Abstract
Purpose
The objective of this work was to develop composite hydrogels based on poly(ethylene glycol) diacrylate (PEGDA) and collagen (Coll), potentially useful for biomedical applications.
Methods
Semi-interpenetrating polymer networks (semi-IPNs) were obtained by photo-stabilizing aqueous solutions of PEGDA and acrylic acid (AA), in the presence of collagen. Further grafting of the collagen macromolecules to the PEGDA/poly(AA) network was achieved by means of a carbodiimide-mediated crosslinking reaction. The resulting hydrogels were characterized in terms of swelling capability, collagen content and mechanical properties.
Results and Conclusions
The grafting procedure was found to significantly improve the mechanical stability of the IPN hydrogels, due to the establishment of covalent bonding between the PEGDA/poly(AA) and the collagen networks. The suitability of the composite hydrogels to be processed by means of stereolithography (SLA) was also investigated, toward creating biomimetic constructs with complex shapes, which might be useful either as platforms for tissue engineering applications or as tissue mimicking phantoms.
Introduction
Interpenetrating polymer networks (IPNs) are composite systems made up of 2 (or more) independently crosslinked networks, at least one of which is crosslinked in the presence of the other. The intimate entanglements between the 2 polymer networks are well known to enhance the mechanical properties of the single networks, and/or to form a new material showing the key functional properties of both components. IPN hydrogels are particularly attractive for biomedical applications, especially for the creation of biomimetic platforms for tissue engineering, which usually combine the typical advantages of synthetic polymers (i.e., designable and tunable properties) with the intrinsic bioactivity of naturally derived biomaterials (e.g., cell-interactive domains and enzymatic degradability). In particular, polymers derived from the extracellular matrix (ECM) are ideal candidates for the development of semi-IPNs, which differ from IPNs in the fact that one network only is crosslinked (e.g., the synthetic polymer), and formed around uncrosslinked chains of a second polymer (e.g., the ECM macromolecules).
Poly(ethylene glycol) (PEG)–based hydrogels have been widely investigated in the last decade in both in vitro and in vivo applications, especially as tissue engineering templates, due to the resistance of PEG to protein adsorption, which makes it a nonfouling material for the design of scaffolds with specific bioactivity (1-11). Hydrogels are usually obtained by the photocrosslinking of acrylated PEG derivatives in aqueous solution, which is particularly advantageous for the design of injectable hydrogel formulations (12-14), as well as for the possible use of rapid prototyping techniques (e.g. stereolithography [SLA]) and other photopatterning approaches for the design of devices with complex or customized structures (15-18). Bioactive PEG-based hydrogels can be obtained by copolymerizing PEG derivatives with short peptide sequences and/or ECM components, which have been previously modified (e.g., acrylated or methacrylated) to be incorporated into the hydrogel network during the crosslinking reaction (1-9). Alternatively, biomimetic semi-IPNs can be synthesized by crosslinking the PEG derivatives in the presence of unmodified ECM molecules, such as hyaluronic acid (19, 20). In this case, in addition to inducing a given cell–material interaction, the ECM component acts as a spacer in the PEG network, thus modifying the microstructure of the network itself.
The aim of this work was to investigate the creation of biomimetic semi-IPNs and grafted IPNs based on PEG derivatives and collagen (Coll), potentially useful for biomedical applications. Semi-IPNs were synthesized starting from aqueous mixtures of poly(ethylene glycol) diacrylate (PEGDA), acrylic acid (AA) and collagen as bioactive molecule. The effect of collagen on the PEGDA/poly(AA) network formed in a photocrosslinking reaction was assessed in terms of swelling behavior and mechanical properties. The use of SLA for the production of hydrogels with complex shapes was also investigated. Further grafting of the collagen macromolecules to the PEGDA/poly(AA) network was explored by using a carbodiimide-mediated crosslinking reaction (21-23).
Materials and Methods
All chemicals used in this study were purchased from Sigma-Aldrich (Milan, Italy), unless otherwise stated, and used as received.
Hydrogel Synthesis
To develop composite hydrogels based on PEGDA and Coll, a 2-step process was employed which involved: (a) the synthesis of semi-IPNs by photostabilizing aqueous solutions of PEGDA (molecular weight 700 Da) and AA, in the presence of collagen; (b) the grafting of collagen to the PEGDA/poly(AA) network by means of a carbodiimide (EDAC)–mediated crosslinking reaction.
