Abstract
Background
Liver fibrosis is the main determinant and predictor of the clinical course of nonalcoholic fatty liver disease (NAFLD). To date, a liver biopsy is still considered the gold standard for staging fibrosis. The aim of this study was to investigate the diagnostic accuracy of the commercial enhanced liver fibrosis (ELF) test manufacturer's cutoff value (≥9.8) in identifying severe fibrosis for adult patients with histologically confirmed NAFLD.
Methods
We tested the ELF test in a clinical practice, prospective cohort of 82 consecutive patients who consecutively underwent percutaneous liver biopsy.
Results
All stages of liver fibrosis were represented in our cohort, and severe fibrosis was present in 15 of 82 patients (18.3%). The stage of fibrosis was significantly associated with ELF score (Spearman's rho = 0.483, p<0.001). The commercial ELF test manufacturer's cutoff identified severe fibrosis with good sensitivity (86.7%; 95% confidence interval [95% CI], 0.69-1.04) and high specificity (92.5%; 95% CI, 0.86-0.99), with a positive predictive value of 72% and negative predictive value of 97%.
Conclusions
Our data could support the use of the ELF test in clinical practice.
Introduction
Nonalcoholic fatty liver disease (NAFLD) represents the most common liver disease in Western countries with a prevalence that has risen to 25%-30% of the general population and with higher figures reported in diabetic and obese patients (1). The histological features of NAFLD include the whole spectrum of liver disease ranging from “simple” steatosis to inflammation and ballooning (nonalcoholic steatohepatitis [NASH]) and progressive fibrosis. Moreover, NASH has recently been estimated as to be the third most widespread indication for liver transplantation in the United States, and it is predicted to become the first indication in the near future (2), and NAFLD overall is becoming a new leading cause of liver mortality due to cirrhosis and hepatocellular carcinoma (HCC) (3-5).
A review of repeated biopsy series pointed out the severity of fibrosis as the main determinant and predictor of the clinical course of the disease for patients with NAFLD (6, 7). In this regard, liver fibrosis represents a key indicator for identifying high-risk cases which deserve a stricter follow-up. Even if liver biopsy has several limitations, it represents the gold standard for the assessment of severity of liver damage in NAFLD (8). A liver biopsy is an invasive procedure and is not reliable for all patients with NAFLD, so unmet need exists in the identification of patients at higher risk of fibrosis with a noninvasive test that could be standardized in clinical practice. Unfortunately liver function tests are not able to reflect the severity of liver fibrosis (9).
The capability of detecting severe fibrosis may also have an innovative impact on public health since a simple test would be helpful for predicting mortality in patients with NAFLD (10).
In 2004, a simple combination of age and circulating extracellular matrix components, such as hyaluronic acid (HA), amino-terminal propeptide of type III procollagen (PIIINP) and tissue inhibitor of metalloproteinase-1 (TIMP-1), was proposed for noninvasive assessment of fibrosis (11) and validated in a meta-analysis (12). These parameters were then included in a commercial test named the enhanced liver fibrosis (ELF) test, but few data are still available to validate this test in an independent prospective NAFLD cohort in real clinical practice.
To fill this gap, we aimed to investigate the the diagnostic accuracy of the commercial ELF test manufacturer's cutoff value (≥9.8) in identifying severe fibrosis for patients with histologically confirmed NAFLD.
Patients and methods
Patients
The study protocol was approved by the local institutional review board and was conducted according to the code of ethics of the World Medical Association (Declaration of Helsinki). The study was completed in the Outpatient Liver Unit of the Gemelli and Columbus Hospital in Rome.
Inclusion criteria were history of persistent or intermittent increase of serum aminotransferases (at least 1.5-fold above normal limit) in 2 determinations obtained during a period of 6 months prior to enrollment, liver ultrasound suggestive of fatty liver at the screening visit, with history of insignificant alcohol intake (<30 g/day for men and <20 g/day for women). Ultrasound diagnosis of liver steatosis was based on the comparative assessment of liver and kidney echogenicity, according to the European Association for the Study of the Liver, European Association for the Study of Diabetes and European Association for the Study of Obesity guidelines (8). Exclusion criteria were evidence of autoimmune liver disease, serological evidence of HCV or HBV infection or seroconverted HBV infection, use of drugs known to promote steatosis (tamoxifen, amiodarone and corticosteroids, chemotherapeutics) in the last 5 years, bariatric or gastrointestinal surgery or clinical signs of overt liver cirrhosis. We also excluded patients with other forms of liver disease by ceruloplasmin and alfa-1-antitrypsin.
