Abstract
Background
DNA mismatch repair deficiency is an important molecular mechanism of genetic instability in gastric cancer, and a high instability at microsatellites is associated with favorable prognosis. We compared mononucleotide and dinucleotide microsatellite instability (MSI) marker panels in 56 paired gastric tumor and normal samples.
Methods
The mononucleotide marker panel (mono panel) consisted of 8 markers: BAT25, BAT26, BAT40, BAT-RII, NR21, NR22, NR24 and NR27. The dinucleotide marker panel (di panel) contained D2S123, D5S346, D17S250, D17S261, D17S520, D18S34 and D18S58. The NCI panel was used as reference panel.
Results
Among 13 gastric tumors showing no hMLH1 or hMSH2 expression, 8 MSI-H (high) and 5 MSI-L (low) were identified. The analytical sensitivities of the NCI, mono and di panels to detect unstable MSI were 61.5% (8/13), 76.9% (10/13) and 84.6% (11/13), respectively. The size change of allele shift was statistically greater in the mono panel than in the di panel (p = 0.02 by Mann-Whitney U-test). The BAT40 (69.2%, 9/13) and D18S34 (76.9%, 10/13) markers showed high sensitivity for determination of MSI status.
Conclusions
To improve the detection rate of MSI in gastric cancer with loss of hMLH1 or hMSH2 expression, the kind of MSI marker may need to be considered more, instead of the repetitive type of marker. Thus, an MSI panel designed with a combination of both BAT40 and D18S34 is suggested for providing more accurate and sensitive MSI analysis in gastric cancer.
Introduction
Gastric cancer is the fourth most common cancer and the second leading cause of cancer-related mortality in the world (1). Updated studies on molecular profiling of gastric cancer reveal heterogeneous characteristics for gastric cancer and define the subtypes on the basis of genomics, gene expression and amplification patterns (2). Recently, the Cancer Genome Atlas (TCGA) project proposed a molecular classification dividing gastric cancer into 4 subtypes. Among them, microsatellite-unstable tumors show elevated mutation rates, including mutations of genes encoding targetable oncogenic signaling proteins; genomically stable tumors, which are enriched for the diffuse histological variant and mutations of RHOA or fusions involving RHO-family GTPase-activating proteins (3). Microsatellite instability (MSI) is defined as alterations in the lengths of microsatellites due to the deletion or insertion of repeating units to produce novel-length alleles in tumor DNA when compared with normal DNA from the same individual (4). MSI can be classified using a panel of 5 markers recommended by the National Cancer Institute (NCI) in 1998 (5). These Bethesda Guidelines have been revised to reflect the fact that MSI-H (high) can be defined when instability is observed at a mononucleotide locus, MSI-L (low) has limited instability at only a dinucleotide locus, and mononucleotide repeats were shown to be more sensitive compared with dinucleotide loci in detecting MSI (6). A meta-analysis of the association between MSI-H and gastric cancer prognosis indicated that MSI-H gastric cancers have an improved prognosis, accompanied with a reduced risk of lymph node (LN) metastasis, tumor invasion and mortality (7). Therefore, it is crucial to perform an MSI analysis using highly sensitive and specific markers that precisely reflect the MSI status.
In this study, we customized and compared mononucleotide and dinucleotide marker panels for classifying MSI status in 56 paired gastric tumor and normal samples.
Materials and Methods
Samples and nucleic acid extraction
A total of 56 paired primary gastric tumor and normal samples were obtained from surgically dissected patients with gastric cancer between 1999 to 2016 at the Daejeon St. Mary's Hospital, Daejeon, Republic of Korea. Loss of the expression of the human mutator L homologue 1 (hMLH1) or the human mutator S homologue 2 (hMSH2) was detected in 13 tumors through immunohistochemistry (IHC), resulting in defective DNA mismatch repair (MMR) (BD Pharmingen, San Diego, CA, USA) (8). Genomic DNA was extracted from all 56 paired gastric tumor and normal samples using the QIAmp DNeasy Blood and Tissue kit (Qiagen, Hilden, Germany) according to tissue protocol. The study protocol was approved by the Institutional Review Board of The Catholic University of Korea, including written informed consent from all participants for clinical and molecular analyses.
