Abstract
Prosthetic joint infection (PJI) is a serious and potentially devastating complication of arthroplasty. Prior arthroplasty, immunosuppression, severe comorbid conditions, and prolonged surgical duration are important risk factors for PJI. More than half of the cases of PJI are caused by Staphylococcus aureus and coagulase-negative staphylococci. The biofilm plays a central role in its pathogenesis. The diagnosis of PJI requires the presence of purulence, sinus tract, evidence of inflammation on histopathology, or positive microbiologic cultures. The use of diagnostic imaging techniques is generally limited but may be helpful in selected cases. The most effective way to prevent PJI is to optimize the health of patients, using antibiotic prophylaxis in a proper and timely fashion. Management of PJI frequently requires removal of all hardware and administration of intravenous antibiotics. This review summarizes and analyzes the results of previous reports of PJI and assesses the prevention and management of this important entity.
Introduction
Prosthetic joint replacement surgery (arthroplasty) is considered one of the most successful orthopedic procedures and can lead to pain relief, restoration of function, and improved quality of life in many patients with arthritis (1). It is estimated that 600 000 primary arthroplasties and 70 000 joints revision were done in the United States in 2003. Over 4 million joint surgeries are expected to be performed by the year 2026 (2-3).
Infection of prosthetic joints constitutes a significant, challenging complication of arthroplasty. The rates of prosthetic joint infection (PJI) after primary procedures range from 1% to 9% (<1% in hip and shoulder prostheses, <2% in knee prosthesis, and <9% in elbow prostheses) (4-6). The rates of PJI are significantly higher after revision procedures (<40%) (5). In 2009, the average cost for treating PJI in the United States was $30,300 for primary hip procedures and $93,600 for hip revision procedure (7). The extrapolated annual cost of PJI in the United States alone ranges from $360 million to $730 million (4). Prosthetic joint infections are also associated with high morbidity, the need for repeat surgical procedures, prolonged hospitalization, and occasional death (4-5).
Clinical manifestations and definitions
Joint pain, erythema, swelling, warmth, and fever are all suggestive of PJI but are not specific. Differential diagnosis of PJI should include fracture, dislocation, prosthesis loosening, hemarthrosis, gout, synovitis, and osteolysis (4). Constant pain is suggestive of an infectious etiology, while pain with motion indicates a mechanical process (8).
The Infectious Disease Society of America (IDSA) defines PJI as: A) the presence of a sinus tract communicating with the prosthesis (definite evidence, sensitivity 20-30%, specificity 100%); B) the presence of purulence surrounding the prosthesis without other identifiable causes (definite evidence, sensitivity 20-30%, specificity 100%); C) the presence of acute inflammation on histopathologic exam at the time of surgery (highly suggestive evidence, sensitivity 95-98%, specificity 98-99%); or D) at least two positive intraoperative cultures or a combination of aspiration and intraoperative cultures yielding the same organism (definite evidence, sensitivity 60-85%, specificity 92-97%) (5, 9). The growth of Staphylococcus aureus in a single intraoperative culture or a joint aspiration could also represent PJI. It is important to note that PJI can still be present even if all of the above criteria are not met. Clinical judgment should be used to determine if the patient had PJI after reviewing all pertinent data (9).
PJIs can be classified according to the onset of signs and symptoms after implantation into early infection (within 3 months after surgery), delayed infection (3-24 months) and late infection (>24 months) (3, 5). The infection can be acquired from: A) the surgical wound (perioperative route); B) a distant focus including oral cavity, lung, gut, urinary tract, or skin (hematogenous or lymphogenous route); or C) contiguous foci including soft tissue, osteomyelitis, or septic arthritis (Per continuitatem route) (5).
