Abstract
Background
Several studies have raised the issue of the inadequacy of CellSearch® to detect the entire pool of circulating tumor cells (CTCs) from blood of cancer patients, suggesting that cells expressing low levels of epithelial cell adhesion molecule (EpCAM) are not recognized by the capture reagent. In this exploratory study, we aimed to evaluate the status of EpCAM in CTCs isolated from a group of metastatic colorectal cancer patients, in 40% of whom, CTC had been found to be undetected by the CellSearch® system.
Methods
CTCs were analyzed using both a microfiltration method (ScreenCell) and CellSearch® in parallel. Furthermore, since EpCAM exists in 2 different variants, we investigated the presence of both its intracellular domain (EpICD) and extracellular domain (EpEX) through immunofluorescence staining of CTCs on filters.
Results
Results from immunofluorescence experiments demonstrated that, overall, EpICD and/or EpEX was expressed in 176 CTCs detected by ScreenCell, while the CellSearch® system was able to capture only 10 CTCs.
Conclusions
This is the first demonstration that the low sensitivity of CellSearch® to detect CTCs in colorectal cancer patients is not due to the lack of EpCAM.
Introduction
Circulating tumor cells (CTCs) released into the bloodstream from both primary and metastatic tumors, represent a precious source of biological material and may provide important information about disease, tumor progression and therapy response (1). However, CTCs are relatively rare compared with other blood cells (2) and are highly heterogeneous (3), thus restricting their use in a liquid biopsy. Therefore, enrichment and detection strategies play a crucial role in the isolation of these cells and represent the limiting step in CTC analysis.
Over the years, several methods have been developed to isolate CTCs from peripheral blood (4), and the development of microfluidic devices reflects the need for highly sensitive, efficient and reproducible procedures (5, 6). To date, the CellSearch® system, an epithelial cell adhesion molecule (EpCAM)–based method, is the only one approved by US Food and Drug Administration (FDA) for CTC counts in metastatic breast, colon and prostate cancer. Nevertheless, studies have clearly shown that the yield of CTCs by CellSearch® is much lower in colon than in breast or prostate cancer (7). Furthermore, it has been widely suggested that EpCAM-based methods are poorly specific to detect CTCs expressing low levels of EpCAM (8).
To better understand this issue, the aim of this exploratory study was to evaluate the status of EpCAM in CTCs undetected through the CellSearch® system in a group of metastatic colorectal cancer (mCRC) patients. For this purpose, we analyzed CTCs using both a microfiltration method (ScreenCell) and CellSearch® in parallel. Furthermore, since EpCAMs exist in 2 different variants, one full-length and the other truncated (9), the presence of the antigen on cell surface was investigated by immunofluorescence staining on CTCs isolated by ScreenCell, using antibodies directed against both the intracellular domain (EpICD) and extracellular domain (EpEX) of EpCAM.
Patients, materials and methods
Patients and blood collection
Blood samples from 10 patients treated for mCRC at the Policlinico Umberto I of Rome, were collected at baseline before the beginning of a new line of therapy. Patient demographics and baseline characteristics are shown in Table I. Informed consent was obtained from all patients. Two blood samples were drawn from each patient: 7.5 mL was collected in a CellSave preservative tube (Janssen Diagnostic, LLC, Raritan, USA) containing ethylenediaminetetraacetic acid (EDTA) and a cell fixative, maintained at room temperature and processed within 72 hours by CellSearch® (Janssen Diagnostic); and 3 mL was collected in a tube containing K2EDTA, stored at 4°C and processed within 3 hours by ScreenCell (ScreenCell, Sarcelles, France). As negative control, blood samples were also taken from 3 healthy volunteers and processed as above described. Further, 3 blood samples from healthy volunteers were used for cell spiking experiments.
Patient characteristics

PS = performance status.
