Abstract
Objective:
To conduct a meta-analysis of prospective and retrospective studies to reveal the relationship between the Glasgow prognostic score (GPS) and overall survival (OS) or progression-free survival (PFS) in patients with lung cancer.
Methods:
Correlative studies were included by searching the databases of PubMed, Web of Science, Embase, and PubMed Cochrane Library until April 16, 2017. We combined the hazard ratios (HRs) and 95% confidence intervals (CIs) to assess the correlation between GPS and OS or PFS in patients with lung cancer.
Results:
Ten studies involving 5,369 participants from several regions were identified through searching databases. In a pooled analysis of all studies, elevated GPS was associated with poorer OS (HR = 2.058; 95% CI, 1.51-2.80; p<0.05). However, the combined data showed no significant relationship between the GPS of 1 or 2, and PFS, respectively. Subgroup analysis showed that the patients with GPS ≥1 had poorer OS compared with those with GPS = 0 (HR = 2.01; 95% CI, 1.75-2.32; p<0.001). A similar trend was observed in patients receiving chemotherapy (HR = 1.66; 95% CI, 1.17-2.36; p<0.05) and surgery (HR = 2.88; 95% CI, 1.59-5.22; p<0.001) when stratified by treatment.
Conclusions:
Increased level of GPS may have a prognostic value in lung cancer. We detected a statistical difference in the association of elevated GPS and poorer OS, though the association was not significant in PFS settings. However, further studies are warranted to draw firm conclusions.
Introduction
Lung cancer is the most common incident cancer and its trends in mortality, which account for 28% in males and 26% in females, have not improved in both men and women for a number of years (1). Most patients were at advanced stages when they were diagnosed lung cancer and the most common treatment are surgery and chemotherapy (2). Although the death rate among lung cancer patients has reduced 16%-20% in adults with smoking history by computed tomography, overall survival is poor (3). The most established prognostic indicator of lung cancer so far is clinical staging status. However, it provides limited information on the disease progression of individual patients and the ideal method to predict the prognosis of lung cancer remains unclear.
A highly proliferative tumor can produce inflammation, as evidenced by increased cytokine and chemokine production, and immune cells gathering in the local tumor microenvironment (4, 5). Mounting evidence has supported that the elevated level of system inflammation was related to the shorter survival time of patients with many cancers by promoting cancer cell proliferation and survival, angiogenesis, and tumor metastasis (6, 7). A variety of inflammatory indices, including the platelet to lymphocyte ratio (PLR), the neutrophil lymphocyte ratio (NLR), the Glasgow Prognostic Score (GPS), and the modified Glasgow Prognostic Score (mGPS) have attracted attention for their prognostic efficiency in cancer patients (8-11). GPS is a useful prognostic score based on C-reactive protein (CRP) and albumin, which present not only the inflammatory status but also the nutritional status. Briefly, patients with both an elevated CRP (>10 mg/L) and hypoalbuminemia (<35 mg/L) were determined as the score of 2. Those in whom only 1 of these biochemical abnormalities was present were allocated a GPS of 1. Where neither of these abnormalities was present, the GPS was allocated as 0.
Although accumulated evidence has shown the clinical utility of GPS in patients with different stages of lung cancer (11-19), data are still scarce and inconsistent. The study conducted by Zhu et al (20) to assess the clinical unity of GPS in non-small cell lung cancer (NSCLC) involved articles that evaluated the prognostic efficiency of GPS as well as mGPS and failed to find the relationship between GPS and progression-free survival (PFS). Therefore, we reenact the inclusion and exclusion criteria and collected the available publications and conducted a meta-analysis to explore the prognostic value of GPS for overall survival (OS) and PFS in lung cancer.
Method
Protocol and registration
This meta-analysis is reported in accordance with the Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) statement (information is provided in supplementary material) and was registered at International Prospective Register of Systematic Reviews (PROSPERO): number CRD42017064259.
Search strategy
The databases of Cochrane Library, Web of Science, Embase and PubMed were thoroughly searched until April 16, 2017 for cohort studies assessing the association between GPS and the prognosis in patients with lung cancer, without any restrictions of language or publication period. The following keywords were used in separation or in combination: “pulmonary neoplasms,” “lung carcinoma,” “pulmonary cancer,” “c-reactive protein,” “albumin, serum,” and “Glasgow prognostic score.” Reviews and reference lists were also manually retrieved from additional publications.
