Abstract
High microsatellite instability (MSI-H)/deficient mismatch repair (dMMR) phenotype is a distinct molecular signature across gastrointestinal cancers characterized by high tumor mutational burden and high neoantigen load. Tumors harboring dMMR are highly immunogenic and heavily infiltrated by immune cells; consequently, they are uniquely vulnerable to therapeutic strategies enhancing immune antitumor response such as checkpoint inhibitors. The MSI-H/dMMR phenotype arose as a powerful predictor of response to immune checkpoint inhibitors with evidence supporting significantly improved outcomes in the metastatic setting. On the other hand, the genomic instability characteristic of MSI-H/dMMR tumors appears to be associated with decreased sensitivity to chemotherapy, and the benefits of standard adjuvant or neoadjuvant chemotherapy approaches in this subtype are being increasingly questioned. Here, we review the prognostic and predictive impact of MMR status in localized gastric and colorectal cancers, and highlight the emerging clinical data incorporating checkpoint inhibitors in the neoadjuvant setting.
Keywords
Introduction
The mismatch repair system is a highly conserved DNA repair mechanism protecting the integrity of the genome against mismatching errors arising during DNA replication. Failure to repair mismatching errors leads to the accumulation of frameshift mutations and generation of several abnormal tumor-specific peptides. These immunogenic neoantigens are recognized as ‘non self’ by T-lymphocytes and trigger a host immune antitumor response. Tumors evolve over time under the selective pressure of the immune system and eventually escape immune surveillance through various mechanisms promoting self-tolerance. 1 The discovery of immune checkpoints and the development of immune checkpoint inhibitors (ICIs), namely anti-programmed cell death protein 1 (PD-1)/programmed death-ligand 1 (PD-L1) and anti-cytotoxic T-lymphocyte-associated antigen 4 (CTLA4) monoclonal antibodies, has emerged as an effective strategy to restore antitumoral immunity by ‘removing the brakes’ off the immune system. Checkpoint inhibitors have revolutionized the management of metastatic microsatellite instability (MSI)/deficient mismatch repair (dMMR) tumors across tumor sites.2,3 MSI is detected in up to 20% of localized gastric cancer (GC) and colorectal cancer (CRC) and presents unique molecular and clinical characteristics calling into question the current standard of care for early-stage disease. The incorporation of ICIs in the armamentarium of cancer therapy in the neoadjuvant setting is an active area of research. In this review, we highlight the opportunities and challenges for novel neoadjuvant strategies, building on retrospective and emerging clinical data incorporating ICIs in the treatment of stage II–III dMMR GC and CRC.
Testing for MSI
Function of the MMR system can be assessed by immunohistochemistry (IHC) or molecular testing, including classic polymerase chain reaction (PCR) and novel next-generation sequencing (NGS) approaches. The four critical genes involved in this pathway (MLH1, MSH2, MSH6, and PMS2) encode proteins functioning as heterodimers.
Under normal circumstances, MSH2 forms a heterodimer with MSH3 or MSH6 to detect and bind single base mismatches or short insertions and deletions. MLH1 partners with PMS2, PMS1, or MLH3 to form a second heterodimer responsible for excision and resynthesis of the corrected DNA bases. Loss of function of any of these genes through mutation or epigenetic silencing results in a failure to repair mismatches and accumulation of mutations. This is reflected in the genome by MSI. Microsatellites are short tandem repeats distributed throughout the genome and identical in all cells from an individual. They are prone to mismatch errors, PCR can detect variability (instability) in the length of these repeated sequences arising as a consequence of dMMR. Alternatively, loss of function of genes involved in the MMR machinery can be easily identified by lack of protein expression on IHC. These two methods are equally valid as initial screening tests and have shown a high degree of concordance.4,5 IHC is generally recommended as an initial screening test due to its wide availability, lower cost, and rapid turnaround time. NGS is emerging as an alternative molecular method to assess MSI. Its main advantages are the possibility to identify the rare cases of dMMR caused by alternative mechanisms and the possibility to carry a complete biomarker analysis in one test. It has shown high concordance with PCR testing when carried both on tissue and plasma [circulating tumor DNA (ctDNA)] samples.6,7 However, ctDNA testing for MSI is insensitive if the plasma tumor fraction is below 0.2%. 7 Despite these promising results, the use of NGS for determination of MSI status requires further validation and should be carried only in centers experienced with these techniques, as recommended by the current European Society of Medical Oncology guidelines. 8 From now on, we will refer to the state of MMR deficiency or MSI-high using the term dMMR to avoid confusion with testing methods and to refer more generally to the phenotype. Mismatch repair status has major implications as a prognostic and predictive biomarker, and testing should be requested at diagnosis for all colorectal and gastric adenocarcinomas.
