Abstract
Purpose
Cranioplasty is a surgical intervention aimed at reestablishing the integrity of skull defects. Autologous bone is still considered the treatment of choice for cranioplasty. The aims of this study were to characterize and evaluate the efficacy of porous hydroxyapatite (HA) to fill skull defects based on its biomimetic characteristics.
Methods
The authors analyzed the postmarketing data of all patients treated with custom-made porous HA prostheses (CustomBone Service). Characterization data in terms of physicochemical analysis and mechanical performance of the porous HA prostheses were also reported.
Results
The low incidence of adverse events (5.72%) due to the use of HA porous custom-made prostheses for cranioplasty is related to the biomimetic performance of the prostheses. The composition and morphology of the porosity enable it to be a useful biomimetic prosthesis for the reconstruction of large and complex skull defects, also able to promote osteointegration.
Conclusions
These collected and analyzed data demonstrate that porous HA is a suitable material to produce custom-made prostheses to repair craniolacunia. It is a biomimetic implant well-tolerated in both adult and pediatric patients and has been shown to be an effective and good alternative for cranial reconstruction.
Introduction
Cranioplasty is a surgical operation aimed at reestablishing the integrity of the skull where there is a defect, usually because of a previous craniotomy. Autologous bone is still considered the gold standard for performing this operation, though it is not always available sometimes abnormal, because it is encumbered by more and more restrictive and costly storage regulations, it gives poor cosmetic results and, finally because may show a high incidence of resorption.
Alloplastic materials such as metals, acrylic and bioceramics have been used to treat cranial bone defects (1). Bioceramics are bioactive and biocompatible biomaterials that integrate well into these defects (2-3-4), and hydroxyapatite (HA) is one of the best-known osteoconductive bioceramics used in clinical practice (5-6-7) because of its structure and chemical composition, which are very similar to the mineral phase of bone. The presence of porosity and a bioactive surface facilitate cell attachment, proliferation and differentiation and, consequently, provide a more biocompatible, osteoconductive ceramic which can favor increased bone formation (8). The chemical composition mimics the mineral phase of bone and as a result of this likeness, the materials seem to be remodeled like normal bone through a cell-mediated process that involves osteoclastic activity. The production of alloplastic implants that are tailored to individual patients’ defects not only improves aesthetic outcomes, but also decreases surgical time, blood loss and the risk of infection (9).
Custom-made prostheses, starting from computed tomography (CT) scan data, were designed and produced in biomimetic porous HA to fit each patient. The main purpose of this study was to review the postmarketing data for cranioplasty carried out with custom-made prostheses based on biomimetic porous HA. To evaluate the clinical efficacy correlated to the properties of custom-made prostheses based on biomimetic porous HA, characterization data based on physicochemical analysis and mechanical performance were also reported.
We also analyzed in detail any complications related to the physical and mechanical properties of the prosthesis, to evaluate the clinical performance of biomimetic porous HA as an effective solution for cranial reconstruction.
Material and Methods
Prosthesis Production
The porous prostheses were prepared by imbibition: cellulose sponges with controlled pore morphology were treated in a HA slurry to obtain the biomimetic porous structures. The organic matrix was totally eliminated during the thermal treatment process. Then the prefinished prostheses were machined by a computer numerical control (CNC) machine tool to achieve the custom design of the prosthesis specific to the patient.
Prosthesis Characterization
Physicochemical, morphological and mechanical characterizations of the final components were carried out. The total porosity of sintered bioceramic was determined by volume to weight ratio and expressed in term of volume percentage. The formation phase in the bioceramics was examined by X-ray diffraction (XRD) using a Bruker D8 Advance (SN 204459). The XRD analysis was performed in the range of 10°-80° (2Θ), counting 1 second per step (real time) of 0.04°. Power X-ray tube: 40 kV and 30 mA; and approximately 1 g of powder is required for the analysis. The presence and size of pores were determined by scanning electron microscope (SEM; Leo 438-VP).