Semi-IPNs
An aqueous suspension of Type I collagen from calf skin (3% w/v, Semed S collagen; Kensey Nash Corporation) was prepared. PEGDA, AA and an acrylate-soluble photoinitiator (Darocur 1173; Basf) were dissolved in distilled water. A given volume of collagen suspension was then added to the above solution and thoroughly mixed, in order to obtain a homogeneous polymer mixture with fixed PEGDA and AA amounts and variable collagen concentrations, according to the following: PEGDA (10% w/v), AA (0.2% v/v), Darocur 1173 (0.3% v/v) and collagen at a concentration of either 0.9% or 1.8% w/v. Control solutions devoid of collagen were also prepared. The concentrations of PEGDA, AA and Darocur 1173 were chosen based on a previous protocol (10), which was slightly modified to reduce the AA amount. With the concentrations used in this study, the molar ratio between PEGDA and AA was 5:1. Hydrogel samples were then obtained by photostabilizing 1.5 mL of the polymer mixture in a 35-mm Petri dish by exposure to UV light (365 nm) for 10 minutes. The average distance between the sample surface and the UV lamp was 3.5 cm.
The resulting semi-IPNs were either subjected to a further grafting procedure, as described in the following, or directly washed in excess distilled water (50 mL) overnight, to remove unreacted chemicals.
A synoptic overview of all the hydrogel formulations prepared in this work (semi-IPNs, grafted IPNs and control samples) is given in Table I. The crosslinking and the characterization of hydrogels based on PEGDA only (i.e., not containing AA) was not investigated, because it has already been reported in the literature (10, 24). The formation of a 3-dimensional hydrogel network for the different formulations tested was itself representative of the reactivity of PEGDA, which was also confirmed by FT-Raman spectra (Fig. 1).
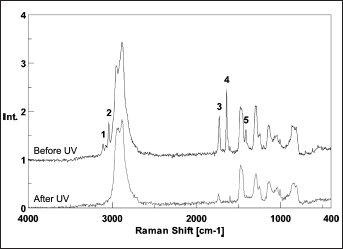
FT-Raman spectra of pure poly(ethylene glycol) diacrylate (PEGDA), before and after UV exposure (365 nm, 1 minute) in the presence of 3% w/w of Darocur 1173. Spectra were acquired averaging on 64 scans and using a laser (1064 nm) power of 390 mW. The characteristic peaks of the acrylate moieties of PEGDA, highlighted in the upper curve, either disappear or are significantly reduced following UV exposure. Highlighted peaks: 1 (3,109 cm−1) and 2 (3,041 cm−1), both ascribable to =CH2; 3 (1,728 cm−1) related to C=O; 4 (1,641 cm−1) related to C=C; 5 (1,413 cm−1), ascribable to CH2=CH.
HYDROGEL FORMULATIONS: PEGDA (10% w/v), AA (0.2% v/v), DAROCUR 1173 (0.3% v/v) WERE USED FOR ALL SAMPLES
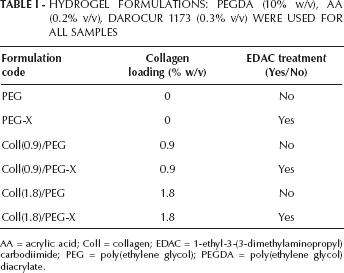
AA = acrylic acid; Coll = collagen; EDAC = 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide; PEG = poly(ethylene glycol); PEGDA = poly(ethylene glycol) diacrylate.
From Semi-IPNs to Grafted IPNs
To induce a chemical grafting of collagen to the PEGDA/poly(AA) network, the semi-IPNs described above were subjected to a crosslinking procedure activated by exposure to water-soluble 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDAC). EDAC is known to induce the formation of peptide linkages (22, 23), which, in this study, may form between the amine groups of collagen and the carboxylic groups brought by either the PEGDA/poly(AA) network or the collagen itself (Fig. 2).
