Abstract
Purpose
For scaffolds in cartilage tissue engineering, it is the principle to design the materials with both favorable mechanical and biological property.
Methods
In this article, collagen hydrogels modified by two ways to improve mechanical strength were applied for in vivo cartilage reconstruction: one is collagen-alginate hydrogel (CAH) representative of mixture, the other is collagen hydrogel crosslinked by genipin (CGH). To investigate the biological activities of the two materials, it was designed as: scaffolds loaded with allogenous chondrocytes were encased in diffusion chamber, and then implanted subcutaneously in SD rats for 8 weeks.
Results
Histologic, immunohistochemical, and RT-PCR results showed that collagen type II and GAG, indicator of cartilage extracellular matrix (ECM) was highly expressed in constructs of chondrocyte-CAH. Significantly lower cell density and expression of cartilage specific protein were shown in constructs of chondrocyte-CGH than that in chondrocyte-CAH. This demonstrated that CAH may provide a more favorable environment for cartilage reconstruction. In addition, the model with diffusion chamber technique was viable for evaluation of scaffolds for in vivo cartilage engineering in immunocompetent host. Instead, directly reconstruction of ectopic cartilage without diffusion chamber suffered from damaged tissue and less neo-cartilage matrix formed.
Conclusions
In conclusion, CAH is realistic as scaffold for in vivo cartilage tissue engineering with both satisfactory mechanical properties and biomimetic activity. Also, the model with diffusion chamber to reconstruct ectopic cartilage in immunocompetent animals is promising for evaluation of scaffolds. This study provided a new insight for in vivo cartilage tissue engineering.
Introduction
Articular cartilage has limited self-repair capability because of the presence of relatively few cells with low mitotic activity, low supply of progenitor cells, and lack of vascularization (1,2). As one of the most promising therapies for cartilage defects, cell-based cartilage tissue engineering has the potential to support the regeneration of large joint defects (3,4).
Material selection is critical for successful cartilage tissue engineering. In the design of scaffolds for tissue engineering, protein-based natural polymers are superior because they contain ligands that can be recognized by cell-surface receptors and have the advantage of known biocompatibility and fewer regulatory constraints (5). Among the natural polymers, the one that has received significant attention is collagen. Collagen gels are highly recommended, especially in cartilage regeneration, because they are highly hydrated three-dimensional networks mimicking the in vivo cartilage matrix. Moreover, it has also been found that collagen matrices can stimulate new collagen production by transplanted cells as compared to other scaffolds.(6) Thus, collagen hydrogels are seen as a “gold-standard” for 3-D tissue culture and serves as a positive control for 3-D cell culture in these studies (7). However, collagen has poor mechanical strength because of its loose network structure and fast degradation rate. Moreover, gel resorption and/or compaction, especially after seeded with cells, also limit the use of purified collagen (8).
There are two ways to improve the mechanical strength of collagen. One is to be combined with a reinforcing phase to form composites, in which proteoglycans such as alginate, chitosan, HA, etc were often used. This attempt also endowed the material with biomimetic properties because natural cartilage is mainly composed of collagen and proteoglycans (9). Alginates are naturally derived polysaccharides that have been extensively used as hydrogel synthetic ECMs in cartilage tissue engineering because of its mechanical and suitable biocompatibility properties, non-toxicity, and biodegradability (2,10,11). It was reported that the collagen type I gel had promoted chondrocyte proliferation, and the alginate gel had an advantage in proteoglycan synthesis (12,13). With the combined advantages and, moreover, with promoted mechanical properties such as reduced compaction, collagen-alginate hydrogel was superior in cartilage tissue engineering (8). Another way to promote mechanical properties of collagen adopted a chemical modification, which can reduce antigenicity and increase resistance of collagen to enzymatic degradation. Among various crosslinking agents, Genipin is favorable because it is a naturally occurring crosslinking agent with significantly lower cytotoxicity than commonly used agents (14-16). Moreover, it was reported to possess anti-inflammatory activity (17). Although the two ways for collagen modification are favorable for the enhancement of mechanical strength, comparison has seldom been performed.