Mechanical Characterization
In the first application of the prosthetic replacement, it is recommended to use a Mayfield headboard in addition, to allow a correct use of neuronavigator; this also provides greater accuracy in the phase of the separation or demolition of the cranial bone. If there were to be a lack of uniformity between the prosthesis and the bone margins of the patient, there would be the possibility to dissect the margins (10, 11) with the use of a diamond bur. In the operating phase, the need can arise to apply a series of normal loads on different points of the prosthesis due to the formation of intracranial pressure in the patient. If this is not the case, the installation can be done by applying gentle pressure (normal force) on the midpoint of the prosthesis.
During the design phase, research was carried out on the most suitable equipment and procedure to be used for the compression test of the CustomBone Service prosthesis.
It was decided to focus the study on the application of a normal force on the midpoint of the prosthesis, since it was not possible to apply isometric forces on nonplanar surfaces.
A tampon was designed that reflects the thumb of a surgeon and would be as adaptable as possible to all of the prostheses. A silicone rubber (25 shore) with an overall diameter of 2.5 cm (1 in = 2.54 cm) was used (Fig. 1). Using hard plastic tampon was excluded because there was the risk of breaking the tampon itself during each breaking test.
Silicon rubber tampon (25 shore).
To allow a proper distribution of the loading forces during the various tests, an adaptable support for each sample was built. The elaboration through computer-aided design (CAD) software allowed the highlighting of the centroid of the outer surface of the examined sample.
A point on the normal direction of the surface was taken as reference point to orient the prosthesis along the 3 coordinate axes (Fig. 2).
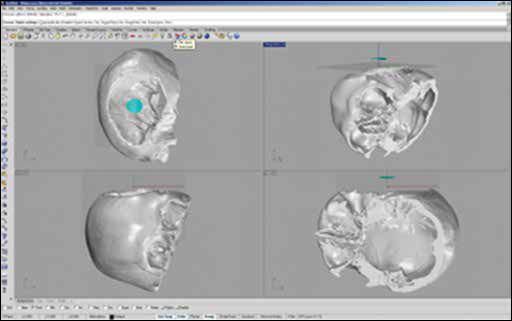
Orientation of the prosthesis along the Cartesian axes.
Once the normal force was calculated, a vertical digital reconstruction of the skull was carried out, and a parallel cutting perpendicular to the center point of the prosthesis was performed. To increase the strength and stiffness of the support, a grid and a containment wall were added inside the support (Fig. 3). The support was built with a 3D printer. Layers of plaster powder wetted with an epoxy glue allowed the entire support to be built layer by layer.
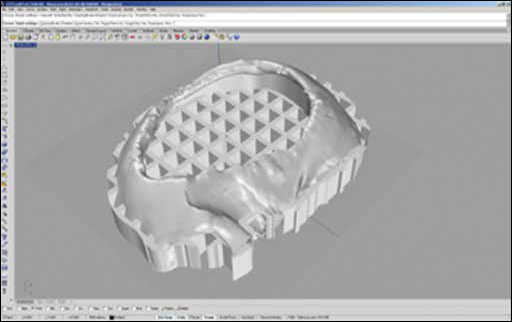
Final support with grill and retaining wall.
For the tests of mechanical breakage, a MTS Insight compression press with a loading cell of 10 kN, full scale, was used (Fig. 4). Sixty prostheses were examined mechanically they were twice if compared to the number of samples used for the test carried out for flexural tests by ENEA. The reason for this was the apparent heterogeneity of forms. It was decided to consider different types of custom-made prostheses as they were represented by the mechanics of the Shell (11). The theory here is quite complex due to the nonuniform state of stress.
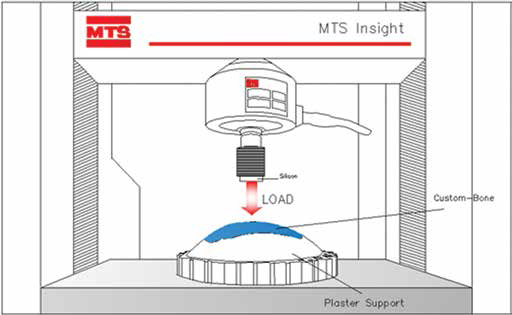
Computer-aided design (CAD) breaking test schema.