Mismatch repair deficiency in GC
GC with MSI is one of the four molecular subtypes described by either The Cancer Genome Atlas or the Asian Cancer Research Group.9,10 Its prevalence is stage dependent and has been reported in up to 20% of node-negative cases compared to less than 5% in metastatic disease. 10 Mismatch repair in GC is most often caused by acquired epigenetic silencing of the MLH1 promoter,11–13 thought to be an early event in gastric carcinogenesis.12,14,15 Methylation events accumulate during GC progression 16 and MLH1 promoter hypermethylation may also rarely occur as a late event in tumor evolution, 17 potentially leading to significant tumor heterogeneity in the dMMR phenotype. 18 In fact, discrepant MMR protein expression between the primary tumor and nodal metastases has been reported 19 and significant MLH1 expression heterogeneity has been reported in 8.9% of dMMR GCs. 20 Whether this tumoral heterogeneity impacts clinical outcomes remains unknown. MLH1 promoter hypermethylation is also the leading cause of MMR deficiency in familial cases and cannot be used to differentiate somatic versus germline GC, 13 unlike CRC. dMMR GCs are enriched with KRAS mutations, they generally lack targetable amplifications (HER2) and are not associated with BRAF V600E mutations.9,21,22
Prognostic and predictive role of dMMR in GC
dMMR status is associated with improved outcomes in resectable GC compared to microsatellite stable (MSS)/proficient mismatch repair (pMMR). This evidence comes from several post hoc analysis of randomized controlled trials 23 and one meta-analysis of 48 studies. 24 More recently, the role of neoadjuvant and adjuvant chemotherapy has been challenged by a few reports assessing the predictive value of MSI status on the efficacy of chemotherapy (Table 1). Post hoc analysis of the CLASSIC, 25 MAGIC, 26 and D1/D2-CRITICS 27 trials have been published. Choi et al. found no improvement in overall survival (OS) with adjuvant chemotherapy with capecitabine and oxaliplatin after R0 resection for the 40 dMMR patients included in the CLASSIC trial. In the MAGIC trial, none of the 12 dMMR patients randomized to perioperative chemotherapy with epirubicin, cisplatin, and fluorouracil showed a pathological response and the 10 dMMR patients treated with surgery alone had an excellent survival (median not reached compared to 20.5 months for MSS patients, hazard ratio (HR) 0.42, p = 0.09). Similarly, post hoc analysis of the D1/D2 and CRITICS trials confirmed the positive prognostic value of the dMMR status and found a poor pathological response [defined as tumor regression grade (TRG) 4, 5 or progression] in 24 of 27 dMMR patients after preoperative chemotherapy with epirubicin, cisplatin or oxaliplatin, and capecitabine (ECX or EOX). The three patients who had a response had substantial mucinous differentiation. Finally, Pietrantonio et al. conducted an individual patient data meta-analysis from four randomized trials (MAGIC, CLASSIC, ARTIST, 28 and ITACA-S 29 ) to further clarify the prognostic and predictive value of the dMMR subtype. 30 Patients with dMMR GC (n = 121) had superior disease-free survival (DFS) and OS at 5 years but did not appear to benefit from the addition of chemotherapy to surgery, while the pMMR subgroup had improved outcomes with the addition of chemotherapy. Five-year DFS for dMMR patients was 70% with chemotherapy and surgery versus 77% with surgery alone [HR: 1.27; 95% confidence interval (CI): 0.53–3.04] and 5-year OS was 75% versus 83% (HR 1.50; 95% CI: 0.55–4.12). Among these studies, only ITACA-S incorporated a taxane in the experimental arm. The addition of taxanes is now standard of care and appears to improve responses regardless of MSI status. Haag et al. reported a retrospective analysis of 101 patients undergoing perioperative chemotherapy, 58 had received fluorouracil, leucovorin, oxaliplatin, and docetaxel (FLOT) including five of nine dMMR patients. 31 The rate of poor responders (defined as TRG 2 and 3) did not differ significantly between pMMR and dMMR patients (79.1% versus 88.9%, p = 0.683). Preliminary results from the DANTE phase II trial showed a similar pathological response to FLOT chemotherapy in dMMR and pMMR tumors, with 47% of the 25 dMMR patients achieving a complete or subtotal regression (TRG1 a/b) compared to 44% of the 295 randomized patients. 32 Subanalysis of the phase III JACCRO-G7 trial 33 and FLOT4 trial 34 has not been published.
Outcomes of patients with dMMR stage II–III GC or GEJ cancer.

TRG 1a: pCR; TRG 1b: <10% vital tumor; TRG2: 10–50% vital tumor.
Central assessment.
pCR in all randomized patients (295 patients including 25 dMMR patients).