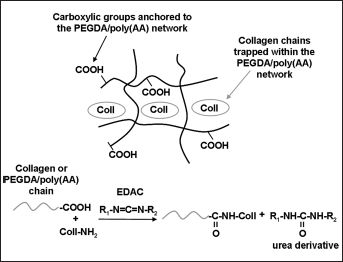
Synthesis of composite PEGDA/Coll hydrogels: a) Schematic representation of semi-IPNs based on PEGDA/poly(AA) and collagen; b) Subsequent grafting reaction of collagen to the PEGDA/poly(AA) network induced by EDAC. AA = acrylic acid; Coll = collagen; EDAC = 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide; IPN = interpenetrating polymer network; PEGDA = poly(ethylene glycol) diacrylate.
Hydrogel samples were subjected to the grafting treatment by placing them in a water solution containing EDAC (14 mM) and N-hydroxysuccinimide (NHS, 5.5 mM), the latter working as a catalyst to improve the rate and the efficacy of the crosslinking reaction (22, 23). NHS is indeed well known to convert the intermediate O-acylisourea group, created between a carboxylic group and EDAC, into an NHS-activated carboxylic group, which is much more reactive toward free amine groups and less sensitive to hydrolysis (23). The needed volume of the EDAC/NHS solution for each sample was calculated by considering a molar ratio of 5:1 between the EDAC and the total amount of carboxylic groups (Coll + AA) theoretically present in each sample (23). The crosslinking reaction was run for 3 hours, after which the samples were soaked in distilled water (50 mL) overnight, to remove unreacted chemicals and/or by-products.
Assessment of Collagen Content
To assess the amount of collagen retained in swollen IPNs, as well as the physical and/or chemical interaction between the collagen and the PEGDA/poly(AA) network in both semi-IPNs and grafted IPNs, an aliquot (1 mL) of the distilled water used to wash the samples (50 mL) was analyzed to detect any protein traces, by means of the bicinchoninic acid (BCA) protein quantification assay. BCA is known to form a purple-blue complex with Cu+1 in alkaline environments, where Cu+1 is obtained from the reduction of Cu+2 achieved in the presence of a protein and proportional to the amount of protein present (25). The BCA assay used in this study had a linear sensitivity range of 200-1,000 μg/mL of protein. Following the manufacturer's protocol, the content of collagen released in the washing solution was measured by reading the absorbance of the purple-blue complex at 562 nm, against a standard curve. Measurements were run in triplicate. The amount of collagen released from each sample was then expressed as the percentage of the theoretical protein loading used for the hydrogel synthesis. Such a loading, calculated from the volume of the starting polymer mixture (1.5 mL) and the collagen concentration, was equal to 13.5 mg and 27 mg for Coll(0.9)/PEG and Coll(1.8)/PEG samples, respectively.
Swelling Measurements
The hydrogel swelling capability in distilled water was assessed by measuring the mass swelling ratio, defined as follows:

where Msw is the mass of the swollen hydrogel sample and Md the mass of the dried sample. For the swelling measurements, each hydrogel type described above was synthesized in triplicate, and from each sample three 6-mm-diameter disks were punched out. After washing in distilled water overnight, the weight of the disks was measured to determine Msw. Before weighing, the hydrogel samples were gently blotted with soft paper to remove excess water from their surface. The samples were then air-dried at room temperature under a chemical hood for 24-48 hours, and weighed again to determine Md. Both Msw and Md values, estimated as described above, did account for the release of any unreacted chemicals and/or collagen from the hydrogel network during the washing, and thus were considered as representative of the true water holding capacity of the samples.
To verify whether the hydrogel formulations were sensitive to ionic strength variations, the swelling ratio of the hydrogels was also assessed in water solutions at different NaCl concentrations (10–4, 10–3, 10–2, 10–1 and 1 M) (26). Results were averaged over 3 independent measurements.
Uniaxial compression tests
After swelling in distilled water for 24 hours, semi-IPN and grafted IPN samples were subjected to uniaxial compression at 20°C by means of a parallel plate rheometer (ARES; Scientific Rheometric), as described previously (27). Such tests were carried out both to assess the mechanical properties of the samples and to verify if the theory of rubber elasticity could be applied to them to evaluate their crosslink density.