We recorded anthropometric data, including height, weight and waist circumference, and reviewed each patient's medical history. Body mass index (BMI) was calculated using the formula: BMI = weight (kg)/height (m2), and obesity was defined as BMI ≥30. The laboratory work-up included routine liver function tests (aminotransferases, total bilirubin, albumin, alkaline phosphatase, γ-glutamyltransferase and coagulation tests), lipid profile (triglycerides, total, high-density lipoprotein [HDL] and low-density lipoprotein [LDL] cholesterol), fasting glucose and insulin levels, creatinine and blood urea nitrogen. Insulin resistance was assessed by homeostasis model assessment (HOMA) (13), with diabetes defined as fasting glucose ≥126 mg/dL or treatment with antidiabetic drugs, according to World Health Organization (WHO) recommendations (14).
The presence of metabolic syndrome was defined as the presence of at least 3 of the following characteristics, according to Adult Treatment Panel III (ATP III) criteria (15): central obesity (waist >102 cm in men and >88 cm in women); hyperglycemia (fasting blood glucose ≥110 mg/dL or diagnosis of diabetes); hypertriglyceridemia (triglycerides ≥150 mg/dL or current treatment for hypertriglyceridemia), hypertension (blood pressure greater than or equal to 130/85 mm Hg or current treatment for blood hypertension) or low HDL cholesterol levels (<40 mg/dL in men or <50 mg/dL in women or current treatment for hypercholesterolemia).
Liver biopsy
Ultrasound-guided percutaneous liver biopsies were performed using the modified Menghini technique. Liver specimens (at least 1.6 cm in length and 5 μm in thickness) were stained with hematoxylin-eosin, Masson's trichrome and periodic acid–-Schiff (PAS) and examined under blind conditions by an experienced pathologist. Steatosis, necroinflammation and fibrosis were assessed according to the modified Brunt criteria, and the liver specimens were evaluated according to the Kleiner NAFLD activity score (NAS) for steatosis, ballooning and lobular inflammation (7, 16, 17).
ELF test assay
Serum HA, PIIINP and TIMP-1 were assayed using the ELFTM Test ADVIA Centaur® Kit (Siemens Healthcare Diagnostics, Germany) on the ADVIA Centaur CP immunochemical analyzer (Siemens Healthcare Diagnostics, Germany), according to the manufacturer's instructions. Serum analyses were performed after instrument calibration and adequate quality control assessment (low-, intermediate- and high-level controls for each serum assay and for ELF score). The ELF score was calculated for the instrument based on the following equation: ELF score = 2.494 + 0.846 In (CHA) + 0.735 In (CPIIINP) + 0.391 In (CTIMP-1). According to the manufacturer, an ELF score ≥9.8 was to be used as an indicator of severe fibrosis.
Statistical analysis
Given the pilot nature of the study, we estimated the required sample size using G*Power software (18). Assuming a nonparametric distribution with a alpha error of 0.05 for a power (1-β) of 0.8, the indicated sample size was 14 patients for each group.
Data were analyzed by t-test and Wilcoxon rank sum test for continuous and ordinal factors (for parametric and nonparametric distributions, respectively) and by Pearson chi-square test or Fisher's exact test for categorical variables.
To assess the influence of clinical variables on the presence of severe fibrosis and/or ELF >9.8, we performed a logistic regression by a backward stepwise with the Wald method including variables with a significant association (p<0.05) at univariate analysis. The goodness of fit was tested by the Hosmer-Lemeshow test for significance, and results were expressed as odds ratios (ORs) and 95% confidence interval (95% CI).
The overall diagnostic accuracy of the ELF test was determined by calculating the area under the receiver operating characteristic (AUROC) curve. The sensitivity, specificity, positive and negative predictive values as well as likelihood ratios were calculated for the best cutoff value identified by ROC curve analysis.
The significance threshold was set up at a p value <0.05. All analyses were performed using the SPSS software package (SPSS 21; SPSS Inc., Chicago, IL, USA).