Microbiology
Staphylococcus aureus and coagulase-negative staphylococci (CoNS) are the most common organisms responsible for PJI (~50%) (4, 10). In patients with rheumatoid arthritis, S. aureus is the most common pathogen (37%) (11). Polymicrobial infections constitute 20% of cases of PJI (12). Other bacterial pathogens include gram-negative organisms, streptococci, enterococci, and obligate anaerobes. Corynebacteria, propionibacteria, and Bacillus spp., which are typically considered contaminants, have been reported to cause PJI (4). Propionibacterium acne is a prominent cause of PJI after shoulder arthroplasty (13-14).
Prosthetic joint infections are occasionally caused by fungi (particularly Candida) and mycobacteria (M. tuberculosis and rapidly growing mycobacteria) (15-19). Occasionally, patients with prior arthroplasty can develop clinical signs and symptoms of PJI without retrieval of organisms from surgical or aspirate specimens. In one study, 7% of cases with PJI were culture negative and had similar outcomes compared to culture positive cases of patients with PJI. Most cases had prior antibiotic therapy (67%) (20).
Pathogenesis
Bacteria are introduced to the joint from the surgical wound during or after the procedure, via hematogenous or lymphogenous route, or by contiguous spread (5). After attachment of bacteria to prosthetic material, they elaborate a fibrous exopolysaccharide material (glycocalyx) creating biofilms (4, 21) (Fig. 1). The formation of biofilms is a critical step for the development of PJI (22). The indwelling metallic prosthesis and polymethylmethacrylate cement lower the amount of bacteria needed to develop an infection and allow microorganisms to persist on their avascular surfaces evading body defenses and systemic antibiotics (4, 23-24). Also, polymethylmethacrylate cement protects microorganisms by disrupting phagocytic, lymphocytic, and complement functions (25).

Scanning electron micrograph (SEM) of a Staphylococcus biofilm on the inner surface of a needleless connector. Photograph by Janice Carr, Centers for Disease Control and Prevention, Atlanta, GA USA. From (21): Donlan RM. Biofilms and device-associated infections. Emerg Infect Dis. 2001; 7(2): 277-281.
Planktonic bacteria near the surface of the biofilms are metabolically active and can disseminate through the upper surface of the biofilm with the aid of phagocytic enzymes. Deep bacteria are typically inactive. They are protected from phagocytosis and are typically resistant to antimicrobials. Decreased antibiotic penetration, nutrient limitation of biofilm cells, and the adoption of new phenotypes can all explain the inherent resistance of biofilms to antimicrobials (22).
Understanding the role of biofilms in the pathogenesis of PJI can impact both diagnostic and management approaches. Biofilm formation can explain the latency of PJI symptoms after arthroplasty, the difficulty of isolating pathogens from joint aspirates, and the failure of treatment in many patients with prosthesis retention (12).
Risk factors
Several risk factors for PJI have been identified. These include older age, obesity, prior surgeries at the site of prosthesis, rheumatoid arthritis, psoriasis, concurrent malignancies, corticosteroid therapy, recurrent urinary tract infection (UTI), poor nutrition status, and lower socioeconomic status. Additional surgical-related risk factors are perioperative and postoperative nonarticular infections, prior infections of the joint or adjacent bones, prolonged surgery duration, and formation of postoperative hematoma (26-30).
In one case-control study, bacteremia during the previous year, non-surgical trauma to the prosthetic joint, and skin and soft tissue infections were associated with the presence of PJI (31). Another study assessed the risk factors for hip revision due to infection. Male sex, hybrid fixation, using cement without antibiotics, femoral head necrosis, hip fracture, and previous hip inflammatory disease were associated with PJI (32). Nasal colonization with S. aureus has been recently proposed as a risk factor for PJI (33). Furthermore, genetic factors may play a role in the development of PJI. One genetic-association study showed that a functional variant of the gene encoding for interleukin-1 beta (IL1B–511*T allele) predisposed to PJI (34).