Indeed, in the case of a perfect rubber-like polymer network subjected to small uniaxial deformations (i.e., for negligible volume changes either in compression or elongation), the following equation derived by Flory (28) might be used to estimate the elastically effective degree of crosslinking:

where σ is the uniaxial stress,
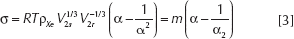
For a rubber-like polymer network and for a < 1, the plot of σ against (α-1/α2) is thus linear, with a slope
It can be demonstrated (28) that the elastic modulus

Considering that the Poisson's ratio
In this study, thin disks of swollen samples (25-mm diameter, n=3-4 for each hydrogel type) were accurately cut and positioned between the parallel plates of the rheometer. The gap was adjusted starting from the original sample height; gap sizes for swollen gels were generally between 0.7 and 0.9 mm. To perform uniaxial compression, a multiple extension mode test was selected, working in a transient motor mode. A constant velocity equal to 0.001 mm/s was then imposed on the upper plate to start the measurement. The evolution of the normal force was recorded as a function of the gap between the plates, until reaching α values of about 0.7, at which point the measurement was stopped. Equation 3 was then applied only in the range of α values between 0.9 and 1.
Since collagen is known to show a rubber-like behavior only if denatured (30), for each sample containing collagen, a hydrogel disk was preliminarily tested both before and after a collagen denaturation treatment, to assess the effect of such a treatment on the uniaxial compression response. The denaturation was performed by soaking the samples in distilled water at 90°C for 15 minutes. Denatured samples were then allowed to equilibrate in distilled water at 20°C, and subjected to compression as described above. The compression curves of a given sample, before and after the denaturation, were almost overlapping, thus suggesting that the mechanical properties of the hydrogels were not affected by the collagen denaturation, at least at the low collagen concentrations used.
Morphological analysis
Scanning electron microscopy (SEM) was used to assess the morphology of hydrogels. Samples swollen in distilled water were lyophilized and then directly observed, both in longitudinal and transverse sections, in a variable pressure mode with a Zeiss EVO scanning electron microscope.
Stereolithography of hydrogels
An SLA 250 device (3D System, Valencia, CA, USA), operating with a He-Cd laser emitting at 325 nm, was modified with a custom-designed elevator-driven build table, described in detail elsewhere (31). This system was designed to use a reduced volume (from 0.3 up to 3 kg) of polymeric solution or suspension compared with that of the standard SLA tank (43 kg). The tank was replaced with a small glass beaker fixed on an adjustable support. The device allowed an accurate setting of the liquid level in order to work in the same conditions as the standard SLA table. The recoating process was deactivated due to the low viscosity of the PEGDA/Coll water mixtures. The SLA construction of hydrogels with a meniscus-like shape was investigated. A minipig meniscus, kindly supplied by the San Raffaele Hospital (Milan, Italy), was used as a reference for a simplified computer-aided design (CAD) model (Fig. 3), then converted to .stl format for SLA processing. A low collagen concentration in the polymer mixture (0.9% w/v) was chosen to reduce the light scattering induced by the collagen molecules during the SLA laser photopolymerization (PEGDA/Coll mixtures are characterized by a whitish color).
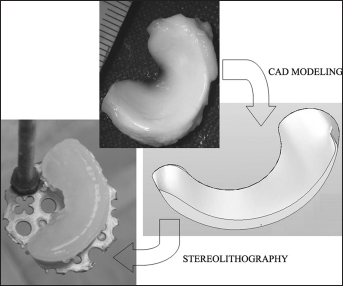
Building of a meniscus-shaped construct by means of stereolithography. Starting from a minipig meniscus (top photograph), a corresponding computer-aided design (CAD) model was created (middle image), which was then processed to build a poly(ethylene glycol) diacrylate (PEGDA)/collagen meniscus by means of stereolithography (bottom left photograph).
Statistical Analysis
Data are expressed as means ± standard deviation (SD). Analysis of variance (ANOVA) was applied to determine the effect of collagen concentration and EDAC treatment on the shear modulus, degree of crosslinking and swelling capabilities of the hydrogels. A probability value of 95% (P<0.05) was used as the criterion for significance.
