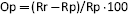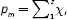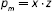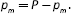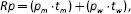Abstract
Laboratory tests are frequently overused and have elevated inappropriateness rates. We previously developed a model to investigate the rate of utilization of tumor markers (TMs) in outpatients as an indirect indicator of inappropriateness. The model was based on the comparison between the number of actually ordered and expected tests, with the latter estimated on the basis of both epidemiological data and recommendations of available clinical practice guidelines.
In this paper we propose an algorithm to distinguish prevalent cases without evidence of disease from those with metastatic spread, on the basis of both epidemiological and clinical information. The algorithm allows for a more precise prediction of the expected TM requests per year, to be compared with the actual number of requested TMs in order to assess possibly inappropriate overordering rates. Moreover, the implementation of the algorithm renders the epidemiologically based model more flexible to develop accurate indicators for appropriateness in the use of TMs in different stages of disease and for different clinical questions. A practical application with CA15.3 requests in breast cancer is presented.
Introduction
Evidence shows that laboratory tests are frequently overused and have elevated inappropriateness rates that do not seem to decline over the years (1). In a situation of widespread overutilization, the control of appropriateness is impaired by the high volume of requests, the variety of health care professionals who may order laboratory tests and the multiplicity of clinical conditions and diseases in which the laboratory may have a potential diagnostic role. However, the most critical factor in the monitoring of appropriateness is the poor clinical information available on test requests of outpatients (2). Notably, the harms related to false positivity occur more frequently with outpatients than with inpatients, since an increased risk of labeling healthy people as sick – or at risk of developing a serious disease – arises in the general population (3). The monitoring of overuse and inappropriateness of laboratory tests in outpatients should be based on a method suitable to be applied to very large records or orders (e.g. at regional or national level) and which is sufficiently flexible to be used in different clinical scenarios.
We previously developed an original model to investigate the rate of utilization of tumor markers (TMs) in outpatients (4). The model was based on the comparison between the number of tests ordered and those expected, with the latter estimated on the basis of both epidemiological data (5) and recommendations of clinical practice guidelines (CPGs) (6). In our previous study, the number of TMs ordered (the reported number of requests per year [Rr]) was compared with the expected number of requested TMs (the predictable number of requests per year [Rp]), assuming that (i) prevalent cases were represented by patients with the most probable clinical status, and (ii) the highest limit of acceptable appropriateness was the determination of the number of TMs recommended by CPGs in the most probable clinical status (6). The percentage of overprescription (Op)–that is, the surplus number of TMs ordered with reference to the expected number of TMs appropriately ordered – was expressed by the formula:
To predict the most probable clinical status of prevalent cases, we considered the ratio between mortality and prevalence, assuming that mortality is a surrogate indicator of relapsed metastatic or advanced disease. For instance, 1.80 and 1.79 patients in every 100 prevalent cases die annually of breast and prostate cancer, respectively (7-9), and therefore, we assumed that prevalent cases are mainly represented by patients followed up without evidence of disease. Thus in breast and prostate cancer, the determination of 2 TMs per year in patients during follow-up can be considered as the highest limit of acceptable appropriateness (6). Conversely, 77.14 patients with pancreatic cancer die annually for every 100 prevalent cases (7-9), and therefore we assumed that prevalent cases are mainly represented by patients with metastatic disease probably treated with anticancer agents. Thus, considering 1 determination before every chemotherapy course, 12 TM determinations per year, can be accepted as the highest limit of acceptable appropriateness in patients with pancreatic cancer (6).
Using similar epidemiological assumptions, a high level of overordering of CA19.9 and CA125 was observed; conversely, the number of CA15.3 actually ordered (1,078,864) was quite similar to the expected one (1,044,470) with a limited 3.3% rate of putative inappropriate requests (4). However, determination of TMs twice per year during the follow-up of asymptomatic patients without evidence of disease is too permissive an assumption, as CPGs are increasingly stringent on the use of CA15.3 testing in breast cancer. In fact, although presently available CPGs are in favor of using the marker in monitoring the response to treatments of patients with metastatic disease, they either recommend against, or do not consider at all, CA15.3 determination in following up asymptomatic women without evidence of disease (10). Prevalent breast cancer cases embrace women followed up without evidence of disease after treatment of the primary tumor, patients with metastatic disease and a relatively minor number of incident cases. In the present study, we showed that the implementation of the algorithm allows for the application of the model developed, also when prevalent cases include patients with different disease conditions in which TMs may or may not be recommended.
Material and methods
Prevalence rates recorded in tumor registries gather all cases having or having had a given malignancy, but not their disease status. Therefore, the number of prevalent cases with advanced disease may only be indirectly inferred on the basis of data regarding mortality and survival rates.
Assuming x as the number of deaths per year for a given type of cancer, and z to be the median survival time in years of patients with metastatic spread, for each year, there are x cases that will die, which should be summated to x cases that will die during the following year, and so forth, as a biological application of Little's law (11).