MSI marker selection and MSI definition
MSI markers were selected to optimize the mononucleotide and dinucleotide marker panels by the repetitive type of MSI. The mononucleotide marker panel (mono panel) consisted of 8 mononucleotide markers: BAT25, BAT26, BAT40, BAT-RII, NR21, NR22, NR24 and NR27. The dinucleotide marker panel (di panel) included D2S123, D5S346, D17S250, D17S261, D17S520, D18S34 and D18S58. These markers were recommended when used in 2 or more studies or recommended due to their clinical significance (9-15). The NCI panel consisting of 2 mononucleotide (BAT25 and BAT26) and dinucleotide (D2S123, D5S346, D17S250) markers was used as reference panel (5). Allelic sizes to match the tumor and normal samples were compared and considered to be MSI unstable if there was a shift of 3 bp or more in the tumor allele (16). All tumors with ≥1 unstable markers were regarded as having some degree of instability and were classified as MSI unstable, whereas tumors were classified as microsatellite stable (MSS) when there were no instable markers. Tumors with ≥40% or with <40% instability were categorized as MSI-H or MSI-L, respectively (17).
MSI analysis
Fifteen MSI markers were divided into groups of 5 each and coamplified in 3 reaction tubes. Briefly, polymerase chain reaction (PCR) was carried out at a final volume of 16 μL containing 2× Taq PCR Pre-Mix (Solgent Inc., Daejeon, Republic of Korea), 0.2 mM of each primer pair and about 50 ng of genomic DNA on a 2720 Gene-Amp PCR system (Applied Biosystems, Foster City, CA, USA). After initial denaturation at 95°C for 5 minutes, the cycling PCR steps were as follows: 95°C for 30 seconds, 60°C of annealing for 30 seconds, and 72°C for 30 seconds, for 30 cycles. A final extension was set to 72°C for 10 minutes. After successful amplifications, 1 μL of PCR products was mixed with 9 μL of Hi-Di formamide and 0.3 μL of GeneScan 500 LIZ Size Standard (Applied Biosystems). PCR amplicons were analyzed using GeneMapper 4.0 Software (Applied Biosystems) under denaturing conditions with an internal LIZ standard on an ABI 3130XL genetic analyzer (Applied Biosystems).
Statistical analysis
An assessment of the normality of data was made using the Kolmogorov-Smirnov and the Shapiro-Wilk tests. The Levene test was used to assess the equality of variances for a variable calculated for 2 panels. The mean between 2 unrelated groups was estimated by Student's t-test when Kolmogorov-Smirnov or the Shapiro-Wilk test showed normal distribution. The Mann-Whitney U-test was used to compare differences between 2 independent groups when the continuous variable was not normally distributed. The statistical analyses were conducted with MedCalc ver. 12.7.2 (MedCalc software, Mariakerke, Belgium), and a p value of less than 0.05 was considered to be significant.
Results
Determination of MSI status
Among 13 gastric tumors showing no hMLH1 or hMSH2 expression, 8 (62%, 8/13) and 5 (38%, 5/13) were classified as MSI-H and MSI-L, respectively. By the degree of MSI status, all 8 MSI-H tumors could be identified concordantly by 3 panels. However, 2 and 3 different MSI-L tumors were only identified by the mono or di panel, respectively, but not by the NCI panel (Tab. I). The analytical sensitivities of the NCI, mono and di panels to detect MSI-unstable gastric tumors were 61.5% (8/13), 76.9% (10/13) and 84.6% (11/13), respectively. However, there was no statistically significant difference when these 3 sensitivities were compared by Fisher's exact test (p = 0.536). The analytical specificity of all 3 panels was 100% (43/43).