Eligibility criteria
The following inclusion criteria were used: (i) the exposure of interest was GPS in patients pathologically diagnosed with lung cancer; (ii) the outcome of interest included OS and PFS with hazard ratios (HRs) and corresponding 95% confidence intervals (CIs); (iii) GPS was measured based on CRP and albumin of serum; and (iv) full text papers published in English.
The exclusion criteria were as follows: (i) reviews, meeting abstracts, and letters; (ii) nonhuman studies; and (iii) if data sets were overlapped or duplicated, only the most recent information was included.
Data extraction
Two independent reviewers (J. Jin and K. Hu) extracted the following information from the eligible studies, and any disagreement between them was resolved by discussion until consensus was reached. The following information was recorded from included studies: first author’s surname, study country, year of publication, study design, length of follow-up (months), sample size, treatment methods, and survival information.
Quality assessment
The risk of bias of each included study was assessed by 2 independent reviewers (J. Jin and K. Hu). Assessment of the quality of the studies was performed by using the Newcastle–Ottawa quality assessment scale (NOS). The full mark is 9 points and studies labeled with a score of 6 or greater were considered of high quality. Discrepancies were resolved by discussion until consensus was reached.
(http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp)
Statistical analysis
Statistical analysis was conducted using STATA (version 12.0; Stata Corporation). We use the HRs in multivariate models whenever available. If there were only univariate models presented, we used the HRs in the univariate models. HRs and 95% CIs of OS or PFS were estimated from the Kaplan–Meier curves if they were not presented directly in the articles, according to the methods reported by Tierney et al (21). Pooled HRs and 95% CIs were synthesized with the Mantel–Haenszel method under a random-effects model, which considers the heterogeneity between studies. Statistical heterogeneity among the studies was evaluated with Q and I2 statistics, with the significance level set at p<0.05 (22). Sensitivity analyses to rule out over-representation of results from a single study in the meta-analysis were performed by excluding each study individually from the meta-analysis. The potential publication bias was evaluated using Begg’s test (23) and p<0.10 was considered as being statistically significant.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee, and with the 1964 Helsinki declaration and its later amendment.
Result
The characteristics of included studies
The flow diagram of this study is presented in Figure 1. A total of 9 publications with 5,369 patients were included in the meta-analysis (11-19). One publication included 2 different cohorts (operative, inoperative) and reported their HRs separately, so we have treated them as 2 studies (11). The sample sizes ranged from 96 to 1,074. Of all the included publications, 3 were prospective studies (12, 15, 19) and the rest were retrospective. Of these studies, 2 (18, 19) were conducted in Europe, and the remaining researches were carried out in Asia. A total of 3,022 patients from 5 studies (11, 13, 14, 16, 17) received surgery as the treatment for lung cancer, and 2,347 patients received chemotherapy. Four studies (12, 17-19) involved patients with advanced stages III or IV, and 5 studies (11, 13-16) included patients with early stages. However, HRs were not reported in 2 studies (13, 17). With the reference of score 0, 5 studies (12-15, 18) provided the HRs for scores 1 and 2. Finally, in this meta-analysis of 10 studies, the HR for lung cancer was calculated for the highest versus the lowest category of GPS (24). Almost all studies defined OS as the time from diagnosis to the day of death or last follow-up, and the definition of PFS was the time between the date of diagnosis and the date of first recurrence or last follow-up. The articles were published from 2003 to 2015 and the NOS scores of the included studies ranged from 6 to 9. The detailed information is shown in Table I.
Flow chart of the study selection.