Then, the number of prevalent cases affected by metastatic disease (pm) every year will be:
that is,
If the overall prevalence of the disease is P, the prevalent cases without metastasis will be
According to these assumptions, it will be possible to derive adjustable and rather accurate estimates of the expected requests for TMs. In fact, the total predictable requests Rp previously considered will become
where tm and tw are the tests to perform in 1 year for the follow-up of each patient with and without metastases, respectively.
Calculations were performed and formulas tested using data published in the previous survey (4).
Results and discussion
The presented algorithm allows for a flexible tuning of the predictable requests according to the clinical conditions of the patients, thus leading to a more accurate estimate of the percentage of the overprescription. The requests for CA15.3 in women with breast cancer was used as the test scenario.
If the recommendations of CPGs were followed (10), CA15.3 would not be requested during the follow-up of patients without evidence of disease, but it would be used only to monitor treatment of patients with metastatic disease. In this case, tw and consequently all of the second term of Equation [5] become 0. Therefore, the number of expected CA15.3 requests Rp will be only (pm·tm) and can be estimated on the basis of data regarding mortality and survival rates of women with metastatic breast cancer (12, 13) as follows: in Italy, 12,000 women die annually of breast cancer; considering that median survival of women with metastatic breast cancer is 3 years, approximately 36,000 (3 × 12,000, according Eq. [3]) prevalent cases of breast cancer would be affected by metastatic disease (486,232 patients would be without evidence of disease, according to Eq. [4]; Fig. 1). Assuming that every case with metastatic disease is monitored monthly with CA15.3 for therapy response, the expected number of CA15.3 determinations would be 432,000 (36,000 × 12, according to Eq. [5]). Therefore, the reported number of CA15.3 requested per year (1,078,864) is considerably higher than the expected one (432,000), with an overprescription rate of 149.7% in comparison with the rate of 3.3% obtained considering all prevalent cases as patients followed up without evidence of disease (4). Figure 1 illustrates the logical flow chart followed to estimate CA15.3 overordering in prevalent cases, considering those cases according to different estimated disease conditions.
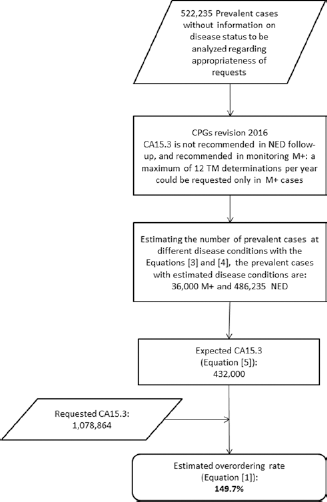
Logical flow chart followed to estimate CA15.3 overordering in prevalent cases in Italy according to different estimated disease conditions of the patients involved. CPG = clinical practice guideline; M+ = patients with metastatic disease; NED = patients without evidence of disease; TM = tumor marker.
We realize that to consider as appropriate 12 tests per year in patients with metastatic disease is a fairly broad assumption, as only some of the patients with advanced disease are treated with chemotherapy regimens and TMs should not necessarily be requested before every cycle (10). Nevertheless, the overordering rate appears high in spite of the broad criteria applied. This simulation corroborates the innovative value of the model developed, here implemented with a proper algorithm, to estimate inappropriateness of TMs by surveying their overutilization. The adaptability of the model to variations of epidemiological information suggests that it is flexible enough to be used to develop indicators for appropriateness in the use of TMs, and seems suitable to be used to monitor the impact of new CPGs on clinical practice, thus aiding in addressing educational interventions toward ordering behaviors still demanding improvement.
Footnotes
Acknowledgements
We wish to thank the Ministry of Health, New National Health IT System (Direzione generale della digitazione, del sistema informativo sanitario e della statistica – Elaborazione a cura dell'Ufficio III – NSIS, flusso di specialistica ambulatoriale – Art. 50 della legge n. 326/2009) for allowing us to use the requested biomarker data. We would like to thank the Interregional Biomarkers Working Group instituted by the Health Commission of the Italian Permanent Conference for Relations Between State, Regions and Autonomous Provinces of Trento and Bolzano (Gruppo Tecnico Interregionale “Miglioramento della Pratica Clinica per l'Utilizzo dei Biomarcatori in Oncologia,” Commissione Salute – Conferenza Permanente per i rapporti tra Stato, Regioni e Province Autonome di Trento e Bolzano) for productive discussions regarding the project proposal. We would also like to thank AIRTUM (Associazione Italiana Registri Tumori) for providing data on cancer incidence and prevalence in Italy. And we would like to thank Antonette Leon for editorial assistance in the manuscript preparation.
Financial support: This work was supported in part by the Italian Association for Research on Cancer (AIRC; Grant Special Program Molecular Clinical Oncology, 5 × 1000, No. 12214).
Conflict of interest: All of the authors declare they have no conflicts of interest.