Microsatellite instability analysis based on 5 or more markers used in gastric cancer studies
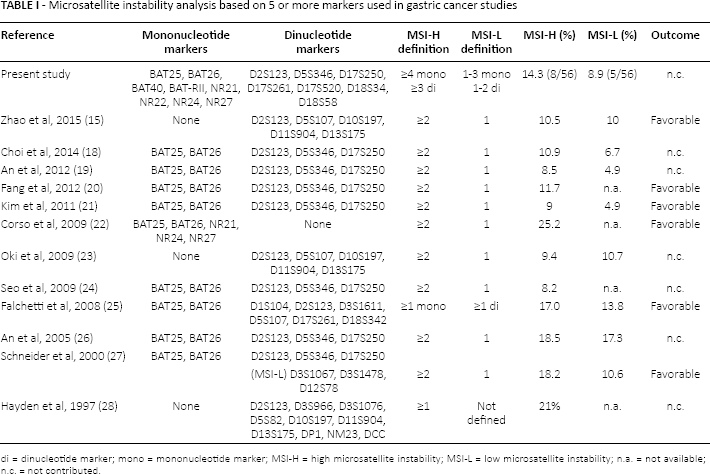
di = dinucleotide marker; mono = mononucleotide marker; MSI-H = high microsatellite instability; MSI-L = low microsatellite instability; n.a. = not available; n.c. = not contributed.
Comparison of the mono and di panels
Estimates for the incidence of unstable MSI in this study ranged from 3 to 10 samples per marker, with a mean of 7.2 samples, depending on each marker. The mean unstable MSI samples per marker was 7.8 (range 6-9) for the mono panel, and 6.6 (range 3-10) for the di panel. The number difference of the MSI-unstable samples per marker between the mono and di panels was normally distributed by the Shapiro-Wilk test (p = 0.054 in mono; 0.320 in di) and was not statistically significant (p = 0.165 by Student's t-test) (Fig. 1).
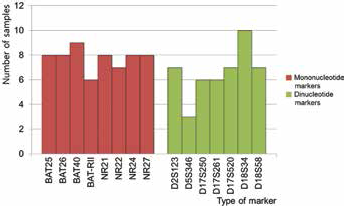
Number of tumor samples scored as microsatellite instability (MSI)-unstable depending on each individual MSI marker. The incidence of MSI in gastric cancer ranged from 3 to 10 samples, with a mean number of 7.2 samples, depending on each marker. The mean number of MSI-unstable was 7.8 in the mononucleotide markers panel, while it was 6.6 in the dinucleotide markers panel. The difference in number of MSI-unstable samples per marker between the mono and di panels was not statistically significant (p = 0.165 by Student's t-test).
On the other hand, the size change of allele shift in MSI-unstable gastric tumors was estimated for the markers between the mono (mean 5.1, range 0-21) and di panels (mean 3.3, range 0-13). The difference in allele shift was nonparametric and statistically greater in the mono panel than in the di panel (p = 0.02 by Mann-Whitney U-test), which increased the detection rate in discrimination of MSI status (Fig. 2).
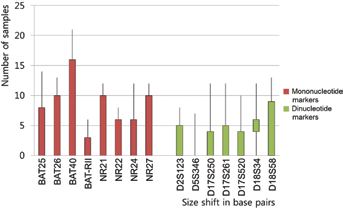
Comparison of the allele size shift between mononucleotide and dinucleotide markers in 13 microsatellite instability (MSI)-unstable gastric cancers with loss of hMLH1 or hMSH2 expression. The size change of allele shift in MSI-unstable gastric tumors was estimated for the markers between the mono (mean 5.1, range 0-21) and di (mean 3.3, range 0-13) panels. The difference of allele shift was statistically greater in the mono panel than in the di panel (p = 0.02 by Mann-Whitney U-test), which increased the detection rate in discrimination of MSI status.
Discussion
No studies directly comparing both mono and di panels to the NCI panel in gastric cancers have been reported before. In this study, both mono and di panels showed better detection rates than the NCI panel, because of higher MSI-L detection. Five MSI-L tumors were detected by mononucleotide BAT40 and BAT-RII as well as by dinucleotide D18S34. If the mono and di panels were used in the interpretation of MSI, 5 of the tumors which were classified as MSS by the panel of NCI panel would have been reclassified as MSI-L. The reclassification to MSI-L does not add value, even though several studies have shown no significant clinicopathological difference between MSI-L and MSS gastric cancers. Determination of the MSI status in gastric cancer is important due to the clinical significance of this phenotype (3). Based on several studies, gastric cancers with MSI-H were associated with significantly better overall survival and a trend toward better disease-free survival compared with MSS/MSI-L gastric cancers after curative surgery (Tab. I). However, our data did not show any association between MSI-H and MSS/MSI-L regarding clinical significance or prognostic outcome. The small sample size (n = 56) and short follow-up duration (median 8.3 months) could have contributed to this discrepancy. Further investigation would require the follow-up of a large number of gastric cancers for a long period of time to establish this association.