Characteristics of all included studies
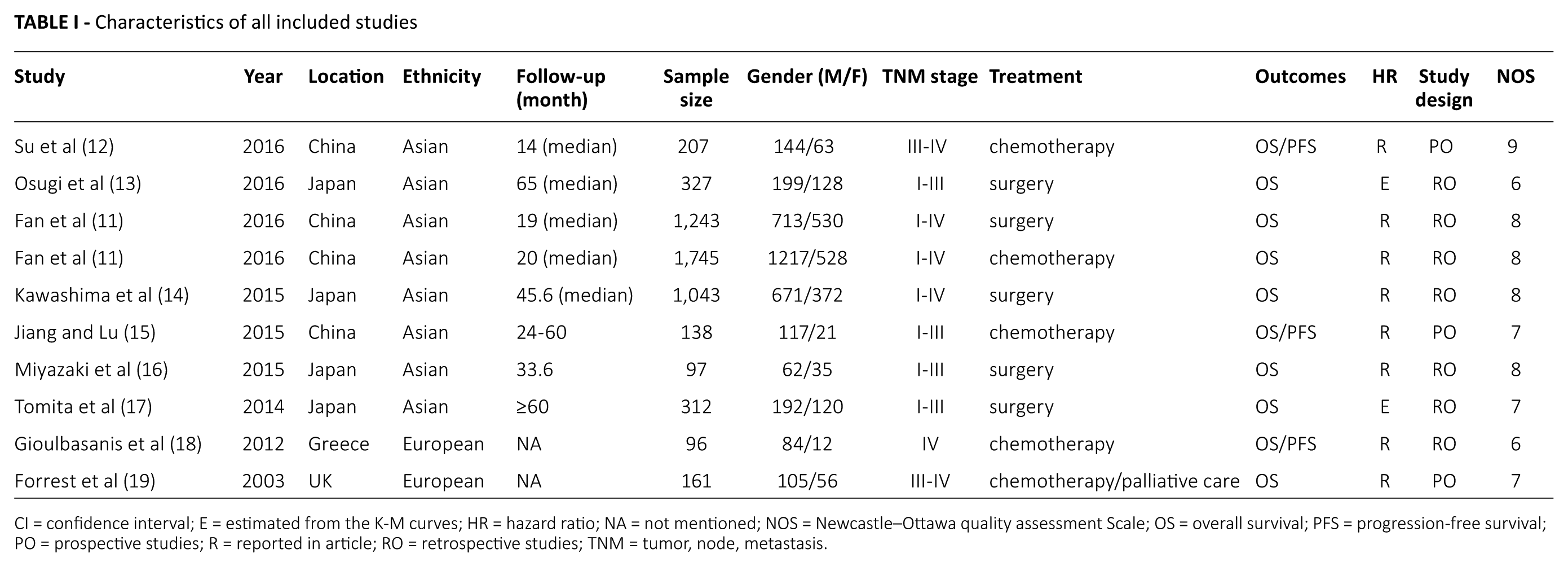
CI = confidence interval; E = estimated from the K-M curves; HR = hazard ratio; NA = not mentioned; NOS = Newcastle–Ottawa quality assessment Scale; OS = overall survival; PFS = progression-free survival; PO = prospective studies; R = reported in article; RO = retrospective studies; TNM = tumor, node, metastasis.
Relationship between GPS and OS in lung cancer
Ten studies with 5,369 patients have provided the data of GPS before treatment and OS in lung cancer. The random effects model shows a significant relationship between elevated GPS and OS in patients with lung cancer (HR = 2.058; 95% CI, 1.51-2.80; p<0.05) with high heterogeneity (I2 = 77.3%, p<0.001, Fig. 2).
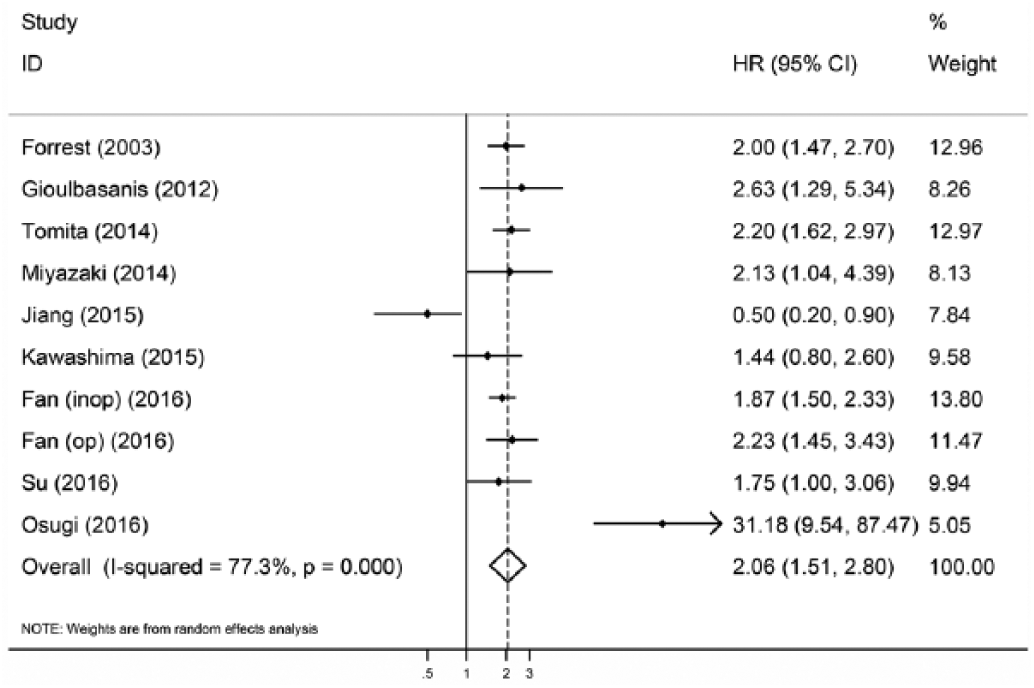
Forest plot of the association between the Glasgow prognostic score (GPS) and overall survival (OS) in patients with lung cancer.