CellSearch® analysis
For CTC enumeration we employed CellSearch® Epithelial Cell Kit (Janssen Diagnostic), as previously described (10). An event was classified as indicating a CTC when exhibiting the phenotype EpCAM+, CK+, DAPI+ and CD45–.
Depletion of leukocytes and ScreenCell analysis
CTCs were first enriched by leukocyte depletion using Dynabeads CD45 (Invitrogen), following the manufacturer's instructions. Briefly, each blood sample was incubated with Dynabeads for 30 minutes at 2°C with gentle rotation. The tube was then removed from the mixer and placed in a magnet for 10 minutes. The supernatant was transferred into a new tube and processed with ScreenCell Cyto kit (ScreenCell), to isolate fixed CTCs for cytological studies, according to the manufacturer's instructions. Three milliliters of leukocyte-depleted blood was diluted in 4 mL of filtration buffer (FC). After 8 minutes of incubation at room temperature, 7 mL of diluted sample was filtered. After washing with phosphate-buffered saline (PBS), the filter was left on absorbing paper to dry at room temperature.
Triple immunofluorescence staining
After hydration with Tris-buffered saline (TBS) for 10 minutes, the filters were incubated overnight at 4°C with goat polyclonal anti-cytokeratin (CK) 20 (N-13, 1:100; Santa Cruz Biotechnology Inc, Rockford, IL, USA) and mouse monoclonal anti-EplCD (4A7, 1:80; Sigma-Aldrich, St. Louis, MO, USA) primary antibodies. The filters were then washed twice in PBS and incubated with donkey anti-goat Alexa Fluor 647 and donkey anti-mouse Alexa Fluor 488 (Molecular Probes, Eugene, OR, USA) secondary antibodies for 45 minutes at room temperature in the dark. After washing in PBS, the filters were incubated with Alexa Fluor 555-conjugated mouse monoclonal anti-EpEX (VU1D9, 1:50; Cell Signaling Technology, Danvers, MA, USA) for 2 hours at room temperature in the dark. Nuclei were stained with 4′, 6-diamidino-2-phenylindole (DAPI; Invitrogen, Rockford, IL, USA) for 15 minutes at room temperature. All antibodies were dissolved in PBS containing 3% bovine serum albumin (BSA), 3% fetal bovine serum (FBS), 0.001% NaN3 and 0.1% Triton X-100. Finally, the filters were mounted with Prolong-Gold Antifade (Invitrogen) on slides and analyzed using a FV1000 Confocal microscope (Olympus FV1000) equipped with a 60× oil immersion objective. A nucleated event was classified as a CTC when exhibiting the phenotype CK20+, EplCD+ and/or EpEX+, in accordance with the CellSearch® CTC definition.
Cell lines and culture conditions
Two human colon cell lines, HCT-116 and HT-29 (Interlab Cell Line Collection, Genova, GE, Italy), were cultured in McCoy's 5A (Sigma-Aldrich), supplemented with 10% FBS, 1% L-glutamine and 1% penicillin-streptomycin (EuroClone SPA, Pero, MI, Italy). Primary cultures of human fibroblasts (HFs), established from a 1 cm2 full-thickness skin biopsy from a healthy donor, as previously described (11), were maintained in Dulbecco's modified Eagle's medium (DMEM) containing 10% FBS. All cell lines were grown in a humidified incubator at 37°C and 5% CO2.