A National Nosocomial Index score (NNIS) of one or more is associated with PJI (30). One point is added for each of the following variables: A) contaminated or dirty wound; B) duration of operation >75th percentile; and C) severe systemic disease (American Society of Anesthesiologists preoperative score ≥3) (35). The baseline Mayo Prosthetic Joint Infection Risk Score for the development of PJI has been recently proposed to stratify patients undergoing total hip or total knee arthroplasty. This scoring system may be used in targeting preventive strategies in patients undergoing arthroplasty (36).
DIAGNOSIS
Imaging studies
Table I presents different imaging methods used for diagnosis of PJI. Plain x-rays should be performed in all patients with suspected PJI to rule out other causes of joint pain (including fracture and loosening of the prosthesis) and to aid as a baseline for diagnostic and therapeutic procedures (9). They can demonstrate abnormal lucencies at the bone-cement surface, malpositioning of the prosthesis, periosteal reaction, cement fracture, transcortical sinus tract, or prosthesis movement on stress films (4, 9). These radiologic findings are usually late (3-6 months) and non-specific as they can be seen in aseptic processes (prosthesis fracture and joint dislocation) (37). The use of imaging modalities other than plain x-ray should not be routinely done unless the diagnosis of PJI is unclear and reoperation is not scheduled (9, 38).
IMAGING STUDIES USED FOR THE DIAGNOSIS OF PROSTHETIC JOINT INFECTIONS
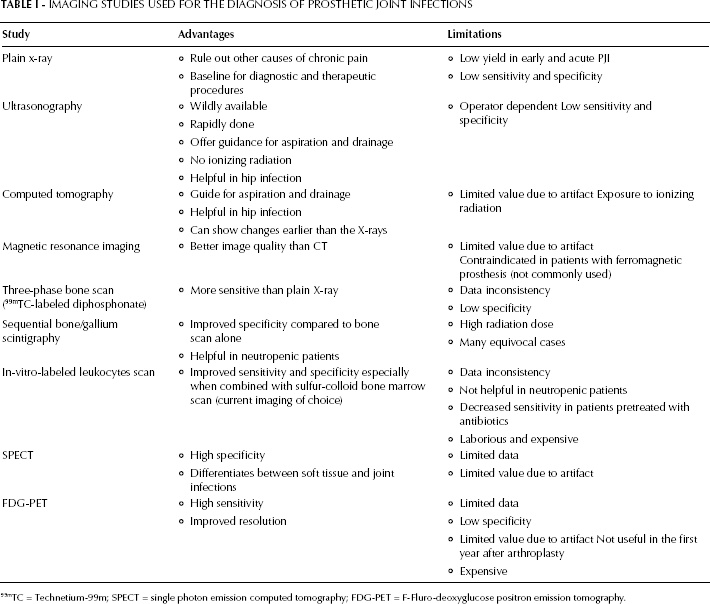
99mTC = Technetium-99m; SPECT = single photon emission computed tomography; FDG-PET = F-Fluro-deoxyglucose positron emission tomography.
Ultrasonography can show fluid collection around the prosthesis and can guide joint aspiration and drainage. It is useful in hip PJI where clinical findings are subtle. It is sometimes difficult to distinguish soft tissue fluid collection from subperiosteal thickening (osteomyelitis) using ultrasonography (40). Computed tomography (CT) may identify subcutaneous abscesses, joint effusions, sinus tracts, bony erosions, and prosthesis loosening (39). Similarly to ultrasonography, CT scans aid in joint aspiration and drainage (40). The use of magnetic resonance imaging (MRI) has been increasing as most recent prosthetic implants are non-ferromagnetic. MRI offers better resolution and can further minimize artifact compared to CT (37, 40).