Results
Assessment of Collagen Content
As expected, the amount of collagen released from the polymeric networks during the washing in distilled water was strongly affected by the EDAC grafting treatment (Tab. II; P<0.0001), regardless of the theoretical collagen loading used for the synthesis. While the low collagen concentrations used in the synthesis did not affect the protein release behavior of semi-IPNs and grafted IPNs, significantly higher contents of collagen in the washing water were detected for semi-IPNs compared with grafted IPNs. This finding thus suggested that the grafting treatment was likely successful in chemically binding the collagen to the PEGDA/poly(AA) network. With regard to grafted IPNs, it is worth underscoring that some collagen might be released in the EDAC/NHS solution during the 3-hour grafting treatment. Although such a protein amount could not be detected and quantified, due to the interference of the EDAC/NHS solution with the BCA assay reagents, it is reasonable to assume that this amount is negligible compared with that released overnight by semi-IPNs during the washing.
COLLAGEN RELEASE AND SWELLING CAPABILITIES IN DISTILLED WATER
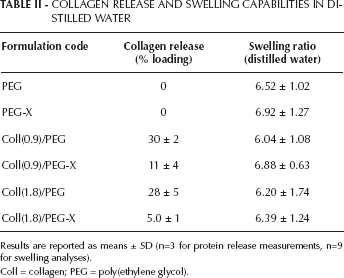
Results are reported as means ± SD (n=3 for protein release measurements, n=9 for swelling analyses).
Coll = collagen; PEG = poly(ethylene glycol).
Swelling measurements
For the hydrogel formulations tested, collagen concentration was not found to affect the swelling capability of the hydrogels in distilled water (Tab. II; P=0.25). However, for each collagen concentration, the grafting procedure by means of EDAC was found to significantly increase the swelling ratios, compared with those of semi-IPNs (Tab. II; P=0.04). Interestingly, when considering control samples devoid of collagen (i.e., samples PEG and PEG-X), the EDAC treatment was also found to increase the average swelling ratio.
With regard to the swelling measurements in NaCl solutions with different ionic concentrations, the results showed that both collagen concentration and EDAC treatment significantly affected the swelling ratio at a given ionic strength (Fig. 4; P<0.0001, for both variables). In particular, for both semi-IPNs and grafted IPNs, an increase of the collagen concentration tended to decrease the swelling capability at a given ionic strength, whereas the grafting treatment by means of EDAC was found to enhance the swelling properties of a given semi-IPN in ionic solutions. As expected, for a given hydrogel formulation, the swelling capability tended to increase when decreasing the ionic strength of the swelling solution.
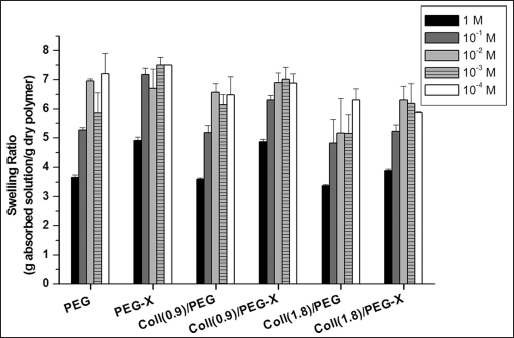
Equilibrium swelling ratios at different ionic strengths (in NaCl water solutions). Results are reported as mean ± SD of three independent measurements. Coll = collagen; PEG = poly(ethylene glycol).
Uniaxial Compression Tests
Typical compressive stress-strain curves of the hydrogels under investigation (i.e., control PEGDA/poly(AA) networks and the corresponding semi-IPNs and grafted IPNs containing collagen) are reported in Figure 5. It is worth underscoring that, for each sample, the compressive test was stopped when reaching values of about 0.7 and that no failure was detected in this range. The further elaboration of the results according to Equation 3 (Tab. III) showed that the addition of collagen to the PEGDA/poly(AA) network in semi-IPNs led to a decrease of the PEGDA/poly(AA) stiffness and degree of crosslinking, likely due to the fact that collagen macromolecules act as physical obstacles for the PEGDA chains during the crosslinking reaction, thus interfering with the crosslinking process itself. However, such a decrease was not significant, at least for the collagen concentrations tested in this study (effect of collagen concentration: P=0.06 for shear modulus, P=0.05 for degree of crosslinking). As expected, the results also confirmed that, following a grafting procedure by means of EDAC, the stiffness as well as the degree of crosslinking of the composite hydrogels increased, and became comparable with those of the PEGDA/poly(AA) network, although the overall effect was not significant (effect of EDAC treatment: P=0.28 for shear modulus, P=0.29 for degree of crosslinking).