Western blot analysis
Antibody specificity was tested through Western blot analysis. To this purpose, 2 epithelial cell lines, HT-29 and HCT-116, were used as positive controls for the expression of EpICD, EpEX and CK20, while lysates of HFs were used as negative control. Cells were lysed in RIPA buffer. Total proteins (100 μg) were resolved under reducing conditions by 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to Immobilon-FL membranes (Merck Millipore, Billerica, MA, USA). Membranes were blocked in TBS containing 0.1% Tween 20 (TBS-T) and 5% milk for 1 hour at 25°C and then incubated overnight at 4°C with the following primary antibodies: anti-CK20, anti-EpEX (VU1D9; Invitrogen), anti-EplCD and mouse monoclonal anti-tubulin (Sigma-Aldrich). Membranes were then incubated with the appropriate horseradish peroxidase (HRP)–conjugated secondary antibody (Santa Cruz Biotechnology) for 1 hour at 25°C. Bound antibody was detected by enhanced chemiluminescence detection reagents (Pierce Biotechnology Inc., Rockford, IL, USA), according to the manufacturer's instructions. Tubulin served to estimate the protein equal loading. The experiments were performed in triplicate to verify results.
Spiking experiments
To evaluate the antibodies' sensitivity on filters, spiking experiments were performed. HT-29 cells were transferred into 3 blood samples (3 mL) from healthy volunteers. The cells were first enriched by leukocyte depletion using Dynabeads CD45 and then isolated using a ScreenCell Cyto kit, as described in the section “Depletion of Leukocytes and ScreenCell Analysis,” above. Triple immunofluorescence staining was carried out on cells isolated on the filter.
Statistical analysis
Quantitative analysis of the expression of EpICD and EpEX was performed by counting cells exhibiting CK20 expression. Results from 3 microscopic fields for each filter were expressed as percentages of positive cells and reported as means ± standard deviation. Data were analyzed using Student's t-test for paired samples assuming a 2-tailed distribution. Only p values <0.05 were considered statistically significant.
Results
Evaluation of antibodies' specificity and sensitivity
In the epithelial cell lines, all of the primary antibodies specifically recognized their target (a band corresponding to a molecular weight [MW] of 40 kDa for EpCAM and 46 kDa for CK20), without the presence of nonspecific bands. Moreover, no signal was detected with any of our antibodies in the mesenchymal cells (HFs), which do not express EpCAM or cytokeratins (Fig. 1).
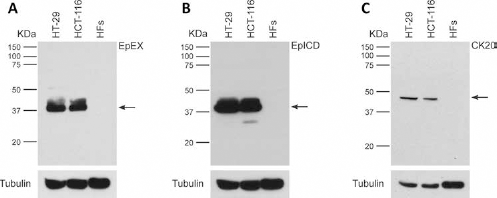
Western blot analysis of EpEX, EpICD and CK20 immunoreactivity in whole cell lysates of the epithelial cell lines HT-29 and HCT-116. The mesenchymal primary cultures of human fibroblasts (HFs) were used as negative control. (
Triple immunofluorescence staining experiments were performed on 3 filters from healthy volunteers, to evaluate the sensitivity of the antibodies against CK20, EpEX and EpICD on filters. All cells isolated by ScreenCell were found to be expressing CK20, EpICD and EpEX (Fig. 2).
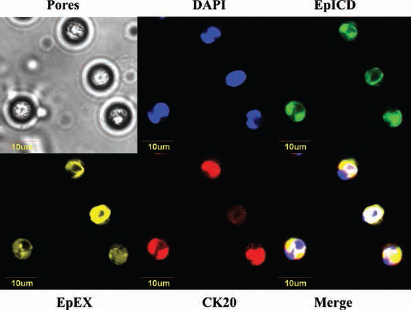
Illustrative images of triple immunofluorescence assay on HT-29 human colon cells. Positive staining for EpICD, EpEX and CK20. CK20 = cytokeratin 20; EpCAM = epithelial cell adhesion molecule; EpEX = EpCAM extracellular domain; EpICD = EpCAM intracellular domain.
CTC detection: alternative methods in comparison
Blood samples from 10 patients with mCRC were analyzed by CellSearch® for CTC enumeration. In 4 of 10 patients (40%) CTCs were found to be absent or were undetected, while in 6 of 10 patients (60%) CTCs were detected, ranging in number from 1 to 2 per 7.5 mL of blood.