Relationship between GPS and PFS in lung cancer
Only 3 studies with 441 patients reported the HRs and 95% CIs of different scores of GPS and PFS. The combined data showed no significant relationship between the score 1 of GPS and PFS (HR = 0.96; 95% CI, 0.74-1.25; p = 0.77) with nonsignificant heterogeneity (I2 = 24.0%, p = 0.27) yielding similar results regarding the relationship between the score 2 of GPS and PFS (HR = 1.56; 95% CI, 0.63-3.90; p = 0.34) (I2 = 84.3%, p<0.05) (Tab. II).
A summary of HRs for the overall and subgroup analyses of GPS and lung cancer
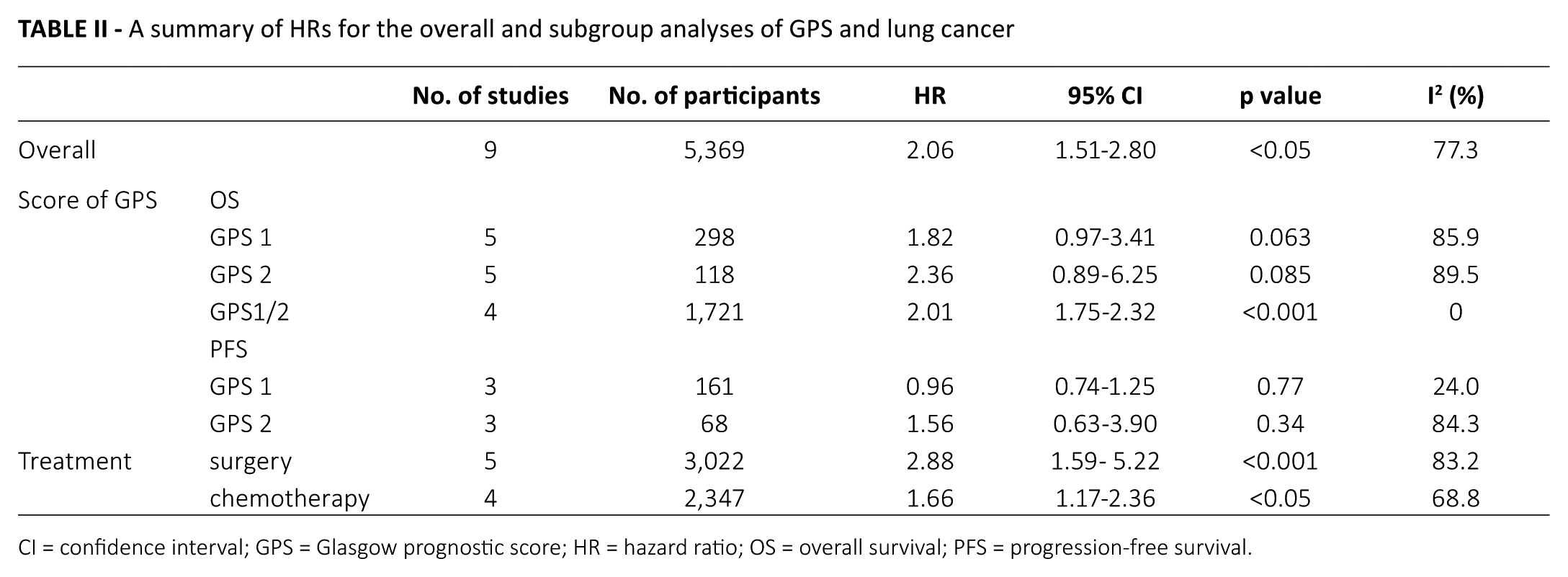
CI = confidence interval; GPS = Glasgow prognostic score; HR = hazard ratio; OS = overall survival; PFS = progression-free survival.