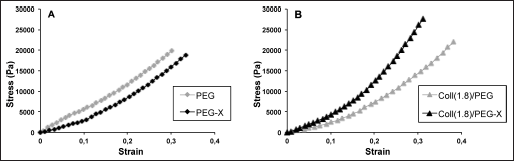
Typical compressive stress-strain curves obtained for PEGDA/poly(AA) networks (A) and semi-IPNs and grafted IPNs containing collagen (B). AA = acrylic acid; Coll = collagen; IPN = interpenetrating polymer network; PEGDA = poly(ethylene glycol) diacrylate.
SHEAR MODULUS AND ESTIMATED ELASTICALLY EFFECTIVE CROSSLINK DENSITY
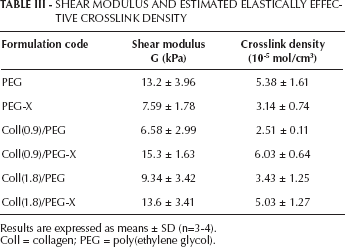
Results are expressed as means ± SD (n=3-4).
Coll = collagen; PEG = poly(ethylene glycol).
It is worth noting that the EDAC treatment, which was thought to affect only the stiffness of the hydrogel formulations containing collagen, was actually found to decrease the stiffness of the PEGDA/poly(AA) network.
Morphological analysis
The morphology of the hydrogels was affected by the presence of collagen macromolecules, as well as by the grafting treatment mediated by EDAC (Fig. 6). In particular, plain PEGDA/poly(AA) hydrogels and grafted IPNs showed a more compact structure than semi-IPNs, with plain PEGDA/poly(AA) hydrogels being particularly non-porous (both EDAC treated and untreated).

SEM micrographs of lyophilized hydrogel samples (longitudinal sections): a) semi-IPN, Coll(0.9)/PEG; b) grafted IPN, Coll(0.9)/PEG-X; c) plain PEGDA/poly(AA) hydrogel (no EDAC). The scale bar is 10 μm, and the magnification is ×2,000. AA = acrylic acid; Coll = collagen; EDAC = 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide; IPN = interpenetrating polymer network; PEGDA = poly(ethylene glycol) diacrylate.
Stereolithography of hydrogels
A meniscus-like shaped hydrogel construct, containing 0.9% w/v collagen, was successfully built by means of SLA as an assembly of 140 layers, in less than 1 hour, starting from the CAD model of a minipig meniscus. The construct showed a smooth surface (Fig. 3) and mechanical properties suitable for handling.
Discussion
The aim of this work was to explore the creation of biomimetic semi-IPNs based on PEG derivatives and collagen, also assessing their processability in complex shapes by means of SLA. PEG-based hydrogels are ideal substrates for the engineering of novel biomaterials, due to the intrinsic PEG resistance to protein adsorption, which makes it possible to design PEG-based materials able to elicit specific cell responses. Biomimetic hydrogels based on photosensitive PEG derivatives (e.g., PEGDA) have been synthesized by incorporating functionalized biomolecules or peptide sequences within the polymer network, and have been successfully employed in tissue engineering studies (1-9, 11). If the biomolecules are not anchored to the PEG network, biomimetic PEG-based semi-IPNs are obtained (20), which show promise for use in the clinical setting. A recent investigation, for example, demonstrated that photostabilized PEGDA/hyaluronic acid semi-IPNs can be transdermally photocrosslinked in humans, for soft tissue reconstruction (19). Although an inflammatory response could be observed in that study, the highly tunable properties of PEG-based semi-IPNs make them susceptible to further optimization (19).
