, VB, VC and VD) in the horizontal and vertical direction, respectively. Throughout, the design width (DW) and length (DL) of the diamonds remains the same. The dimensions of the auxetic stent are 18-mm length, 1.8-mm outer diameter and 0.14-mm strut thickness.
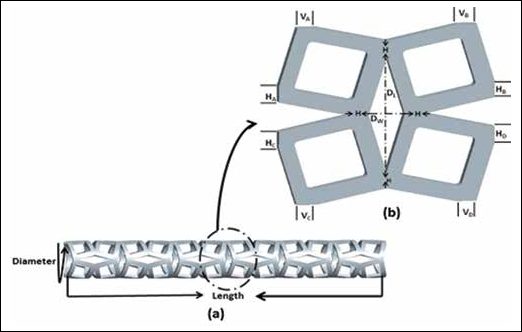
a) Three-dimensional model of auxetic coronary stent design; b) Single unit cell of auxetic coronary stent.
Materials and Methods
The fabrication of the new auxetic coronary stents was carried out using a biocompatible medical grade 316L stainless steel material supplied from Minitubes, France. The composition of 316L stainless steel contains balanced iron, 18% Cr, 14% Ni and 2.5% Mo.
Fabrication of Auxetic Coronary Stent
Process of Laser Cutting
The fabrication of the new auxetic coronary stent begins with the process of laser cutting. The design of the auxetic coronary stent was first developed using AutoCAD software, and then it was translated into a computer and numerically controlled (CNC) CAGILA program. This new auxetic stent design has arrays of squircles and diamonds in the x and y axes. A Starcut 12FM pulsed Nd: YAG laser (Rofin Baasel, Germany) was used for the cutting of the 316L stainless steel tube and fabrication of the auxetic stent.
Surface Treatment of the Auxetic Coronary Stent
Process of Acid Pickling
This method was carried out to effectively eradicate the slag and oxide scales that formed after the process of laser cutting, by submerging the auxetic coronary stents in a solution to complete the acid pickling process. This method mainly removes the unwanted material from the stent. Acid pickling solution (150 mL) for the coronary stents was prepared by combination of pickling solution boron trifluoride (BF3) and oxidizing agent hydrogen peroxide (H2O2). The stents were immersed in 150 mL of pickling solution and ultrasonicated. Subsequently, the stents were rinsed again for 30 seconds with propanol and were placed in an oven for drying at temperature 90°C for 10 minutes.
Electropolishing Process
The next surface treatment method which was employed after acid pickling of the stents was electropolishing. The purpose of the electropolishing process was to make the stent surface smooth, round off edges effectively and reduce the strut thickness of the auxetic stent to the desired level. Power was supplied to the auxetic stent which acted as an anode, and the stent was polarized to the desired level of current and voltage. An electrode heater provided a constant temperature to the electrolyte solution basin. A time controller provided timing for the polishing of the stent, and the stainless steel electrodes inside the basin acted as cathodes.
Heat Treatment (Annealing)
The annealing of the auxetic stents was carried out in a vacuum furnace (Carbolite, Germany), in which a heating rate of 30°C/minute was set, and the temperature was incrementally increased from room temperature to 960°C. This was done to remove the internal stresses from the stent material and to make the stent ductile. The dwelling time of the auxetic stents within a quartz tube inside the furnace was 3.5 hours, and the stents were brought back to room temperature under vacuum with the same mechanism, as shown in Figure 2.

Heat treatment (annealing) setup.
In Vitro Mechanical Testing
In vitro mechanical testing was carried out to analyze and evaluate the mechanical performance of the auxetic coronary stent. The in vitro testing was done experimentally through the inflation device and examined under an optical microscope. The inflation device was filled with normal saline and attached with the balloon catheter. The auxetic coronary stent mounted on balloon catheter was expanded through inflation device by applying pressure increments of 1 atm.
Results and Discussion
Formation of Auxetic Design Pattern
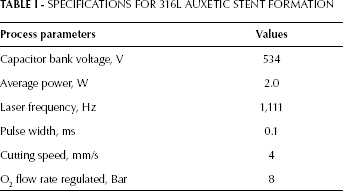
The design pattern of the auxetic stent comprising squircles and diamonds was cut by the interpolation of 316L stainless steel tubing movements in both linear and rotational directions with the help of a flash lamp pumped Nd: YAG laser. The profile of the laser-cut auxetic coronary stent was monitored through an optical microscope and is depicted in Figure 3a. The parameters for auxetic design pattern formation are shown in Table I. No evidence of microcracking was found along the struts of the auxetic stent. From the scanning electron microscope (SEM) micrographs (JEOL-instrument JSM-6490A), it was established that the edges of the auxetic stent struts after laser cutting were quite sharp, and the slags were attached at the cutting zone, as shown in Figure 3b.

a) Auxetic design on laser-cut stent; b) SEM micrograph showing sharp edges along with slags.
SPECIFICATIONS FOR 316L AUXETIC STENT FORMATION
Effect of Acid Pickling on the Auxetic Coronary Stent Design
The unit cells (comprising hollow squircles and diamonds) and struts of the auxetic stent were clearly visible after acid pickling, as shown in Figure 4. The strut thickness of the auxetic stent was reduced by 1 μm after the acid-pickling process. For this auxetic design, time and temperature influenced the pickling rate and played a vital role in the proper elimination of the slags and in the subtraction of surface irregularities. The specifications for removal of slags and metal oxides are listed below in Table II.