Conversely, to select optimal scaffolds for cartilage tissue engineering and regeneration, it was suggested that application in immunocompetent animal models is promising (18). Apparently, direct application for cartilage repair is costly, which requires large amounts of animals, complicated and strict surgical operation, and long restoration period. Ectopic cartilage engineering may be an alternative, which may be an applicable resource to heal cartilage defect (18-20). However, host reaction that might destroy the chondrocytes, against the scaffold, and subsequently deform the tissue (20), impedes the development of ectopic cartilage engineering in large animals. In a previous study, ectopic cartilage has been successfully reconstructed with collagen hydrogel scaffolds and allogenous chondrocytes in a diffusion-chamber system embedded subcutaneously in rabbits (21). The engineered tissue proved to be a potential candidate for cartilage repair. This model presented a new insight for the application of engineered tissue in vivo. Conversely, it may be a useful model for inspection of engineered tissue including assessment of scaffolds in large animals. Moreover, as in the in vivo environment, especially in the immunocompetent host, it may be close to the outcome when directly implanted in situ. Thus, the model of ectopic cartilage reconstruction with diffusion chamber for estimation of engineered tissue, especially selection of scaffolds, should be further investigated.
In this article, collagen-alginate hydrogel (CAH) and collagen hydrogel crosslinked by genipin (CGH), which were different in the way of collagen modification, were used as scaffolds for cartilage tissue engineering. A comparative study between the two kinds of scaffolds was performed utilizing the model of ectopic cartilage construction with chondrocytes carried scaffolds in diffusion-chamber applied in immunocompetent SD rats. Collagen hydrogel (CH) without any modification was used as control. Simultaneously, the feasibility of the model as a means for material selection was investigated.
Materials and Methods
Fabrication of scaffolds
Collagen type I was extracted from calf skin with pepsin in acetic acid. Purification of the solubilized collagen was accomplished by sodium chloride fractionation and fibril assembly. A concentration of 7 mg/mL collagen type I solution was adjusted to neutralization by using HEPES-buffer, 1M NaOH. CGH was prepared by genipin-crosslinking collagen hydrogel, and detail referred to 2.4 cell seeding experiment.
Ice cold alginate solution of concentration of 20 mg/mL was added to neutralized collagen type I solution such that the final concentrations of collagen and alginate prior to cell encapsulation were 5 mg/mL each. The compounded solution was stored at 4°C ready for use. As control, 5 mg/mL neutralized collagen solution was prepared ready for use.
Preparation of diffusion chamber
Diffusion chamber was fabricated by using Ultra-high Molecular Weight Polyethylene (UHMWPE) as material of the ring (10 mm outer diameter and 6 mm inner diameter, 5 mm in height). Only one side of the ring was sealed with a membrane of pore size 0.22 μm (Millipore Uk Ltd, Harrow, UK) by adhesive sealant (Millipore Uk Ltd, Harrow, UK). The chambers were sterilized by γ-irradiation.
Rat chondrocyte isolation and culture
Chondrocytes were isolated by sequential enzymatic digestion from the articular cartilage of joints of new-born SD rats: first with 0.25 mg/mL typsinase and then with 2-mg/mL collagenase type II in alpha-modified Eagle's medium (α-MEM) (Gibco, Gaithcrsburg, MD, USA) for 16 hours at 37°C. After digestion, isolated chondrocytes were filtered through a 180 μm filter to remove any undigested cartilage particles, washed several times with PBS, and cells were resuspended in α-MEM media supplemented with 1% ascorbic acid, 10% FBS (Gibco. Gaithersburg, MD, USA), and 1% penicillin-streptomycin.
Cell seeding experiment
When the cells of the second passage attained 80% to 90% confluency, they were trypsinized using 0.25% trypsin/EDTA and counted using a hemocytometer. Cell viability was confirmed to be >95% before encapsulation as shown by trypan blue staining, and a stable cell metabolism was maintained. Cell pellets were used to prepare samples: 1) After centrifugation, one part of the cell pellets was resuspended and mixed in prepared collagen-alginate gel solution for a final cell concentration of approximately 5.0×10 7 cells/mL. The cell-gel composites were then injected into each diffusion chamber. The collagen-alginate mixtures were then gelled by exposing the lower surface of the membrane of diffusion chamber to a 75 mM CaCl2, 30 mM HEPES, 150 mM NaCl and 10 mM KCl buffer for 15 min at 37°C. 2) One part of the cell suspension was loaded in collagen gel solution with cell concentration of 5.0×10 7 cells/mL, and then injected into diffusion chamber. Genipin, with a final concentration of 0.05% was added to crosslink collagen. 3) After loading the cells into the scaffolds, the residue side of the chamber was sealed. 4) Cell-CAH and cell-CGH composites without diffusion chamber were prepared with the same procedure as those in diffusion chambers except injecting to diffusion chambers.
In the control group, chondrocyte encapsulated in CH with and without diffusion chamber were prepared as described previously (21).