Subgroup analyses
To detect the potential source of heterogeneity, subgroup analyses stratified by treatment and different scores of GPS were conducted (Tab. II, Fig. 3). As shown in Table II, GPS of 1 or 2 did not predict poor OS in patients with lung cancer (Fig. 3B, C). However, the patients with a GPS ≥1 had a significantly poorer OS compared with those with a GPS = 0 (HR = 2.01; 95% CI, 1.75-2.32; p<0.001) (I2 = 0%; p = 0.91) (Fig. 3D). We also performed a subgroup analysis based on treatment, to further explain the results of this meta-analysis. Among patients receiving surgery, elevated GPS was correlated with shortened OS (HR = 2.88; 95% CI, 1.59-5.22; p<0.001) (I2 = 83.2%; p<0.001). A similar trend was observed in patients receiving chemotherapy (HR = 1.66; 95% CI, 1.17-2.36; p<0.05) (I2 = 68.8%; p = 0.012).
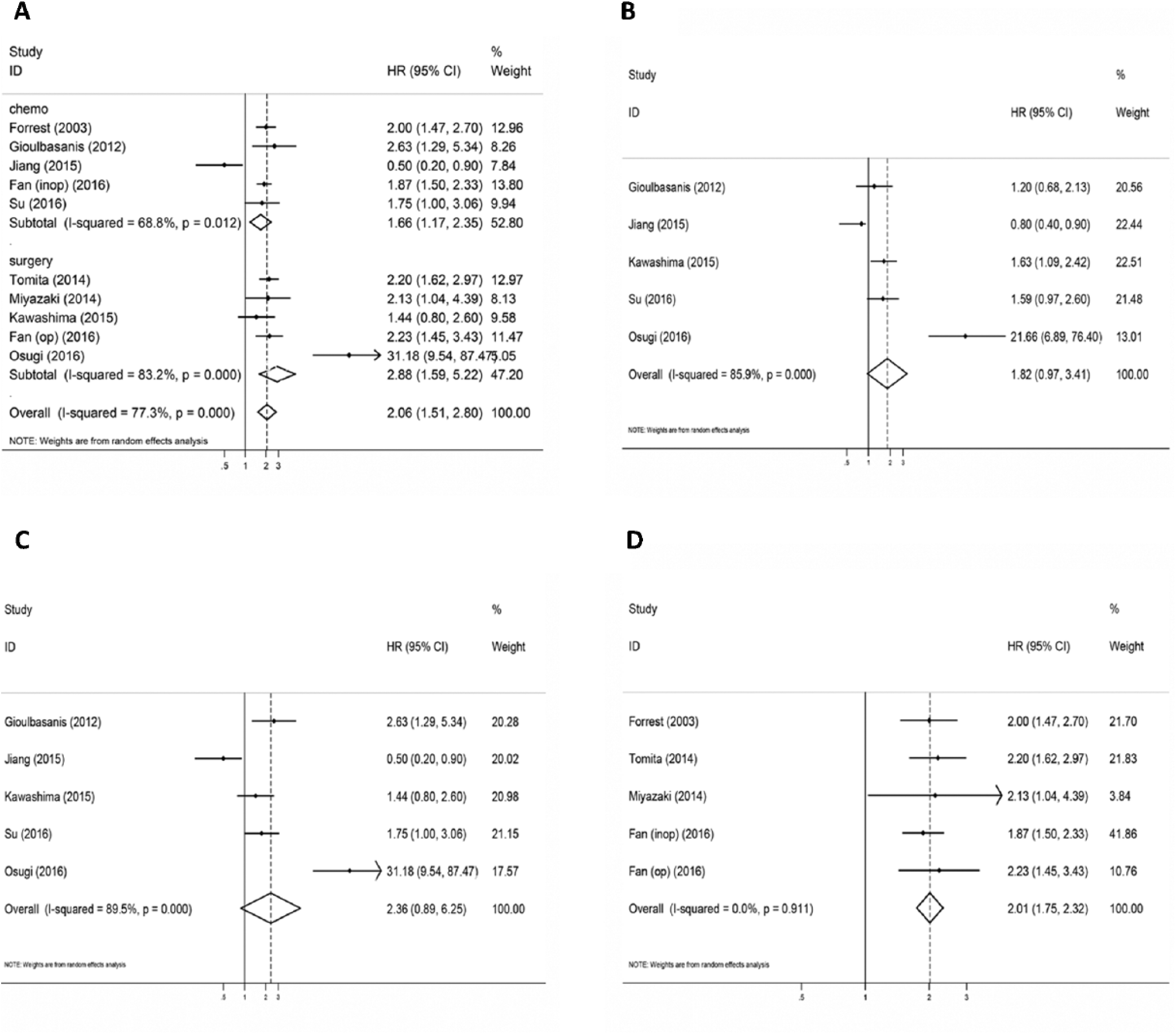
Forest plot of the association between the Glasgow prognostic score (GPS) and overall survival (OS) in patients with lung cancer stratified by treatment and GPS.