Photographs showing close-up view of single unit cell (a) and full-length view of acid-pickled auxetic coronary stent (b).
SPECIFICATIONS FOR REMOVAL OF SLAGS AND METAL OXIDES

D.I. = de-ionized water.
Achievement of Surface Smoothness
The removal of the surface peaks and valleys (i.e., surface roughness) was achieved using the appropriate specification of the electropolishing parameters as shown below in Table III. Material reduction and surface smoothness were obtained by the supply of direct current to the anode (i.e., the auxetic stent) which polarized the auxetic stent to the desired level of current and voltage. The effects of anodic polarization subsequently started transmitting metal material (ions) from anode to cathode (which was a stainless steel plate), which in turn established anodic brightness as well as anodic leveling, and removed peaks and valleys from the rough surface of the anode (i.e., the auxetic stent). As depicted below in Figure 5, surface definition and characteristics of the stent were significantly improved after electropolishing.

Photograph showing a close-up view of the polished stent.
SPECIFICATIONS FOR SURFACE SMOOTHNESS

Auxetic Stent Profile
The auxetic stent profile (i.e., strut thickness and metal ratio) achieved was observed by SEM (Fig. 6). Sharp, flat geometrical features of the acid pickled auxetic stent were transformed into well-rounded, smooth edges. The auxetic stent profile was controlled by time, voltage and current. To achieve the desired strut thickness and surface smoothness, voltage was kept constant while the other parameters were varied. Current was applied to transfer material from auxetic stent (anode) to cathode, and to achieve surface smoothness by the removal of peaks and valleys at a certain polishing time. Polishing time was a very crucial parameter for reducing the thickness of the stent struts and for rounding of the sharp edges of the auxetic stent. Consequently, the thickness of the auxetic coronary stent was reduced, as illustrated in Figure 7.

SEM micrographs showing polished stent: outer surface with round edges (a) and close-up view of inner luminal surface (b).
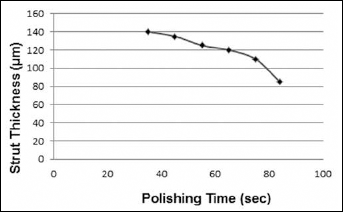
Range of polishing times required for the reduction of auxetic stent strut thickness.
Effectiveness of Annealing on Auxetic Coronary Stent
It was established that the heat treatment of the auxetic stents in the annealing process was very effective, and it not only improved the mechanical properties (i.e., internal stresses were removed and became more ductile when subjected to balloon expansion) of the stent but also improved the quality of the stents, as shown in Figure 8. This was validated by subjecting the auxetic stents to balloon expansion using angioplasty balloon catheters, as described in the following section.

Annealed coronary stent.
In Vitro Mechanical Analysis of Auxetic Coronary Stent
Diameter and Length Before and After Expansion
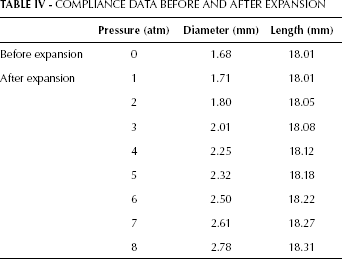
In vitro testing was carried out to observe the auxetic coronary stent behavior before and after expansion. Inflation and deflation of the stent was done experimentally by applying pressure with the inflation device, and the results were examined under an optical microscope. Prior to giving any pressure to the balloon catheter–mounted stent, diameter and length were measured. The expansion of the stent was examined after the pressure was applied at increments of 1 atm, and measurements were taken accordingly. Table IV illustrates the compliance data for the unexpanded and expanded auxetic coronary stent. The compliance data show that the auxetic coronary stent expanded in both radial and longitudinal directions.
COMPLIANCE DATA BEFORE AND AFTER EXPANSION
When pressure was applied, horizontal diamonds widened gradually, and increasing pressure rotated the squircles, while simultaneously, the vertical diamonds widened, as shown in the SEM micrograph in Figure 9.