In this work, semi-IPNs based on PEGDA and collagen were synthesized, by photocrosslinking a PEGDA-based network around collagen macromolecules. Low collagen concentrations (0.9% and 1.8% w/v) were used in the polymer mixture based on the following: First of all, the steric hindrance of collagen macromolecules is high, considering their triple helix conformation and high molecular weight (about 300 kDa), compared with that of the PEGDA used in this work (700 Da). Therefore, collagen macromolecules are very likely to interfere with the crosslinking reaction of PEGDA, leading to semi-IPNs with poor mechanical properties (indeed the stiffness of a polymer network increases at higher crosslink density or degree of crosslinking (28)). The low collagen loadings used in this work were thus likely to allow for the synthesis of semi-IPNs suitable for handling and further analyses. Secondly, for the further investigation of SLA-built semi-IPNs, low protein amounts in the polymer mixtures were preferred, to reduce the laser light scattering induced by collagen macromolecules.
With the specific aim of modulating the properties of the PEGDA/Coll semi-IPNs, the grafting of collagen to the PEGDA network was explored. To covalently bind the collagen to the PEG chains by means of a carbodiimide (EDAC)-mediated reaction, AA was added to the PEGDA solution before crosslinking. In this way, a PEGDA/poly(AA) network was obtained, with anchored carboxylic groups that could potentially react with collagen amine residues during the grafting treatment mediated by EDAC (Fig. 2). The amount of collagen retained in swollen semi-IPNs, due to the physical interaction between collagen macromolecules and PEGDA/poly(AA) network, as well as the efficacy of the EDAC treatment, were assessed by measuring the release of collagen in distilled water. The results showed that, for semi-IPNs, about 70% of the loaded collagen was retained, whereas such an amount increased up to 90%-95% in case of further chemical grafting mediated by EDAC. The swelling capability and the mechanical stiffness of both semi-IPNs and grafted IPNs were then evaluated to investigate how and to what extent those properties could be tuned by the grafting reaction.
To analyze the swelling results, it is worth recalling the microstructural parameters of a hydrogel network that affect its swelling capability – i.e., the crosslink density and the presence of pendant fixed charges on the polymer backbone. The former opposes the deformation of the chains to a more elongated state, thus limiting the swelling process (i.e., higher swelling attained for lower crosslink density) (28). In contrast, in the case of polyelectrolyte crosslinked polymers, the swelling force is greatly increased as a result of the localization of charges on the polymer chains. This is due to the different concentration of mobile counterions established between the hydrogel and the external solution, with more counterions present in the gel to ensure macroscopic electric neutrality, which induces more solvent to enter the network (28). For the same reason, polyelectrolyte hydrogels are also particularly sensitive to variations of the pH and ionic strength of the external solution (28).
The whole set of hydrogels prepared in this work showed swelling ratios in distilled water of between 6 and 7 (Tab. II). Since PEGDA/poly(AA) networks are polyanionic (due to the dissociation of carboxylic groups in COO- and H+), the sensitivity of the hydrogels to ionic strength variations was also evaluated. As expected, enhanced swelling ratios were measured when lowering the ionic strength of the external solution, for all formulations (Fig. 4) (28). The presence of collagen in semi-IPNs slightly changed the swelling ratios at given ionic strengths. Conversely, the EDAC treatment, which enabled the reaction between carboxylic and amine groups (the former located either on the PEGDA/poly(AA) network or the collagen chains, and the latter located only on collagen) was expected to strongly affect the hydrogel swelling response. This was confirmed by the swelling ratio measurements, which highlighted significant differences between semi-IPNs and grafted IPNs, with higher swelling ratios attained by grafted samples at given ionic strengths. Interestingly, such a behavior, detected in distilled water as well, was also displayed by samples devoid of collagen (denoted PEG and PEG-X).
As resulting from the mechanical tests (Tab. III), the addition of collagen to the PEGDA/poly(AA) network, to obtain semi-IPNs, led to a decrease of the crosslink density of the polymer network, due to the steric hindrance of collagen macromolecules discussed above, which is likely to limit the crosslinking reaction. In spite of the lower crosslink density, which would cause a higher swelling capability, semi-IPNs displayed swelling properties very close to those of PEGDA/poly(AA) samples. This can be ascribed to the fact that the steric hindrance exerted by collagen triple helices might sharply limit the elongation of the PEGDA/poly(AA) network chains during the swelling, and the overall electrostatic charge within the gel is changed due to the polyampholyte nature of collagen molecules. From the morphological point of view, the presence of collagen in PEG-based semi-IPNs led to a porous and irregular microstructure, compared with that of PEGDA/poly(AA) hydrogels.