Results
Patient characteristics
We recruited 82 consecutive patients with a histological diagnosis of NAFLD. Following to Clinical and Laboratory Standards Institute (CLSI) guidelines (19), we also tested 20 healthy donors from the local population to verify adherence to CLSI EP 28A3C (20).
The demographic characteristics of the study population are summarized in Table I. Briefly, the prevalence of metabolic syndrome was 20.3%, while that of diabetes was 15.8%. All stages of fibrosis were represented in our cohort, and 18.3% of patients had significant fibrosis at liver biopsy. NASH was present in 18 out of 82 (22%).
Demographic and clinical characteristics of patients in study
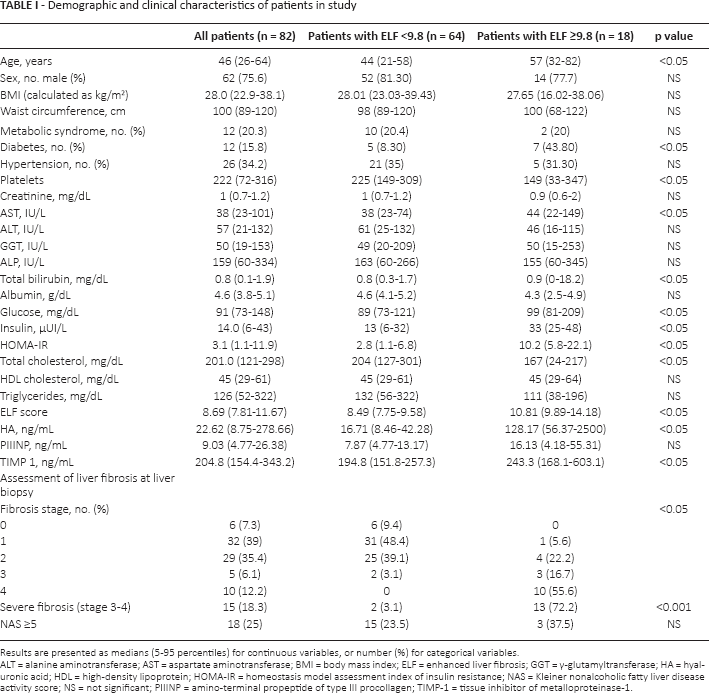
Results are presented as medians (5-95 percentiles) for continuous variables, or number (%) for categorical variables
ALT = alanine aminotransferase; AST = aspartate aminotransferase; BMI = body mass index; ELF = enhanced liver fibrosis; GGT = γ-glutamyltransferase; HA = hyaluronic acid; HDL = high-density lipoprotein; HOMA-IR = homeostasis model assessment index of insulin resistance; NAS = Kleiner nonalcoholic fatty liver disease activity score; NS = not significant; PIIINP = amino-terminal propeptide of type III procollagen; TIMP-1 = tissue inhibitor of metalloproteinase-1.
We also compared demographic and clinical features between patients with not-severe fibrosis and severe fibrosis at an ELF score cutoff of 9.8, according to the manufacturer's guidelines (Tab. I). Patients with ELF >9.8 had significantly lower platelet counts (p<0.05) and total cholesterol (p<0.05), higher levels of glucose (p<0.05), basal insulin (p<0.05) and HOMA (p<0.05) and higher prevalence of severe fibrosis (p<0.001).
The stage of fibrosis was significantly associated with ELF score (Spearman's rho = 0.483, p<0.001) (Fig. 1).
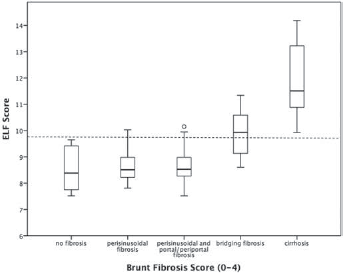
Mean values for enhanced liver fibrosis (ELF) scores and liver fibrosis in nonalcoholic fatty liver disease (NAFLD) patients by fibrosis stage. Dashed line represents the ELF cutoff of 9.8.