Radionuclide imaging can be helpful in the diagnosis of PJI but its role is limited due to low specificity. Three-phase bone scintigraphy can show increased uptake of Technetium-99m (99mTC)-labeled diphosphonate on the surface of bone mineral matrix in cases of PJI. However, this can also be seen in cases of sterile inflammation, bone remodeling, fracture, and aseptic prosthetic loosening (37). Sequential bone/gallium scintigraphy technique offers enhanced specificity by comparing spatial distribution congruity of the two scans (41). In vitro -labeled leukocytes scan using indium-111(111In) oxyquinolone or 99mTC- hexamethylpropyleneamine oxime (HMPAO) has been associated with improved accuracy of PJI diagnosis (37, 42). The combination of leukocyte scan with sulfur colloid bone marrow scan offers even better results (43). This method has an overall accuracy of 88% to 98% and is the current imaging procedure of choice (9, 40).
Single photon emission computed tomography (SPECT)/CT scan enables more localization of the radio-tracer uptake compared to other radionuclide imaging techniques (44). The specificity of SPECT/CT is markedly increased when combined with leukocyte scanning (45). Fluro-deoxyglucose positron emission tomography (FDG-PET)/CT offers high resolution imaging with excellent sensitivity for the diagnosis of PJI (46). Other investigational methods include the use of 99mTC-interleukin-8, 111In- Biotin scintigraphy, 99mTc-Ciprofloxacin, 99mTc-recombinant annexin V scintigraphy, radiolabeled synthetic fragments of ubiquicidin, 99mTc-labeled bacteriophages (SPECT) and thymidine kinase (PET). These methods may have a prospective role in diagnosis of PJI (37).
Laboratory studies
Testing for white blood cell count (WBC), erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP) is recommended in all patients with suspected PJI (9, 38). These markers are usually elevated in cases of PJI. However, high false-positive rates are not uncommon, particularly after arthroplasty. The utility of using them is improved when normal baseline labs are available (35). Combining ESR and CRP can lead to better accuracy than using either one of them (47). Different cut-offs have been suggested for the diagnosis of PJI (39, 48). Limited data exist regarding the diagnosis utility of procalcitonin, interleukin-6 (IL-6), and soluble intracellular adhesion molecule-1 (sICAM-1) (39).
Microbiologic studies
Blood cultures should be obtained in patients with suspected PJI if they have fever, concomitant infection elsewhere, infection of cardiovascular devices, or if their infection is due to S. aureus (9).
The specific diagnosis of PJI often requires isolation of organisms from culture of joint fluid or surgical tissues. Arthrocentesis should be performed in all patients with suspected acute PJI unless the infection is evident, surgery is scheduled, and initiation of antibiotics can be delayed until after surgery (9). Arthrocentesis should also be performed in patients with a chronic painful prosthesis with high clinical suspicion of PJI or with elevated ESR or CRP. Aspirated fluid should be sent for cell count, gram stain, aerobic and anaerobic cultures (9, 38). Withholding antibiotics for medically stable patients for at least two weeks before arthrocentesis or surgery is recommended as it leads to higher yield of identifying the pathogen (9).
The suggested cutoff point for WBC in joint fluid to diagnose PJI is >4200 WBC/μl and >80% polymorphonuclear cells (PMN) for hip joints. This cutoff is lower (>3000 WBC/μl) when combined with elevated ESR and CRP (49). The suggested cutoff point for knee joints is >1700 WBC/μl and >65% PMN (50). One colorimetric chemical strip that detects leukocyte esterase has been shown to be predictive of PJI (sensitivity 80.6%, and specificity 100%). This technique is cheap and provides real time results, but has limited utility when blood is present in the synovial fluid (51).
Combining intraoperative synovial fluid culture with periprosthetic tissue culture leads to a high sensitivity (90%) and specificity (100%) (52). Obtaining swab cultures is not recommended as it has lower diagnostic yield (39). At least three surgical samples (preferably six) should be obtained and sent for aerobic and anaerobic cultures. Gram stain of tissue specimens has a low sensitivity (9).
Histopathological studies
Histopathological examination of periprosthetic specimens is an important tool for the diagnosis of PJI. The finding of ≥5 neutrophils per high-power field in x40 magnification is indicative of PJI (sensitivity >80% and specificity >90%) (39). Frozen sections can aid surgeons in deciding whether to perform resection or revision arthroplasty in cases in which perioperative evaluation did not confirm PJI. However, this modality is limited by tissue sampling, and the availability and expertise of the pathologist (9).