The test samples had a surface area of between 66 and 170 cm2. Every single prosthesis was inspected and controlled with the aid of a Dinolite Dino Capture Digital Microscope able to capture images with a resolution of 640×480 pixels with ×25 optical zoom, so that they reveal every imperfection.
The breaking test was conducted with mechanical compression on a single support point (Fig. 4) thus applying a normal force. The loading cell was preloaded with a pressure of 100 N to avoid any interruptions due to background noise. Once pressure was applied, an instant failure of the prosthesis was expected. This resulted in the development of 2 different cases: the occurrence of a first peak of interruption or a total breaking of the prosthesis. In the first case, a verification of the prosthesis was carried out trying to highlight the formation of new cracks or eventual breakage. Otherwise, the prosthesis was relocated inside the press and recharged for a new breaking test of the actual breakage.
Postmarketing Data
With each prosthesis, a questionnaire was sent so that the surgeon could fill it in at the end of the intervention and send it to the manufacturer to carry out surveillance of intraoperative events (MDD 93/42/EEC - Annex X and MEDDEV 2.12/2). In the above-mentioned questionnaire, surgeons were asked to indicate, for each installation performed, intrasurgical observations and feedback on the product (packaging, usability, etc.). All reported postoperative events were recorded using a form in which it was asked to indicate causes, actions taken and possible correlation between the event and the prosthesis. If these postoperative events were related or were suspected to be related to the prosthesis, the surgeon was asked to fill in a further report.
Reported complications were defined as any adverse postoperative event that occurred with the implant that did not allow it to fulfill its proper function. Surgery-related adverse events which happened with the patients, such as postoperative hematoma, brain edema and ischemia, hydrocephalus, seizures or any other cause of neurological worsening, were not taken into account because of their total irrelevance for the prosthesis behavior. Preoperative adverse events were considered to be those concerning all of the events from shipping the prosthesis to its arrival in the operating room. Intraoperative events, however, referred to problems that occurred during surgery.
Postoperative reports were divided, following the directions of the aforementioned directive on the supervision of medical prostheses, into related events (incidents) and unrelated events – i.e., those that did not fall within the definition of incident.
All of the data collection forms reporting postoperative material related complications collected by the company were analyzed and treated in compliance with MDD 93/42/ECC Art. 8 and following current medical device vigilance regulations (MEDDEV 2.12/1).
Results
Physicochemical and Morphological Data
The XRD analysis clarified the HA phase (Fig. 5). Quantitative analysis of the crystalline HA showed a crystalline HA amount of 98 +/- 2 (vol.%), and the Ca/P molar ratio (molar ratio) was determined to be 1.67±0.02.
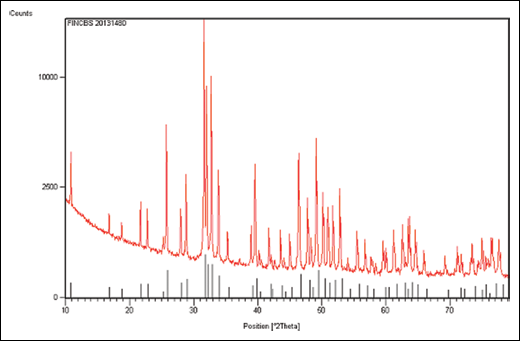
X-ray diffraction (XRD) analysis of batch 20131480 (upper) compared with hydroxyapatite pattern (lower, ref. code 09-0432).
SEM images showed the presence of macroporosities and microporosities in the bioceramic prosthesis. Microstructures of the bioceramics examined on the surfaces are displayed in Figures 6 and 7. The macroporosities (Fig. 6a, b) and the microporosities (Fig. 7a, b) can be observed.

a, b) SEM pictures of the macrostructures of sintered porous hydroxyapatite.

a, b) SEM pictures of the microstructures of sintered porous hydroxyapatite.
Mechanical Data
Through several simulations, it was shown that a surgeon during installation of a custom made prosthesis applies an average force of ≅78 N, but never more than 118 N.