With regard to the effect of the EDAC treatment, as expected, the grafting of collagen to the PEGDA/poly(AA) networks resulted in stiffer IPNs with higher crosslink densities and a more compact microstructure, when compared with semi-IPNs. In particular, the stiffness and the degree of crosslinking yielded by grafted IPNs were comparable to those of collagen-free PEGDA/poly(AA) networks. However, grafted IPNs displayed higher swelling capabilities than semi-IPNs. Although the number of carboxylic groups of the PEGDA/poly(AA) network available for ion dissociation was decreased, due to the carbodiimide-mediated condensation reaction, additional fixed charges were brought by polyampholyte collagen in the resulting IPNs. The overall concentration of mobile counterions within the gel was likely increased, thus enhancing the swelling capability. For a complete characterization of semi-IPNs and grafted IPNs, further swelling analyses should be performed to evaluate the hydrogel sensitivity to physiologically relevant variables other than the ionic strength, such as the pH and the presence of proteins in the swelling medium. For the purposes of the present study, the effect of ionic strength variations on the hydrogel swelling provided further evidence of the efficacy of the grafting reaction.
An unexpected finding was that the EDAC treatment led to a significant decrease of the crosslink density of PEGDA/poly(AA) samples (Tab. III). If such a decrease could explain the higher swelling capability of PEG-X samples at fixed environmental conditions (Tab. II; Fig. 4), the reasons the carbodiimide treatment is able to lower the elastically effective degree of crosslinking of PEGDA/poly(AA) networks (i.e., the one estimated through the mechanical tests) appear unclear and need further investigation. Overall, the reported results showed that the swelling and the mechanical properties of PEGDA/Coll semi-IPNs could be modulated by the grafting reaction and, to a lesser extent, by the loaded collagen amount. It is interesting to note that the values of stiffness obtained in this study were roughly consistent with those reported in the literature for similar PEGDA hydrogels (24). Moreover, both PEGDA molecular weight and PEGDA concentration are 2 additional variables known to affect the swelling and the mechanical properties of the resulting hydrogels, which can be tuned to mimic those of soft tissues, including cartilage (24).
As a further aim of this study, the production of PEGDA/Coll semi-IPNs with complex shapes (e.g., meniscus-like) was successfully attained by SLA. Rapid prototyping techniques, such as SLA, are particularly attractive in the biomaterial field for the production of customizable biomimetic constructs. Application of SLA to PEG-based hydrogels has been reported in the literature (15, 16, 18, 31), as well as the use of SLA to produce cell-loaded hydrogels with defined macrostructure (15, 16). Such hydrogels have been proposed either as tissue engineering platforms (15, 16, 18) or as tissue mimicking phantoms (31, 32) for in vitro studies. In this work, we demonstrated that PEG-based semi-IPNs, in addition to PEG-based hydrogels, can be processed by means of SLA. Our preliminary test showed an excellent resolution with respect to the CAD model. The typical drawbacks reported for SLA on hydrogels, such as high shrinkage and curling, were not observed, as also shown by the picture reported in Fig. 3. For the particular semi-IPN investigated (i.e., PEGDA/Coll mixture), further studies should be performed to find the optimal SLA building parameters as well as the dimensional resolution of the resulting construct. However, the possibility to use SLA to build PEGDA/Coll semi-IPNs, together with the documented possibility to control the semi-IPN properties by tuning the synthesis process, make these materials very attractive for exploitation in the biomedical field.
Conclusion
In this study, semi-IPNs based on PEGDA and collagen, potentially useful for biomedical applications, were prepared by photocrosslinking aqueous mixtures of PEGDA, AA and collagen. Remarkably, the microstructural parameters of the semi-IPNs, affecting their morphological, mechanical and swelling properties, could be modulated not only by changing some synthesis variables (e.g., PEGDA and/or collagen concentration), but also by grafting the collagen molecules to the PEGDA/poly(AA) network, by means of a carbodiimide-mediated crosslinking reaction. Moreover, the promising capability of PEGDA/Coll semi-IPNs to be processed in complex shapes by means of SLA was demonstrated.