Surgical experiment
Each sample was surgically inserted into a pocket subcutaneously at the back of the adult SD six-month-old rats under sodium barbital anesthesia. 1) In the experimental groups, rats were divided into two groups (three in one group), with one group carrying chambers and the other group for constructs without chambers. And in one group, each rat carried six samples with three cell-CAH composites on the left side and three cell-CGH composites on the right. Another group carried samples without diffusion chamber under the same conditions. 2) As control, three rats were used with cell-CH in diffusion chamber on the left side and only cell-CH on the right. Wounds were carefully rinsed with 0.9% saline solution and closed with suture.
Histologic and immunohistochemical staining
Rats were sacrificed by an intravenous injection of euthanasia solution after eight weeks and samples were harvested for analysis. For samples with diffusion chamber, cell-scaffolds constructs were removed from the chambers. Harvested specimens were then fixed immediately in 10% PBS buffered formalin over night, and then dehydrated in gradient ethanol series, embedded in paraffin, sectioned in 5-μm thickness. Cross-sections were stained with hematoxylin and eosin (HE) and safranine O.
Type II collagen was immunohistochemically stained using polyclonal antibody to type II collagen (No.bs-0709R, BIOS, China) according to the instructions.
Semi-quantitative RT-PCR
Another part of the explants was detected for the expression of cartilage matrix-specific genes using RT-PCR analysis for aggrecan, type II collagen. Detailed procedure was according to previous work (21). In short, total RNA was extracted from the tissue using Trizol reagent (Invitrogen), and then was reverse transcribed using SuperScript II (Invitrogen). After amplifications performed, PCR products were separated by electrophoresis in a 1.5% agarose gel and detected by ethidium bromide staining. Primer sequences for aggrecan (AGC) are 5′-TCAGGAACTGAACT-CAGTGG-3′ and 5′-GCCACTGAGTTCCACAGA-3′, for type II collagen (COLII) are 5V-AGTGGAAGAGCGGAGA-CTA-3′ and 5′-GACAGGCCCTAT-
GTCCACAC-3′, for GAPDH are 5′-AACTCCCTCAA-GATTGTCAGCA-3′ and 5′-TCCACCACCCTGTTGGCT-GTA-3′. Results are shown as mean ratio±standard deviations. Statistical significance between two groups was determined by one way ANOVA. P≤.05 was considered statistically significant.
Results
Macroscopic observations
A thin fibrous tissue with a light vascular network encapsulated all chambers and only minimal fibrotic reaction was observed (Fig. 1A). The thin fibrous capsule surrounding the chamber was easily peeled from the chamber. From its appearance, tissue in the chamber with CAH as scaffolds was opaque and pale in color like native cartilage (shown in Fig. 1B), and vascular invasion could not be observed. Tissue in CGH was light blue in color as for the effect of crosslinker (Fig. 1C). In the control group, although tissue with CH was white and cartilage-like, certain degrees of degradation were observed (Fig. 1D).
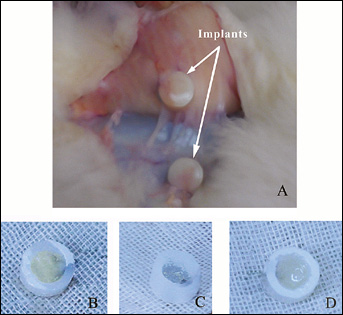
Macroscopic appearance of specimens after eight weeks subcutaneous implantation in rats. A thin fibrous capsule was observed around the chambers (A). Inside the chamber, constructs with CAH as scaffolds appeared opaque and pale in color (B). Tissue in CGH was light blue in color (C). Although tissue with CH was white, degradation was seen (D).
In the groups without chamber, the tissues with CAH and CGH were both encapsulated by a fibrous capsule and surrounded by vascular networks (Fig. 2), indicating medium fibrotic reaction. Comparatively, fibrotic reaction was more intense in CGH groups than CAH. In the control group, tissue could not be found, which may be degraded completely.
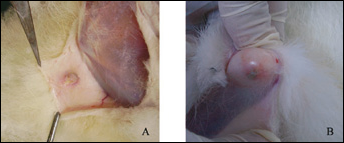
Gross morphology of specimens after eight weeks implantation directly at subcutaneous site. The tissues with CAH (A) and CGH (B) were both encapsulated by a fibrous capsule and surrounded by vascular networks.