To assess potential factors influencing the ELF score, we performed a binary logistic regression analysis. The multivariate analysis included the following variables: age, glucose, insulin, HOMA, total cholesterol and adjustment for sex of patient. Only basal insulin levels appeared to have a correlation with ELF score >9.8, with OR = 1.14 (95% CI, 1.02-1.28; p = 0.018).
Relationship between ELF score and liver fibrosis
To assess the capability of ELF test to assess the presence of severe fibrosis, we compared patients with stage 0-2 fibrosis with patients with stage 3-4 fibrosis. Patients with stage 3-4 fibrosis were older, had lower platelet counts, albumin, alanine aminotransferase (ALT) and total cholesterol levels. Moreover, stage 3-4 patients had significantly higher fasting glucose and insulin levels, higher insulin resistance as assessed by homeostasis model assessment index of insulin resistance (HOMA-IR) score and a higher percentage of diabetes diagnoses (Tab. II).
Demographic and clinical features of patients with no or mild (stages 0-2) vs. severe fibrosis (stages 3-4)
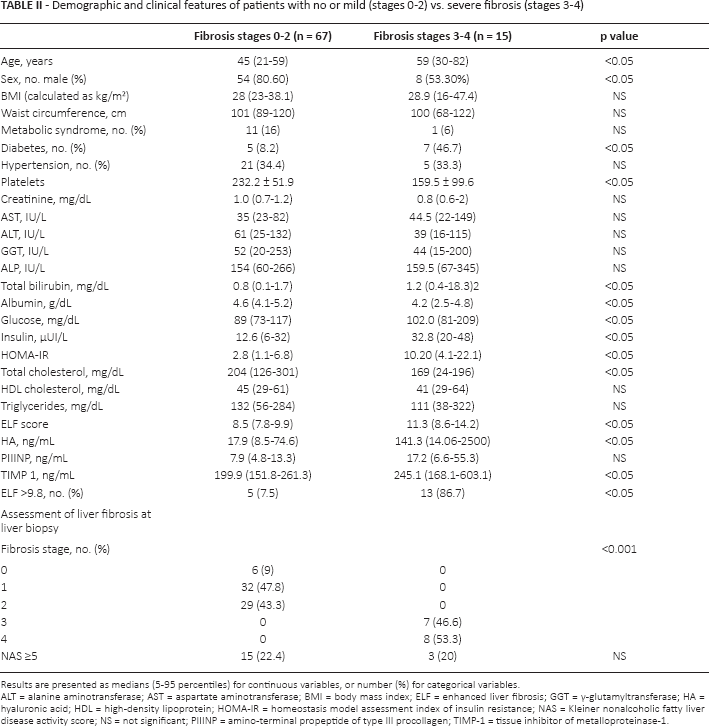
Results are presented as medians (5-95 percentiles) for continuous variables, or number (%) for categorical variables
ALT = alanine aminotransferase; AST = aspartate aminotransferase; BMI = body mass index; ELF = enhanced liver fibrosis; GGT = γ-glutamyltransferase; HA = hyaluronic acid; HDL = high-density lipoprotein; HOMA-IR = homeostasis model assessment index of insulin resistance; NAS = Kleiner nonalcoholic fatty liver disease activity score; NS = not significant; PIIINP = amino-terminal propeptide of type III procollagen; TIMP-1 = tissue inhibitor of metalloproteinase-1.
Median ELF score was significantly elevated in patients with severe fibrosis (11.3 vs. 8.5; p<0.05) even if no significant difference was noted regarding NASH status. Among the components of ELF, HA and TIMP-1 circulating levels were significantly higher in stage 3-4 fibrosis.
The ELF score showed a highly significant association with severe fibrosis, with OR = 7.59 (95% CI, 1.55-37.16; p<0.05) at multivariate analysis adjusted for age, sex, diabetes, glucose and basal insulin levels, HOMA and platelets count.
We tested the diagnostic capability of ELF to detect severe fibrosis in patients with NAFLD by ROC curve, the AUROC curve was 0.948 (95% CI, 0.88-1; p<0.001) (Fig. 2), and by applying the cutoff of 9.8, we found a good sensitivity (86.7%; 95% CI, 0.69-1.04) and high specificity (92.5%; 95% CI, 0.86-0.99) with a positive predictive value of 72% and negative predictive value of 97%.