Other techniques
The yield of isolating bacteria from biofilm is greater after dislodging them from the explant biofilms using sonication techniques, followed by either culturing or molecular methods (53, 54). Multiplex polymerase chain reaction (PCR) of sonicate fluid is more sensitive in diagnosing PJI (96%) than cultures of sonicate fluid (67%, p=0.016) or periprosthetic tissue (71%, p=0.031) (53). The sensitivity is even greater when combining sonicate fluid PCR with sonicate fluid culture (55, 56). In one study, the sensitivity of combining the two previous tests was reportedly 100%, even in patients who were pretreated with antibiotics (56). Bead-mill process is another novel technique that uses glass beads to agitate and liberate organisms from biofilms. It may shorten time to isolate the organisms and improve the release from biofilms (57).
Automated ribotyping is a rapid and accurate method for diagnosing PJI (58, 59). However, the use of automated ribotyping is limited by the high cost and need for a previous bacterial culture (60, 61). Another recently developed technique is matrix-assisted laser desorption ionization coupled with time of flight analysis mass spectrometry (MALDI-TOF/MS). This technique relies on detecting peptide and protein ions of bacteria through spectra originating from relative charges and masses (61). It can rapidly and reliably identify pathogens responsible for PJI to the species level. Ibis T500 Universal Biosensor (Ibis Biosciences, precursor to the PLEX-ID system; Abbott Laboratories, Chicago, IL, USA) based on PCR-electrospray ionization/mass spectroscopy (PCR-ESI/MS) technique can detect bacteria, viruses, fungi, and protozoa, as well as antimicrobial resistance genes (61, 62). It was evaluated in patients with total knee arthroplasty and was able to detect pathogens in 17/44 (38%) cases of presumed aseptic failure (63).
PREVENTION
Due to the tragic complications of PJI, focusing efforts on prevention should be considered of the highest importance. Table II delineates several key methods for prevention of PJI.
STRATEGIES FOR PREVENTION OF PROSTHETIC JOINT INFECTIONS (PJI)

CAD = Coronary Artery Disease; MRSA = Methicillin-resistant S. aureus.
Data modified from (69): Matar W Y, Jafari SM, Restrepo C, Austin M, Purtill JJ, Parvizi J.
Preventing infection in total joint arthroplasty. J Bone Joint Surg Am. 2010;92 Suppl 2:36-46.
The host
Screening and optimization of patients is an important strategy in preventing PJI (64). Control of dentogingival pathology, obstructive uropathy, and dermatologic conditions before arthrocentesis have been shown to decrease the risk of PJI. Using prognostic tool calculators to assess the risk of PJI can aid surgeons in identifying patients with high risk for treatment failure and to opt for adjusting these factors before arthroplasty (4). Glucose and hemoglobin control, cardiac function optimization, smoking cessation, and improving nutritional status are vital ways to prevent PJI (64, 65). Screening and treatment of asymptomatic bacteriuria has been attempted with limited data as a method to prevent PJI (66).
Antibiotic prophylaxis
The use of prophylactic antibiotics has been shown to decrease the risk of PJI (relative risk 81%, p<0.001). Cefazolin, cefuroxime, or oxacillin are the antibiotics of choice in most but not all institutions (4, 67). The dose of cefazolin should be increased from 1 g to 2 g for patients who weigh >80 kg. The use of clindamycin or vancomycin is appropriate in patients with previous allergy to betalactams. A second dose of cefazolin should be used during prolonged surgeries. Vancomycin is often used in hospitals with high rates of methicillin-resistant S. aureus (MRSA) or in patients who have a history of previous MRSA infection or colonization. Antibiotics should be administered within one hour of surgical incision and should not be continued beyond twenty-four hours (68, 69).