Histologic and immunohistochemical observations of the tissue in chamber
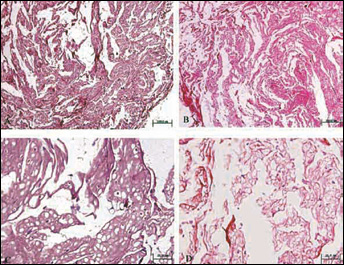
As shown in Fig. 3A and Fig. 3D, histologic examination of the tissue in the chamber with CAH indicated normal features of cartilage with round chondrocytic cells which were densely and homogeneously distributed. And as indicated by safranine-O staining with a mass region of deep red color, proteoglycan and GAG were intensively deposited around the cells (Fig. 4A). In samples with CGH as scaffold, cells were aggregated only in some regions and the number of cells was much less than in tissue with CAH (Figs. 3B and 3E). It could also be observed that deep GAG deposition centered only in some areas (Fig. 4B). In the control group with CH as scaffold, cells were densely distributed (Fig. 3C, F) with proteoglycan deposited around (Fig. 4C), but scaffold degradation was obvious.
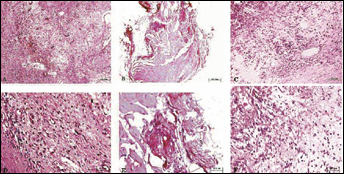
HE staining of sectioned tissue inside diffusion chamber after it was implanted in vivo for eight weeks (A, B, C: original magnification × 100; D, E, F: original magnification × 400). The tissue in the chamber with CAH showed round chondrocytic cells which were densely and homogeneously distributed (A, D). In samples with CGH as scaffold, cells were aggregated only in some regions and the number of cells was much less (B, E). Although cells were densely distributed, scaffold degradation was obvious (C, F).
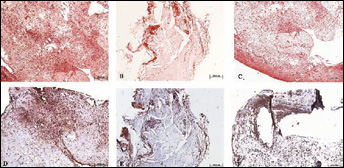
Safranine-O staining and immunohistochemical staining of cross sections of harvested tissues inside diffusion chamber after eight weeks (original magnification × 100). Deep red color of ECM shown by Safranine-O staining indicated intense GAG deposition around cells for constructs with CAH as scaffolds (A). While deep GAG deposition centered only at some area for constructs with CGH (B). Immunohistochemical staining showed type II collagen was strongly positively stained (dark brown staining) in tissue with CAH (C). Only the region with dense distributed cells showed positive anti-type II collagen antibody staining in tissue with CGH (D). In the control group with CH as scaffold, GAG deposition (C) and positively stained type II collagen (F) was shown in the area without degradation.
Immunohistochemical staining showed that type II collagen, indicator of cartilage differentiation, was strongly positively stained (dark brown staining) in tissue with CAH (Fig. 4D). Comparatively, only the region with dense distributed cells showed positive anti-type II collagen antibody staining in tissue with CGH (Fig.4E). In the control group, the area without intense degradation showed positively stained type II collagen (Fig. 4F).
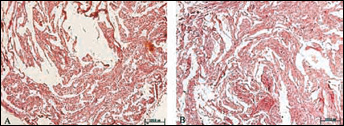
From Figure 5 showing tissues without chamber, there were modicum round-shaped cells dispersed, and the matrix degraded much more than in groups with chamber. The color stained by safranine-O was relatively fainter (Fig. 6). There was little difference between constructs with CAH and CGH without diffusion chamber.
HE staining of sectioned tissue directly embedded at subcutaneous site after eight weeks (A, B: original magnification × 100; C, D: original magnification × 400). There were modicum round-shaped cells dispersed, and the matrix degraded much more in both tissue with CAH (A, C) and CGH (B, D) as scaffolds.
Safranine-O staining of sectioned tissue directly embedded at subcutaneous site after 8 weeks. (original magnification × 100) Both constructs with CAH (A) and CGH (B) as scaffolds showed GAG deposition.
RT-PCR analysis and densitometry
Figure 7 showed the RT-PCR results. It was revealed that expression of aggrecan (1.09-fold) and type II collagen (0.55-fold) in CAH groups was significantly higher than that in CGH (accordingly 0.06- and 0.028-fold). Moreover, CAH showed even significantly higher expression of cartilage specific matrix than CH.