Postreplication MMR is an important mechanism for maintaining microsatellite stability through the correction of base change mismatches and insertion/deletion events. Premutational intermediates are identified and processed by heterodimers of the MutS and MutL family of proteins. The MutSα complex (MSH2 and MSH6) can recognize base substitutions as well as single-base insertion/deletion loops (IDLs) in either the parental or nascent strand. On the other hand, MutSβ (MSH2 and MSH3) can recognize IDLs of 2-4 bases in addition to single-base IDLs. MutL heterodimers are recruited by MutS complexes to the mismatch or IDL, and consist of MLH1 dimerized with either PMS2 (MutLα), PMS1 (MutLβ) or MLH3 (MutLy). MutLα is considered as the major MutL complex for fixing of both large and small IDLs, while MutLy and MutLβ have little or no known role in MMR (29).
In this study, the relative MSI sensitivity of the individual markers making up the mono or di panels varied. The BAT40 (69.2%, 9/13) and D18S34 (76.9%, 10/13) markers showed highly sensitivity for determination of MSI status in gastric cancer with loss of MMR protein expression, but even the worst marker was more sensitive than the D5S346 marker of the NCI panel. The D5S346 marker was reported to show relatively lower sensitivity (50%-59%) compared with other dinucleotide markers such as D2S123 (72%-89%) and D17S250 (50%-81%) (30, 31). Moreover, D5S346 and D17S250, as 2 NCI panel dinucleotide markers, showed low sensitivity (33% and 50%, respectively) in early onset gastric cancer (32). Because some individuals have 2 identical forms of this microsatellite, the locus is rendered uninformative for loss-of-heterozygosity analysis (33).
It was widely acknowledged that dinucleotide repeats, of which there are 3 in the NCI panel, were less sensitive and specific and less specific for the detection of MMR deficiencies than mononucleotide markers (6). In stark contrast, mononucleotide markers are relatively stable and tend to be monomorphic. The hMLH1-, hMSH2- or human postmeiotic segregation increased 2 (hPMS2)-mutated tumors lead to pronounced instability at both mononucleotide and dinucleotide markers. However, human mutator S homologue 6 (hMSH6)-mutated tumors were mainly associated with instability at mononucleotide repeats. Interestingly, the hMSH6 protein participates in the correction of base–base mismatches and single nucleotide deletions/insertions only. hMSH6-deficient tumors are usually stable at dinucleotide repeats, and their MSI status would be controversial. Thus, false-negative MSI results may be caused by the use of only dinucleotide markers, particularly for MSH6-defective tumors (34).
In cases without matching normal tissues, the MSI status of the tumors could be determined using mononucleotide markers, comparing the tumor allele sizes with the common allele sizes in the population (35). The dinucleotide repeats in the NCI panel are polymorphic, and matching normal tissue is usually needed for an MSI determination. Although the potential limitations of the reference panel proposed by NCI were described (6), laboratories can use the panel directly or refer to this panel to validate their own set of markers.
Nonetheless, dinucleotide markers are still considered useful to reflect the intention of the NCI panel, because dinucleotide markers would reduce the effects of confounding factors that affect the fidelities of mononucleotide loci. Theoretically, the fidelities of mononucleotide and dinucleotide repeats in the genome are maintained by mechanisms of MMR and other biological procedures – i.e., length alternations in simple repeat sequences might also be driven by some biological procedures in addition to MMR (15). Dinucleotide markers would be obligatorily included, more than mononucleotide markers, to reduce confounding factors that affect the fidelities of mononucleotide loci.
In conclusion, to improve the detection rate of MSI in gastric cancer with loss of hMLH1 or hMSH2 expression, the kind of MSI marker may need to be considered more, instead of the repetitive type of marker. Thus, an MSI panel designed with a combination of both BAT40 and D18S34 is suggested for providing more accurate and sensitive MSI analysis in gastric cancer.
Footnotes
Financial support: No grants or funding have been received for this study.
Conflict of interest: None.