In the same patients, we performed a leukocyte depletion followed by CTC isolation through ScreenCell assay. Triple immunofluorescence staining experiments were carried out on filters to evaluate the coexpression of CK20 (marker of colorectal cancer cells), EpICD and EpEX. For each filter, 3 microscopic fields were analyzed, and we were able to detect 176 CK20+ CTCs, with a range in number from 4 to 33 per 3 mL of blood. EpICD was found expressed in 98.9% ± 4.5% out of the 176 CTCs analyzed, while EpEX was found in 87.0% ± 19.6% out of the 176 CTCs analyzed (difference in EpICD and EpEX expression in CTCs was found to be statistically significant, p = 0.003). Specifically, in 3 of 10 patients, all CTCs expressed both EpEX and EpICD (Fig. 3A). In 6 of 10 patients, all CTCs expressed EpICD, while variably expressing EpEX (Fig. 3B). A subpopulation of EplCD–/EpEX+ CTCs was only identified in 1 patient, although in a very small fraction of the CTCs (2.3%) (Fig. 3C).
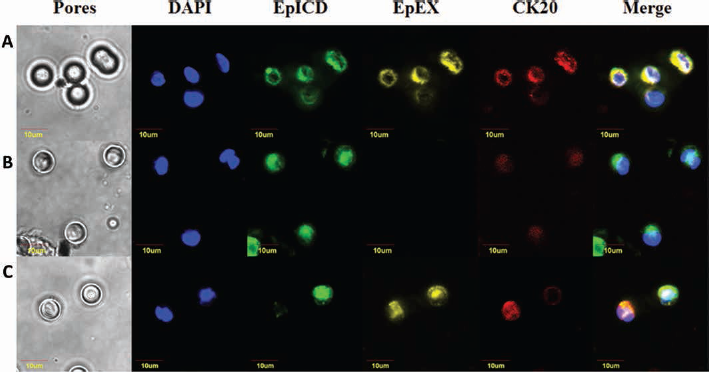
Illustrative images of triple immunofluorescence assay on circulating tumor cells. (
No correlation was found between CTCs detected by CellSearch® and EpEX expression at immunofluorescence. In fact, 64 EpEX+ CTCs were found in 4 patients with no CTCs with CellSearch® analysis. In 2 patients with 1 CTC (total number of CTCs = 2) with CellSearch®, we found 24 EpEX-expressing CTCs using ScreenCell. Lastly, in 4 patients with 2 CTCs with CellSearch® (total number of CTCs = 8), 62 cells detected by ScreenCell were found to be expressing EpEX. Table II shows the detailed results.
CTC detection using CellSearch® and ScreenCell devices
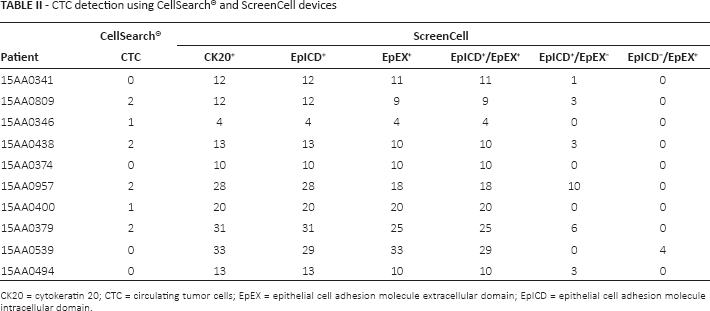
CK20 = cytokeratin 20; CTC = circulating tumor cells; EpEX = epithelial cell adhesion molecule extracellular domain; EpICD = epithelial cell adhesion molecule intracellular domain.
CTCs from healthy volunteers were not found, using either CellSearch® or ScreenCell (data not shown).