From the data shown in Table I, it can be seen that the maximum value reached for each type of surface clearly exceeded the maximum value that is applied by a surgeon during installation.
RESULTS OF BREAKAGE TESTS OF PROSTHESES
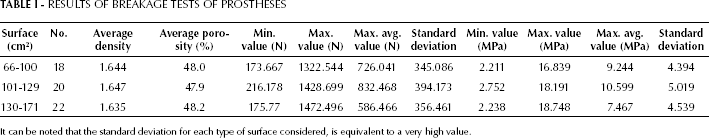
It can be noted that the standard deviation for each type of surface considered, is equivalent to a very high value.
ULTIMATE COMPRESSIVE STRENGTH OF BONE AND CARTILAGE
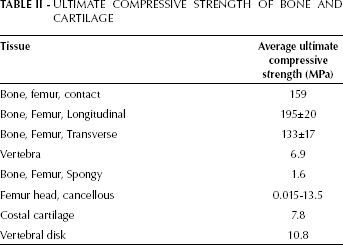
Performing a comparison of the values obtained in the breaking tests, with breakage of different types of human bones, it can be seen that these present a higher resistance than the prosthesis made from porous HA (Tabs. II and III). The reason for this is that human bone is composed of a large amount of collagen. This major feature provides high elasticity to the bone, thus avoiding the occurrence of any pseudoplastic-type breakage during a crash. The prosthesis instead, being build with ceramic components in porous HA, will suffer therefore a fragile-type fracture with a sharp break.
COMPARISON OF CHARACTERISTICS OF POROUS HYDROXYAPATITE IN RELATION TO CORTICAL BONE AND CANCELLOUS BONE (13)
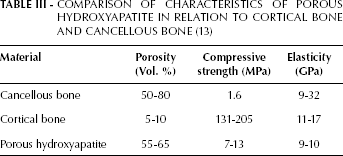
Comparing the compression resistance between components in porous HA implants and Custom made prosthesis (12), it is noted that the value of breakage is very similar.
Postmarketing Data
From 1 November 1997 to 30 April 2013, 2697 patients underwent a cranioplasty operation with the implants of 2900 custom-made porous HA prostheses (CustomBone Service/Fin-Ceramica Faenza). The implants were distributed almost entirely in Europe; only 32 implants were carried out in the rest of the world.
Regarding the total population:
The pediatric population included 128 boys and 67 girls between the ages of 0 and 14 years;
The adult population included 1612 men (66%) and 840 women (34%);
There were data for 50 patients of unknown age.
In 602 (21%) patients, the CustomBone Service prosthesis was the second-line treatment (13% due to the autologous bone resorption and 8% caused by rejection of other materials), and therefore their data were not correlated directly to the etiology of the initial event.
Furthermore, the choice of using the custom-made prosthesis made with porous bioceramic was attributed mainly to decompression (54%) and tumor removal (10%) compared to 65% of traumatic cases and a 15% of tumoral cases.
One hundred forty-three patients had wide and complex lacunae and required a double prosthesis to reconstruct the necessary curvature; 57 of these (40%) were bifrontal lesions, 34 (24%) were frontal-parietal-temporal, and 52 (36%) were located elsewhere.
From 2900 procedures performed, we received 64 intraoperative reports, equal to 2.21% of total interventions. Of the reports received, most of these were because of breakage of the prosthesis during implantation ((48 implants: 1,66%) of the total) or due to an error in sizing the prosthesis (6 implants: 0,21%) even though the prosthesis conformed to the design approved by the surgeon. Only in 13 cases were other solutions used, therefore patients who received a HA prosthesis were found to number 2684.
On all of the 2887 implanted prostheses, 102 events (3.53%) regarding a postoperative occurrence were reported. Of these, only for 3 was it established that the nature of the event should be classified as an incident (1 was attributed to a premature fracture, 2 a spontaneous dislocation), while the other 99 cases were unrelated events and distributed as follows:
51 infections (1.77% of the total)
35 fractures (1.21%)
5 mobilizations (0.17%)
4 spontaneous dislocations due to an extradural hematoma (0.14%)
4 tumor recurrences (0.14%).