Sensitivity analysis and publication bias
Significant heterogeneity was discovered among all studies (I2 = 77.3%, p<0.01), even after subgroup analysis. The influence of each single study set on the combined HRs was evaluated by excluding each study individually from the meta-analysis. The results showed that the pooled HRs for OS were robust in our study (Supplement Fig. 1. Available online at www.biological-markers.com). Moreover, Begg’s test and the funnel plot showed no evidence of obvious publication bias (p = 0.858) (Supplement Fig. 2. Available online at www.biological-markers.com).
Discussion
The current meta-analysis summarizes the results of prospective and retrospective studies involving 5,369 patients. By combining the HRs and 95% CIs from 10 studies, we showed the prognostic impact of pretreatment GPS on OS and PFS in patients with lung cancer. Our results revealed that elevated GPS was significantly associated with poorer OS (HR = 2.058; 95% CI, 1.51-2.80; p<0.05), but it was not significantly correlated with PFS. However, the subgroup analysis, stratified by the GPS, showed that GPS of 1 or 2 had no prognostic effect for OS. Furthermore, stratified analysis by treatment showed that a higher GPS had consistent prognostic value in a diverse subgroup population.
Elevated levels of systemic inflammation are associated with worse survival in solid tumors (25, 26). Readily available blood-based parameters, including NLR, PLR, and mGPS, could reflect the cancer-related inflammatory status and are used as prognostic factors in lung cancer (9, 10, 27-29). Systemic inflammation in patients with malignancy is considered to reflect the cytokine profile produced by the tumor (30). CRP, which is caused by microbial invasion and is synthesized by liver cells, is a reflection of local inflammation in the circulation (31). Its prognostic value in patients with various types of cancer was investigated in many studies (32-38). Furthermore, albumin can reflect the nutritional status of patients with cancer and malnutrition is correlated with worse survival (39). The GPS, which evaluates both the level of systemic inflammation and the nutritional status, may be a potential prognostic predictor for lung cancer, which has been assessed in many studies (11-19).
In addition, we noticed that the previous study searched literature until August 31, 2015, and 12 studies were included (20). However, the primary outcome of 5 studies (40-44) were mGPS, not GPS. Unlike the previous study, we strictly limited the rule of inclusion and exclusion, and we enrolled 3 additional articles published in 2016, conducting a study with a relatively larger number of participants with detailed information. Also, the research conducted by Zhu et al. (20) did not evaluate the association between GPS and PFS in patients with NSCLC, which was different from ours. The results of our study supported elevated GPS as a prognostic factor for OS in lung cancer, which is consistent with most other studies. In addition, we observed that the GPS of 1 or 2 may be associated with poorer PFS, but due to the limited number of studies we were able to include, the results are not significant and must be interpreted with caution. Although most survival data were extracted from multivariate models adjusted for the potential confounding factors, including gender, age, cancer stage, treatment, and other biomarkers, a few studies only provided the survival data on univariable analysis. This may lead to the high heterogeneity. However, due to the limited information presented in the studies, it was not possible to perform a subgroup analysis according to different confounding factors.
The current research has several limitations. First, high heterogeneity existed in this meta-analysis. The heterogeneity was notably reduced after we omitted the Osugi et al (13) and Jiang and Lu (15) studies, but the pooled HR was not attenuated. So the heterogeneity may be caused by these 2 studies, though they have no significant bias after careful evaluation, and our result remained stable whether they were included or not (Supplement Tab. I. Available online at www.biological-markers.com). Second, participants from enrolled studies were at different clinical stages. A total of 2,693 patients with advanced stages III and IV, and 2,676 patients with early stages I and II, were involved in this meta-analysis, but we were not able to perform a subgroup analysis according to different clinical stages.
Generally, our study demonstrates the prognostic role of increased GPS for poor OS, but not for PFS in lung cancer by meta-analysis. However, given the limitations mentioned above, these findings should be treated with caution when applied to clinical practice. More prospective cohort studies are warranted to test our results.
Footnotes
Disclosures
Financial support: This study was funded by Technology Support Program of Science and Technology Department of Sichuan Province (2014SZ023).
Conflict of interest: None of the authors has financial interest related to this study to disclose.