Discussion
The basic concept of inadequacy of EpCAM-based methodology to detect the entire pool of CTCs relies on the fact that the cells expressing low levels of EpCAM are not recognized by capture reagents (12), particularly in mCRC where CTCs are found to be undetected by the CellSearch® system in a significant proportion of patients (13). Efforts have been made to clarify the biological reasons for the CellSearch® failure, addressing the crucial question of CTC heterogeneity (14) and studying the potential mechanisms that might contribute to the dynamic expression of EpCAM (15).
In several studies the loss of EpCAM was frequently attributed to an epithelial mesenchymal transition (EMT) program activation leading in turn to a down-regulation of epithelial markers (8), although it has been demonstrated that CTCs undergoing EMT might maintain selected epithelial traits (15). This process, as well as the transcription factors regulating EMT induction or epigenetic mechanisms, could explain the undetection rate of CTCs by CellSearch®. In our work, a total of 10 CTCs were found through CellSearch®, and all patients had a CTC number below the cutoff value. These data conflicted with those obtained using the ScreenCell device in the same group of patients, where all CTCs isolated were found to express EpICD and/or EpEX.
Further explanations could be taken into account, from a immunochemistry point of view. It has already been shown that CTC immunomagnetic enrichment is critically dependent on the EpCAM clone used (16). Furthermore, it has been suggested that different conformational states of EpCAM might mask the epitopes and negatively affect the antibody binding (17). However, the transmembrane glycoprotein is also a substrate for different proteases which cleave EpCAM at the extracellular domain, resulting in ectodomain shedding (18). It is clear that use of antibodies directed against the domain containing cleavage substrate could give rise to false negative and thus to misinterpretation of results. Particularly, a proteolytic cleavage between Arg80 and Arg81 might lead to the shedding of the first epidermal growth factor (EGF)–like domain containing the epitope (amino acids 27-59) which is recognized by VU1D9, the EpCAM clone used as a capture reagent for CTCs by CellSearch®. This would be true even if the results from immunofluorescence experiments performed on filters employing the same clone (VU1D9) used as the CellSearch® capture reagent, demonstrated that 89.1% of the CTC population expressed EpEX.
In addition, a small proportion (2.3%) of EpEX+ CTCs was found not to express EpICD. Recently, Seeber et al, using immunohistochemistry on tissue specimens, demonstrated that 164 out of 640 colorectal patients (25.6%) recruited in the study had lost the expression of EpICD, retaining EpEX at the plasma membrane. This might have been due to a regulated intramembrane proteolysis (RlP)-independent activation of EpCAM, resulting in a selective degradation of EpICD and retention of EpEX on cell surfaces (9).
In light of the above-described in “Results” for this present study we believe that the challenge might be the conjugation of EpCAM to nanoparticles, more than the clone chosen. It has been recently suggested that the binding affinity of an antibody does not always match the cell capture efficiency (19). However, it is clear that when an antibody is immobilized on magnetic core nanoparticles as well as a solid-phase surface, its binding activity is usually less than that of a bead-unconjugated antibody (20). From a chemical point of view, in this regard, some steric hindrance certainly plays a key role. Indeed, the distance and/or the covalent bond between the antibody and the bead surface, not leaving out also the nanoparticle diameter, might limit the antibody orientation (21).
In conclusion, we demonstrated for the first time that, at least in colorectal cancer, EpCAM is retained at the CTC level, shedding light on the fact that the undetection rate of CTC reported in some cancer types is not a matter of EpCAM, and that other mechanisms might contribute. Further investigations are needed to better understand the issue. The optimization of the EpCAM-beads complex, through decreasing the steric hindrance, and/or the choice of antibody clone, preferably directed against a domain which is often not a cleavage substrate, might improve the performance of CellSearch®, as well as of other EpCAM-based methods, in order to drive the CTC-based liquid biopsy into current clinical practice.
Footnotes
Financial support: This work was supported by the Italian Ministry for University and Research (MIUR) (grant number C26N147NSX) and A.R.Ger.On. Onlus.
Conflict of interest: The authors have no conflicts of interest to declare.