In 21 of these cases, the patients were reported to have experienced complications such as to lead to the choice of another therapeutic solution.
If we analyze the 51 cases of infection, we can see that 13 led to the explantation of the prosthesis because of the choice of other methodologies or because it was decided to leave the gap. Among others, 4 were awaiting for back-up custom-made prosthesis implants, 5 were successfully treated in situ with antibiotics, 1 was treated with a reoperation and repositioning of the prosthesis, while 28 had a positive outcome with the removal and substitution with a back-up custom-made prosthesis after treatment of the infection. The infections were classified as not correlated events because, for all of these cases, other reasons were given by the surgeons.
In 2 cases, the removal was followed by the death of the patient for other independent complications. Finally, for 8 patients, the surgeon opted for another solution.
Of the 5 cases of mobilization recorded, 1 was solved with implantation of the back-up custom-made prosthesis, 3 with repositioning the prosthesis, while in the last, in which mobilization was not a dislocation but only a displacement of a few millimeters, was solved with conservative treatment. The 4 events reported for spontaneous dislocation due to an extradural hematoma were solved using the back-up custom-made prosthesis, as was the case for all of the other cases of tumor recurrence.
Focusing our analysis on the events characterized by fractures, if it appears quite intuitive that the 35 fractures appeared after second trauma and had no correlation with the prosthesis, it is useful to note that, of these, 12 were treated conservatively: 6 with evidence of self-repair and 6 with monitoring, waiting for self-repair; 12 were removed and replaced with the back-up custom-made prosthesis. At the end of this study, one patient was still pending to receive an implant.
In addition to this, we can observe that about 71% (25/35) of these fractures took place in males; 80% (28/35) appeared as a result of trauma and 51% (20/35) as a result of an operation for decompression. The distribution of these events was fairly uniform, showing a low correlation with the anatomical localization. Furthermore, the majority (9/35 [26%]) were observed on the frontal-parietal-temporal zone. We can also note that more than 70% of the fractures are present in patients up to 30 years of age and that for the age range 0-40 years, the percentage rises to over 80%. The average age of patients in whom a prosthesis fracture occurred was approximately 24 years.
Patients more exposed to adverse events following a cranioplasty were likely to be children. In fact, the incidence rate rose significantly for pediatric patients, and, out of 208 implants performed on boys and girls between 0 and 14 years, 8 fractures were reported, 5 infections and 1 mobilization, for a total of 14 adverse events, which represented 6.73% of the total. This figure should be compared with adverse clinical events seen in adult patients (15 years and older): in adult patients, more types of adverse clinical events were found (premature fracture, spontaneous dislocation, extradural hematoma and tumor reoccurrence, in addition to the aforementioned), but out of 2,635 implants, adverse clinical events occurred in 85 (3.23%). So, proportionally, adverse events in pediatric patients were more than twice those in adults.
These data confirm, as reported in the instructions for the use of custom-made porous HA prostheses, that they are not indicated for the reconstruction of large and complex bone voids in pediatric patients younger than 7 years, because the risk of an adverse event rises further (5 out of 60 implants, which is 8.33%).
However, many surgeons have still chosen this solution for pediatric patients because of its intrinsic characteristics. In fact, according to a recent study (14), the only material that caused a low incidence of adverse events was titanium (4%), which is not advisable for use with pediatric patients, because of its rigidity. So while a custom-made prosthesis in porous HA has an incidence rate for adverse events in pediatric patients of 6.73%, it is, however, more acceptable than other materials with higher incidence rates for adverse events, such as polymethylmethacrylate (PMMA; 9.6%), autologous bone (14.9%) or cements (26.7%).
It is also interesting to study the etiology of, and underline the fact that, if for some diseases the incidence rate is similar in children and adults, for others, it varies considerably, with very different percentage rates for patients operated on due to malformations (15.38% in children and 0.99% in adults), or due to vascular problems (2.40% in children and 11.08% in adults), or even patients who are operated on for the removal of a tumor (10.10% in children and 15.83% in adults). In contrast, there are similar percentage rates for traumatized patients (64.90% in children and 65.05% in adults) and in patients operated on due to other reasons (7.21% in children and 7.02% in adults).