Expanded auxetic coronary stent unit cell, in which horizontal diamonds have widened and squircles rotated.
Foreshortening
A foreshortening test was carried out, and the auxetic stent exhibited zero percent foreshortening. The lack of foreshortening in the auxetic coronary stent was due to the auxeticity of the coronary stent design, which causes the coronary stent to expand in the longitudinal direction. This is an advantage of an auxetic coronary stent over existing coronary stents where foreshortening causes the problem of stent migration. The present stent undergoes no foreshortening effects and will thus prevent the problem of migration after the stent is expanded in the coronary artery.
Elastic Recoil
For elastic recoil testing, the inflation device pressure was reversed from 8 atm to 0 atm. No evidence of damage in the stent was found during the elastic recoil test. Minimum elastic recoil would be favorable for clinicians to raise the inflation pressure in comparison with coronary artery diameter so that the auxetic coronary stent can properly adhere to the walls of the blood vessels.
Surface Morphology Analysis
A surface morphological analysis of the auxetic coronary stents was carried out by SEM (JEOL-Instrument JSM-6490A) to determine the effects of processes involved in the adopted fabrication route on the topography of the auxetic stent, as mentioned earlier in the sections “Fabrication of auxetic coronary stent”, “Surface treatment of the auxetic coronary stent” and “Heat treatment (annealing)”. SEM micrographs of the inner luminal surface of the auxetic stents were taken at magnifications of ×850 and ×1,500, as shown in Figure 10, and they revealed that the porous and irregular oxide layer was deposited on the inner surface of the stent.

SEM micrographs showing annealed stent surface (magnification ×850 and ×1,200).
After the electropolishing process, an SEM micrograph of the auxetic stent samples was taken at a magnification of ×1,200, as shown in Figure 11a. It was evident that the definition of the squircles and diamonds of the auxetic geometry was quite clear, and the edges were fairly rounded off. The rounded edges are desirable in the case of an auxetic coronary stent to prevent perforation and injury to the surrounding arterial wall. The grain boundaries of the auxetic stent are quite apparent in the SEM micrograph taken at a magnification of ×850, as presented in Figure 11b. The luster and surface brightness in the SEM micrograph revealed that the passivation or electropolishing was done effectively, which not only made the auxetic stent corrosion resistant but also improved the structural integrity of the stent.

SEM micrographs showing electropolished auxetic stent: a) smooth surface (magnification × 1,200), b) obvious grain boundaries (magnification ×850).
The SEM micrographs of the auxetic stent were captured after annealing (heat treatment) of the stents at magnifications of ×850 and ×1,200, as shown in Figure 12. It was observed that the annealing process introduced ductility into the auxetic stents by reducing the size of the grain boundaries, which in turn improved the fracture toughness and structural integrity of the auxetic stent. It was also found that this improvement in mechanical properties of the auxetic stent was very crucial, because the auxetic stent will be deployed into the coronary artery by balloon expansion and will be anchored into the surrounding tissue through plastic radial and longitudinal deformations.

SEM micrographs showing irregular and porous oxide layers on the inner surface of the stent (magnification ×850 and ×1,500).
Chemical Fingerprinting of the Auxetic Stent Surfaces
The analysis of the elemental composition of the inner (luminal) and outer surfaces of the laser-cut, electropolished and annealed auxetic stents was carried out using energy dispersive spectrometry (EDS; JEOL-Analysis Station, JED 2300). The main acquisition parameters for EDS were accelerating voltage: 20.0 kV and probe current: 1.00000 nA. The high oxide levels on the inner surface of the laser-cut stent indicated the presence of oxide layer formation. It was also established that the high chromium content on the inner and outer surface indicated an effective passivation and electropolishing process, which introduced corrosion resistant (barrier layer) of chromium oxide on the inner and outer surface. It was also noticed that the annealing process reduced the size of the grain boundaries, which in turn improved the overall structural integrity and surface topography of the auxetic stent.
Advantages and Benefits of the Auxetic Coronary Stent
The new auxetic coronary stent presents some advantages and benefits as compared with the available 316L stainless steel coronary stents. An auxetic design with perfect round edges minimizes the risk of vessel wall reaction; the auxetic coronary stent can be optimized for specific vessel diameter and length for optimal luminal adaption; the 85-μm struts in the stent structure minimize the risk of thrombogenesis and restenosis; and due to its inherent anisotropic mechanical properties and NPR, the auxetic coronary stent will have good mechanical adhesion with the arterial wall and will retain the luminal patency of the coronary artery. Furthermore, these properties prevent any stent migration problems by expanding in both radial and longitudinal directions.
Conclusion
This study focused on the development of a new auxetic coronary stent for the treatment of coronary heart disease. It is proposed that the new balloon-expandable auxetic stent will have good mechanical adhesion or anchorage on the arterial wall by virtue of its having suitable mechanical properties. The anisotropic properties of the stent are based on its auxetic design, which make it a perfect match for the anisotropic structural properties of coronary vessels. The size of the auxetic stent increases in both radial and longitudinal directions when expanded radially through the balloon catheter, and the stent exhibits no foreshortening. It is believed that the auxetic property of the stent design will effectively maintain the luminal patency of the coronary artery and will prevent any problem of stent migration.
The expansion of the auxetic coronary stent in both directions will improve mechanical anchorage with the walls of arteries. Due to its auxetic design, the stent will anisotropically maintain the luminal patency of the coronary artery. This feature of the present stent has an advantage over the commercially available stents; current commercially available stents have a mismatch between stent structure and arterial wall due to their isotropic properties, which is why these stents do not offer favorable anchorage with the arterial walls.