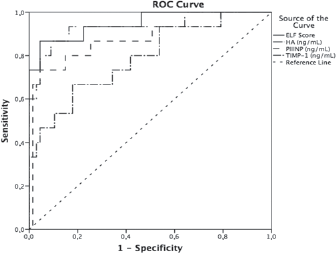
Receiver operating characteristic (ROC) curve analysis. Diagnostic performance of enhanced liver fibrosis (ELF) test score and its components to detect severe fibrosis (stages 3-4). HA = hyaluronic acid; PIIINP = amino-terminal propeptide of type III procollagen; TIMP-1 = tissue inhibitor of metalloproteinase-1.
Discussion
The main finding of our study was that the commercial cutoff value for the ELF test is effective in identifying patients with NAFLD with severe fibrosis. To the best of our knowledge, our study is the first that aimed to assess the diagnostic performance of the commercial ELF score >9.8 in a consecutive NAFLD cohort in clinical practice. In fact, we studied the ELF in a prospective unselected consecutive NAFLD cohort recruited in a real practice setting.
In our series, the rate of false positives was 5/18 (27.7%), moreover the ELF cutoff of >9.8 correctly identifying the presence of severe fibrosis (stages 3-4) in 13 of 18 patients (72.3%), and the test correctly excluded 62 of 64 patients without severe fibrosis (96.8%). We also found a strong correlation of ELF score with severity of fibrosis in the multivariate analysis, with an OR of 7.59 (95% CI, 1.55-37.16; p<0.05).
Our results were similar to and confirmed a recent report by Fagan et al on mixed chronic liver disease (21). Also in our series, with similar figures to those of Fagan et al, the highest percentage of patients with a false positive ELF score (4/5; 80%) had stage 2 fibrosis, pointing to at least 2 possible explanations: (i) a possible misclassification of liver biopsy results or (ii) the high value of ELF score may reflect the active extracellular matrix turnover of the fibrogenic process. Moreover, in the cohort studied by Fargan et al, the presence of liver steatosis was associated with a lower misclassification of liver fibrosis stage, confirming the reliability of the ELF score in NAFLD patients.
Recently some authors have outlined the need for different and specific ELF test cutoff for the diagnosis of severe fibrosis in patients with chronic hepatitis (22, 23). This is not surprising, as the original diagnostic algorithm was built on a broad but heterogeneous population of patients with chronic liver diseases showing a diagnostic variability according to etiology. Despite liver fibrosis being a common feature of almost all chronic liver diseases in the advanced stages, differences in fibrosis distribution (e.g., periportal fibrosis in chronic hepatitis C or perisinusoidal fibrosis in NAFLD) and potential confounding effects deriving from necroinflammation and apoptosis could explain performance variability. Our study confirmed the capability of manufacturer's ELF cutoff score to identify patients with progressive NAFLD due to higher stages (stages 3-4) of fibrosis.
Our study had some limitations: few lack an independent validation within a separate cohort, a possible tertiary center recruitment bias, and not yet data regarding the prognosis of the cohort since the cross-sectional observational design of this study could not sort out these points. However, the available literature on ELF performance in other chronic liver diseases and the possibility of using a commercial kit on a large-scale population may encourage a future validation in an independent cohort.
Considering the high prevalence of fatty liver in the general population and the epidemic levels of diabetes and obesity, a noninvasive assessment of progressive disease is an urgent need for public health stakeholders and physicians (24). The availability of a noninvasive tool to detect liver fibrosis may optimize the use of liver biopsies and may be helpful in effectively monitoring the disease in clinical practice and in clinical trials.
In conclusion, the high prevalence of NAFLD and the rising cost of health care systems both drive the need for screening tests to identify patients with advanced fatty liver disease requiring specific treatments and follow-up. Our data could support a wider use of the ELF test in clinical practice, although specifically designed studies on large populations are needed to confirm the test's efficacy as a screening method.
Footnotes
Financial support: This study was supported by grants PRIN 2010-2011 (prot. no. 2010C4JJWB) (to A. Grieco) and Catholic University Linea D1 (to A. Grieco) and Linea D3 (to A. Gasbarrini and A. Grieco). Siemens Healthcare Diagnostics, Germany, provided the reagents for the ELF test (to C. Z.).
Conflict of interest: The authors declare that they have no competing interests.