Discussion
Autologous bone is still today considered the gold standard for the repair of craniolacunia because of its tolerability and biological properties. However, its use presents some, as-yet-unresolved problems such as lack of availability, resorption and the unsatisfactory aesthetic results in some cases. The unavailability of the bone material may be due to trauma with multifragmentary skull fractures that cannot be reconstructed and preserved, or, in cases of skull destruction, due to lesions.
Furthermore the resorption of autologous bone can occur following repositioning at the cranial level and has a variable incidence in the literature ranging from 2% to 50% (15, 16), especially frequent in the pediatric population (17, 18), or it occurs when the bone is preserved in the subcutaneous adipose tissue at the abdominal level (19). Finally, because of the dimensions of the operculum, which do not coincide with the bone lacuna, the use of autologous bone can be encumbered by a lack of fusion and by an unsatisfactory aesthetic result.
To remedy these problems, many safe and reliable materials have been developed in recent decades that can be used in craniolacunia, such as, for instance, PMMA (20-21-22) and porous polyethylene (23).
The postmarketing data enable us to evaluate the efficacy of custom-made prostheses in porous HA for the reconstruction of cranial bone defects due to trauma or associated with elective procedures such as prosthesis revision, malformation and bone tumor treatment.
The slight differences existing between the percentage of adverse events that occurred in patients undergoing first-line treatment (3.42%) and those undergoing second-line treatment (3.97%) makes bioceramic porous HA, in our opinion, the material of choice in those cases in which another material has already failed, in situations that are often complex and that occur in patients with a high number of morbidities.
Conclusion
The mechanical properties of the bone are determined by the same methods used to study similar properties of metals (24). The breaking test of the implants for custom-made prostheses for cranial defects showed that all samples tested and brought to breaking, exceeded the maximum value that a surgeon applies during installation of the prosthesis. In Table I it was noted that the average value of resistance is greater in prostheses that presented surfaces between 100 and 129 cm2. Prostheses with very large surface curvature or with complex geometry, such as a front prosthesis, present difficulties in the distribution of compressive forces and therefore show a greater propensity to lower resistance to breakage. Comparing the values obtained between bars of porous HA and custom-made prosthesis showed that the values for breaking were similar to each other. This makes it clear that the resistance of a custom-made prosthesis depends mainly on the specific geometry.
All of these factors that emerged from the study of the mechanical characterization data were confirmed by the analysis of data recorded from postmarketing surveillance, in particular with regard to the events related to fractures of the prostheses.
The extensive series of cranioplasties carried out with only one material but never reported in the literature demonstrate that porous HA is a safe and effective material that is well-tolerated in adult and pediatric patients and that it meets the requirements necessary to cover a craniolacunia. The initial fragility (lesser strength) of the prostheses has led to a correction in the design, to the manufacture of a thicker prosthesis and to a scrupulous refinement of the surgical technique that must be learned by surgeons before undertaking cranioplasty operations.
This study, carried out on postmarketing data, showed clinical evidence for the efficacy of custom-made prostheses based on porous HA, in the reconstruction and bone integration (25, 26) of large cranial bone defects in humans. Results from this study suggest that such materials represent a safe and predictable alternative for autografts and allografts and synthetic materials such as metals, polymers and cements. Knowledge of the bioceramic properties and application of surgical techniques are critical aspects for good clinical outcomes.
In addition, this study aimed to lead to a different conception of implants in pediatric patients – to move from a clear differentiation between patients who are older or younger than 7 years, to a system that allows, as main solution the use of CustomBone Service prosthesis, with due exceptions involving pediatric patients in whom it is more appropriate to select a different surgical solution.
Footnotes
Acknowledgments
For their scientific assistance, the authors would like to thank Dr. G. Celotti (ISTEC-CNR, Faenza) for XRD and Drs. G. Raiteri and M. Scafè (ENEA, Faenza) for mechanical testing (flexural test).