The operating room
Several preventive methods related to the operating room are recommended, including preoperative skin preparation, draping, shorter operative time, addition of antibiotics to cement, and controlling the operating room environment (69, 70).
Other methods
As shown in Table II, several controversial methods have been used for prevention of PJI, but more data are needed. Additionally, few innovative approaches have been proposed. Researchers in the field of biomedical sciences have modified implants to overcome bacterial colonization (71-76). These implants are also known as “smart implants” (71, 72). The implantation of copper and silver ions on Ti6AIV4 alloy surfaces has been shown to stimulate osteoblast proliferation and prevent S. aureus and E. coli colonization (73). Bioglasses doped with gold nanoparticles showed antibacterial properties against S. aureus (74). Another approach is modifying implant surfaces to prevent bacterial adhesion. One study showed that using a poly (ethylene glycol)-based monomolecular layer that adsorbs on titanium surfaces was associated with greater stimulation of bone formation and reduction in bacterial adhesion compared to uncoated titanium surfaces (75).
An anti-adhesin vaccination that targets S. aureus attachment to biofilms and bacterial invasion into osteoblasts has been attempted in animal studies (77). Unfortunately, the efficacy of these vaccines was limited due to the variable expression of surface proteins on S. aureus (78). Other vaccinations that boost Th17 activity or target S. aureus protein A (SPA) are under development (79).
Different enzymes have been shown to disrupt biofilms. Studies in vitro and in nematodes showed that deoxyribonuclease (DNAse) can disrupt immature biofilms, increase susceptibility to antibiotics, and increase survival (80). Other studies showed that dispersin B can disrupt biofilms from vascular catheters in vitro, and can decrease catheter colonization in rabbits (81, 82). Extracellular amylase has been shown to prevent biofilm formation by S. aureus and Vibrio cholerae (83).
MANAGEMENT
Surgical treatment
Surgery is essential in the management of patients with PJI. Multiple surgical options are available, including debridement and retention of the device, one- or two-stage exchange, resection arthroplasty, arthrodesis, and amputation. Many factors can affect the choice of surgery including duration of symptoms, time from arthroplasty, causative organisms and susceptibility pattern, prosthesis loosening, options for reconstructive surgeries, medical comorbidities, surgeon's expertise, and patient's preference (4, 9).
Debridement and retention of the device is a good option for patients who have early PJI (<30 days), short duration of symptoms (<3 weeks), stable prosthesis, and in patients without sinus tract or abscesses (9, 82). Patients with PJI due to MRSA, gram-negative organisms, and patients with S. aureus who are not treated with rifampin combination therapy have higher risk of failure when retention of the device is performed (9, 84-86).
A one-stage exchange involves removal of all hardware material and polymethylmethacrylate cement, debridement of bone and soft tissue, and placement of new prosthesis with antibiotic-impregnated cement (9). This procedure is more commonly performed in Europe than in the United States (3). The indications of this procedure include total hip arthroplasty, good soft tissue and bone stock, easily treated organisms, susceptibility to oral antibiotics with high oral bioavailability, use of antibiotic impregnated cement, and no need for bone grafting (85, 87). Advantages include lower morbidity rate and diminished cost (9).
A two-stage exchange is the procedure of choice in the United States for management of chronic PJI with unstable prosthesis. This procedure involves removal of all hardware and cement, debridement, and placement of antibiotic impregnated cement and spacer. Reimplantation of new prosthesis is usually performed from 2 weeks to several months after removal (9). This procedure is optimal for patients who can tolerate two surgeries and who have adequate bone stock (88). This procedure is a good option for patients with PJI due to MRSA, enterococci, and Candida species (9). Reimplantation after four to six weeks of intravenous antibiotics and two to eight weeks of antibiotic-free period are associated with high success rates (89). In cases of infection by less virulent organisms (i.e., excluding MRSA, enterococci or multi-drug resistant gram-negative organisms), reimplantation can be done as early as two to six weeks after hardware removal (1). Decrease in ESR and CRP can aid in deciding the optimal time of reimplantation (9).