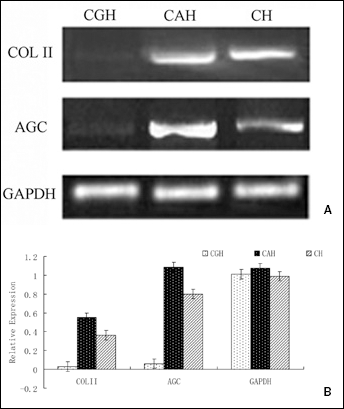
Reverse transcription-polymerase chain reaction analysis and of specimens with CGH, CAH, and CH as scaffolds in diffusion chamber (A). Genes of type II collagen (COL II), Aggrecan (AGC) were detected. Densitometry of PCR products of specimens (B) showed X-fold changes of AGC, COL II in relation to gene GAPDH. Mean values ± standard deviation are derived from three samples. COLII and AGC groups showed statistically significant differences between samples (P≤.05).
Discussion
Scaffolds play a crucial role in tissue reconstruction. As natural polymers, collagen scaffolds are useful matrices for cartilage tissue engineering but lack mechanical strength (22,23). The approaches to improve its strength include physical incorporation of elements and chemical modification such as crosslinking. Although the two ways are effective in improving mechanical properties, there were few reports on the comparison, especially of biological properties. In addition, advanced model for the assessment of scaffolds is scarce.
Between the two methods for collagen modification, our study showed that hybrid polymer CAH was superior to CGH as scaffold for ectopic cartilage reconstruction in SD rats. Macroscopic observation, histologic and immunohistochemical assays, and RT-PCR results indicated the feature of cartilage for CAH-chondrocyte constructs: white color in appearance, densely distributed round-shaped cells, and expression of cartilage indicator such as aggrecan and type II collagen. In contrast, only a limited and minor region showed the characteristic of cartilage for CGH-chondrocyte composites. The difference in the constructs may be attributed to the fact that CAH was more biocompatible and suitable for the proliferation and growth of cells, resulting in the high expression of cartilage extracellular matrix, while CGH was less cytocompatible. Although cytotoxity of genipin is much lower compared to most crosslinking agents, it was suggested that crosslinking may have an adverse effect on cell proliferation. Comparatively, alginates are known for their suitable biocompatibility properties which would have less impact on the biological properties of composite polymers. This was reflected in the results that fibrotic reaction was more intense in CGH than in CAH. Moreover, as the natural cartilage is mainly composed of collagen, proteoglycans and water, addition of alginates in collagen gels may provide a favorable biomimetic environment for cells to proliferate and produce extracellular matrix.
To the best of our knowledge, scaffolds for cartilage tissue engineering are required to meet multiple and often conflicting mechanical and biological design requirements. The mechanical strength of both collagen-based scaffolds (CAH and CGH) has unquestionably been improved, as evidenced by the reports (8,16). Previous work showed gel contraction was improved after modification (21,24). Our study also showed that, after two months implantation in vivo, chondrocyte-mediated gel degradation was effectively inhibited, while tissue constructed with CH scaffold degraded even with the protection of diffusion chamber. This demonstrated enhanced mechanical properties through modification. But accompanied with the addition of other components or crosslinking to improve mechanical properties, biological performance would be affected. How to maintain or improve the biological properties after collagen modification becomes crucial. Thus, scaffolds with both favorable mechanical and biological properties were more suitable for tissue engineering. From this point of view, CAH was more realistic compared to CGH.
On the other hand, selection of a favorable model for the assessment of scaffolds for cartilage tissue engineering is also critical. It was suggested that ectopic cartilage engineering in competent animals was promising (19,20), except that it would suffer from immunoreaction and vascular invasion by host. The diffusion chamber technique was adopted in this study to reconstruct cartilage tissue as it can effectively preclude invasion by host cells and vascular invasion (21). Results showed that leakproof chambers were effective in avoiding direct contact with the host tissue. Vascular invasion, which may result in calcification and bone formation, was also prevented. Without the protection of chambers, the attack to allogenous cells and scaffolds were obvious as indicated by little cells remained and mass areas of degradation. In addition, the CH group without chamber could not be found. This may be caused by host immunoreaction. All this demonstrated directly implanting samples subcutaneously was not fit for the assessment of scaffolds. Thus, the diffusion chamber system is a useful model for the assessment of scaffolds for cartilage tissue engineering.
CONCLUSION
This study successfully reconstructed ectopic cartilage in immunocompetent host and demonstrated that 1) for scaffolds with collagen, CAH with better cytocompatibility and mimicked property was superior to CGH; 2) with improved mechanical properties, the biocompatibility of CAH is also favorable, indicating it was realistic for in vivo cartilage tissue engineering; 3) the diffusion-chamber system is effective in preventing host immune rejection and vascular invasion, providing a favorable model for the assessment of scaffolds for cartilage tissue engineering.