5-FU, 5-fluorouracil; CAPOX, capecitabine and oxaliplatin; CT, chemotherapy; CRT, chemoradiotherapy; DFS, disease-free survival; dMMR, deficient mismatch repair; ECF, epirubicin, cisplatin and fluorouracil; FLOT, fluorouracil, leucovorin, oxaliplatin, and docetaxel; GC, gastric cancer; GEJ, gastro-esophageal junction; HR, hazard ratio; MSS, microsatellite stable; NR, not reached; OS, overall survival; pCR, pathological complete response; pMMR, proficient mismatch repair; RFS, recurrence-free survival.
Taken together, these data suggests that dMMR tumor status is associated with decreased benefit from adjuvant or neoadjuvant chemotherapy, owing to both improved baseline prognosis and decreased sensitivity to chemotherapy compared to patients with pMMR GC. The current recommended standard of care for fit patients with locally advanced (cT2-T4 or N+) GC is either perioperative chemotherapy typically with FLOT or upfront surgery followed by adjuvant chemotherapy; this indication is irrespective of biomarker analysis.37,38 Knowledge of MMR status adds relevant prognostic information and may be used for individualized therapeutic decision-making. Patients with resected early-stage, node-negative dMMR GC appear to have an excellent prognosis with surgery alone and may potentially avoid chemotherapy after multidisciplinary discussion. While an absence of benefit from neoadjuvant chemotherapy cannot be definitively concluded in view of the limited numbers and retrospective nature of available data, considerable debate remains as to whether patients with stage II-III dMMR GC otherwise eligible for perioperative chemotherapy would be better served by proceeding directly to surgery. Further prospective data are needed to conclusively change the standard of care for stage II-III dMMR GC.
The use of checkpoint inhibitors in the neoadjuvant setting
Given the impressive efficacy of ICIs in metastatic dMMR GC 23 and previously mentioned caveats with neoadjuvant chemotherapy, it is natural to incorporate these agents at earlier stages. Several trials adding a PD-1/PD-L1 inhibitor to a perioperative chemotherapy backbone are currently open to accrual for patients with gastric or gastro-esophageal junction (GEJ) adenocarcinoma and will include a minority of patients with dMMR tumors. Among these, Al-Batran et al. reported a complete pathological response (TRG1a) rate of 63% (5/8) with the addition of atezolizumab to FLOT in the DANTE trial, compared to 27% (4/15) with FLOT alone. 32 A chemotherapy-free approach using dual ICIs was also demonstrated to be feasible and highly effective in the NEONIPIGA and INFINITY phase II trials.36,39 In total, 32 patients with cT2T4Nx dMMR gastric or GEJ tumors were treated with 12 weeks of neoadjuvant nivolumab and low-dose (1 mg) ipilimumab followed by surgery and an additional 9 months of adjuvant nivolumab in the NEONIPIGA trial. All but one patient underwent surgery, 58.6% achieved a pathological complete response (pCR). Treatment was well tolerated, the rates of surgical complications and adverse events were as expected with 25% grade 3–4 toxicity. At a median follow-up of 12 months, 30 (93.7%) patients remained alive without relapse. Similarly, 15 patients received a single dose of tremelimumab and 12 weeks (3 doses) of neoadjuvant durvalumab followed by surgery in cohort 1 of the INFINITY trial, pCR rate was 60%. Two patients had disease progression after a median follow-up of 13.4 months, one had heterogeneous pMMR/dMMR status.
Is this the dawn of a new surgery-free era?
There is a strong biological rationale to incorporate ICIs in the management of dMMR early-stage GC, preliminary results from ongoing trials are encouraging and truly have the potential to change current standard of care. Surgery has been the cornerstone of potentially curative treatment and the integration of systemic therapy has improved outcomes but has never been considered curative by itself. The unprecedented rates of pCR in the NEONIPIGA trial coupled with the durability of responses observed in the metastatic setting are now challenging the need for surgery for the subset of patients who achieve a complete clinical response, particularly when considering the significant morbidity of gastrectomy and long-term consequences in terms of quality of life. Many questions remain to be answered before this paradigm shift can be implemented in clinical practice. The main caveat for nonoperative management of GC is the suboptimal negative predictive value of preoperative staging and/or surveillance 40 – the incorporation of ctDNA to surveillance protocols may mitigate this to some extent. Longer follow-up will also be required to determine the durability of response, patterns of recurrence and feasibility of salvage gastrectomy. Cohort 2 of the phase II INFINITY trial (NCT04817826) will help answering these questions by investigating the use of dual checkpoint inhibitor blockade with durvalumab and tremelimumab for resectable dMMR gastric or GEJ adenocarcinomas. The protocol includes a non-operative management strategy for patients achieving clinical complete response (cCR) after 12 weeks of therapy. 41 Finally, the best strategy to achieve response and long-term DFS with or without surgery remains to be explored. Based on a limited number of patients and limited follow-up, response rates to induction chemotherapy with FLOT and anti-PD-1 for 8 weeks appear comparable to a 12-week anti-CTLA4 and anti-PD-1 regimen.32,36 In the metastatic setting, response rates (70 versus 55%) and OS (not reached versus 38.9 months) favored the combination of ipilimumab and nivolumab over chemotherapy with nivolumab for the 34 dMMR patients treated with ICIs in the phase III trial Checkmate-649. 42 Table 2 summarizes currently active clinical trials incorporating ICIs in the management of patients with resectable gastric or GEJ adenocarcinoma.