Resection arthroplasty or arthrodesis can be performed in nonambulatory patients, patients with limited bone stock, severe comorbidities precluding major surgeries, failed two-staged therapy and those with difficult-to-treat organisms (9). Amputation is indicated in some cases of necrotizing fasciitis, severe bone loss, and previously failed arthroplasty. The functional outcome of amputation is poor (9, 90, 91).
Medical treatment
Rifampin is the mainstay of therapy in patients with PJI due to S. aureus, especially in those who underwent retention of the device or one-stage prosthesis exchange (12). Rifampin and fluroquinolone have great biofilm penetration, and this combination has been shown to be associated with high rates of successfully curing infection of retained prostheses (92, 93). Rifampin has also been shown to be active against P. acne and Enterococcus faecalis in vitro (94, 95). Rifampin should not be used as a monotherapy due to the high likelihood of developing resistance (96).
Fluroquinolones are also useful in management of PJI (97). However, their efficacy may be limited by induced resistance in vivo, mainly when treating Pseudomonas aeruginosa infections (98). For candidal infections, caspofungin is associated with higher biofilm penetration and drastically reduced biofilm dispersion, as compared with fluconazole (99). A summary of IDSA recommendation for medical treatment of PJI is presented in Table III (9).
ANTIBIOTIC MANAGEMENT OF PROSTHETIC JOINT INFECTIONS

FQ = Fluroquinolone.
May be used in patients with potential of implant loosening and loss of bone stock, and in those who are unsuitable or refuse further surgeries.
May be used for infections due to gram-negative organisms after FQ treatment Modified from (9): Osmon DR, Berbari EF, Berendt AR, et al. Executive summary: diagnosis and management of prosthetic joint infection: clinical practice guidelines by the infectious diseases society of America. Clin Infect Dis. 2013; 56(1): 1-10.
Novel approaches
Recent studies have reported a promising role of biofilm-disrupting agents in the management of PJI. N-acetylcysteine can disrupt mature biofilms by breaking disulfide bonds, and it has bactericidal effect against several pathogens (100). Using cationic polylysine peptide targeting the polysaccharide intercellular adhesin can decrease biofilm stability by disrupting the charge-charge interaction (101). Another approach is using nanotechnology to maximize local release of antimicrobial agents. Carvacrol, an essential oil component, was encapsulated in poly(dl-lactide-coglycolide) nanoparticles. This combination was able to change the elasticity and stability of the biofilm by allowing the delivery of the antibiotic-laden nanoparticles (102). The use of chelating agents such as ethylendiaminetetraacetic acid (EDTA) and desferrioxamine B (DFO) has been associated with decreased growth of S. aureus biofilms. However, the use of lower concentration of DFO resulted in an increased biofilm growth (103).
Photodynamic therapy provides a novel way to kill most pathogens (bacteria, fungi, viruses, and protozoa) and it is not affected by microbial resistance. Photoantimicrobials produce reactive oxygen species after they are applied topically and taken up by pathogens. A single treatment of methylene blue and 670 nanometer non-thermal activating light was associated with >99.9% reduction in MRSA and P. aeruginosa biofilms (104).
Laser generated shockwave can deliver non-thermal pulses that remove biofilms. Two studies have shown reduction of bacteria after using shockwave pulses (105, 106). Finally, the use of metabolite profiles can help shed more light on the pathogenesis of PJI, as well as identify new therapeutic properties (107).
Conclusions
PJI is associated with significant morbidity and mortality. New diagnostic molecular and radiologic techniques have been recently proposed. Modifying implants to incorporate anti-biofilm and antimicrobial agents has a promising future in the field of PJI prevention. The development of biofilm disrupting agents, laser generated shockwave, photodynamic therapies, and metabolomics are new techniques that may aid in management.