Active trials for patients with stage II–III gastric–GEJ adenocarcinoma.

cCR defined as the absence of residual disease on. Radiological examinations, tissue and liquid biopsies, in the absence of salvage gastrectomy.
p Stage III.
yp N+ or R1.
pCR and ctDNA negative.
ctDNA, circulating tumor DNA; DFS, disease-free survival; EFS, event-free survival; FLOT, fluorouracil, leucovorin, oxaliplatin, and docetaxel; OS, overall survival; pCR, pathological complete response; PD-1, programmed cell death protein 1; RFS, recurrence-free survival.
Mismatch repair deficiency in CRC
Mismatch repair deficiency in CRC arises by one of two distinct mechanisms. 48 Germline mutation of one of the genes involved in MMR (Lynch syndrome) accounts for approximately 25% of cases, while sporadic cases account for approximately 75% of cases and are attributed to a loss of expression of MLH1 caused by hypermethylation of the MLH1 promoter, associated with the CpG island methylator phenotype and BRAF V600E mutation. 49 The presence of either of these two molecular features essentially excludes Lynch syndrome. Right-sided tumors tend to be sporadic while rectal tumors tend to be associated with Lynch syndrome. dMMR is found in 15–20% of cases in stage II, 10–15% in stage III, and 4–5% in stage IV colon cancer,50,51 the prevalence is lower in rectal cancer across all stages (1–10%).52,53 dMMR is an early event in colorectal carcinogenesis even in sporadic cases and although inter-tumoral heterogeneity of MMR status has been a subject of debate, discordance between primary tumor and metastases seems extremely rare. 54 dMMR CRC is enriched with mutations in RAS, BRAF, HER2, and NTRK fusions; all are considered mutually exclusive.55,56
Prognostic and predictive value of dMMR for adjuvant therapy in CRC
A number of retrospective studies and post hoc analysis have evaluated the role of MMR as a prognostic and predictive biomarker in stage II–III CRC, 57 a complete review would be beyond the scope of this article. The largest evidence comes from two separate pooled individual patient data analysis of the Adjuvant Colon Cancer Endpoint (ACCENT) database,50,51 results are summarized in Table 3. dMMR confers an improved prognosis at earlier stage, such as N0 (stage II, HR: 0.27, 95% CI: 0.10–0.74, p = 0.011) or N1 disease [1–3 involved nodes, adjusted HR (aHR): 0.65, 95% CI: 0.45–0.93, p = 0.0132], but not for N2 disease (>3 involved nodes, aHR: 1.22, 95% CI: 0.94–1.59, p = 0.1403). The presence of the BRAF V600E mutation does not appear to confer an adverse prognosis for dMMR patients with stage II–III colon cancer,51,58,59 although it may mitigate the positive prognosis associated with dMMR 60 in stage III disease. The association between dMMR status and lack of efficacy of 5-fluorouracil (5-FU)-based adjuvant chemotherapy in colon cancer has been a matter of debate for the past decade. In an analysis by Sargent et al. 61 in 2010, 102 patients with dMMR stage II colon cancer had worse survival when treated with adjuvant 5-FU-based adjuvant chemotherapy compared to surgery alone (HR: 2.95, 95% CI: 1.02–8.54, p = 0.04). Subsequent pooled analysis and meta-analyses have confirmed the more favorable prognosis of stage II dMMR colon cancer and lack of benefit from adjuvant chemotherapy; however, no detrimental effect was observed.50,57 Current guidelines do not support routine adjuvant chemotherapy for patients with stage II dMMR colon cancer while oxaliplatin-based chemotherapy may be considered for high-risk stage II disease. Three to six months of oxaliplatin-based chemotherapy per usual standards is recommended for patients with dMMR stage III colon cancer.62,63 The addition of oxaliplatin significantly improved OS compared to FP alone (HR: 0.52, 95% CI: 0.28–0.93) in the most recent ACCENT database analysis. 51 Despite optimal management, it is estimated that up to 30% of patients with dMMR colon cancer will experience recurrence. The phase III POLEM 64 and ATOMIC 65 trials are investigating the addition of an anti-PD-L1 inhibitor to a chemotherapy backbone as adjuvant therapy for stage III dMMR colon cancer.
Predictive and prognostic value of MSI from two ACCENT pooled analysis.
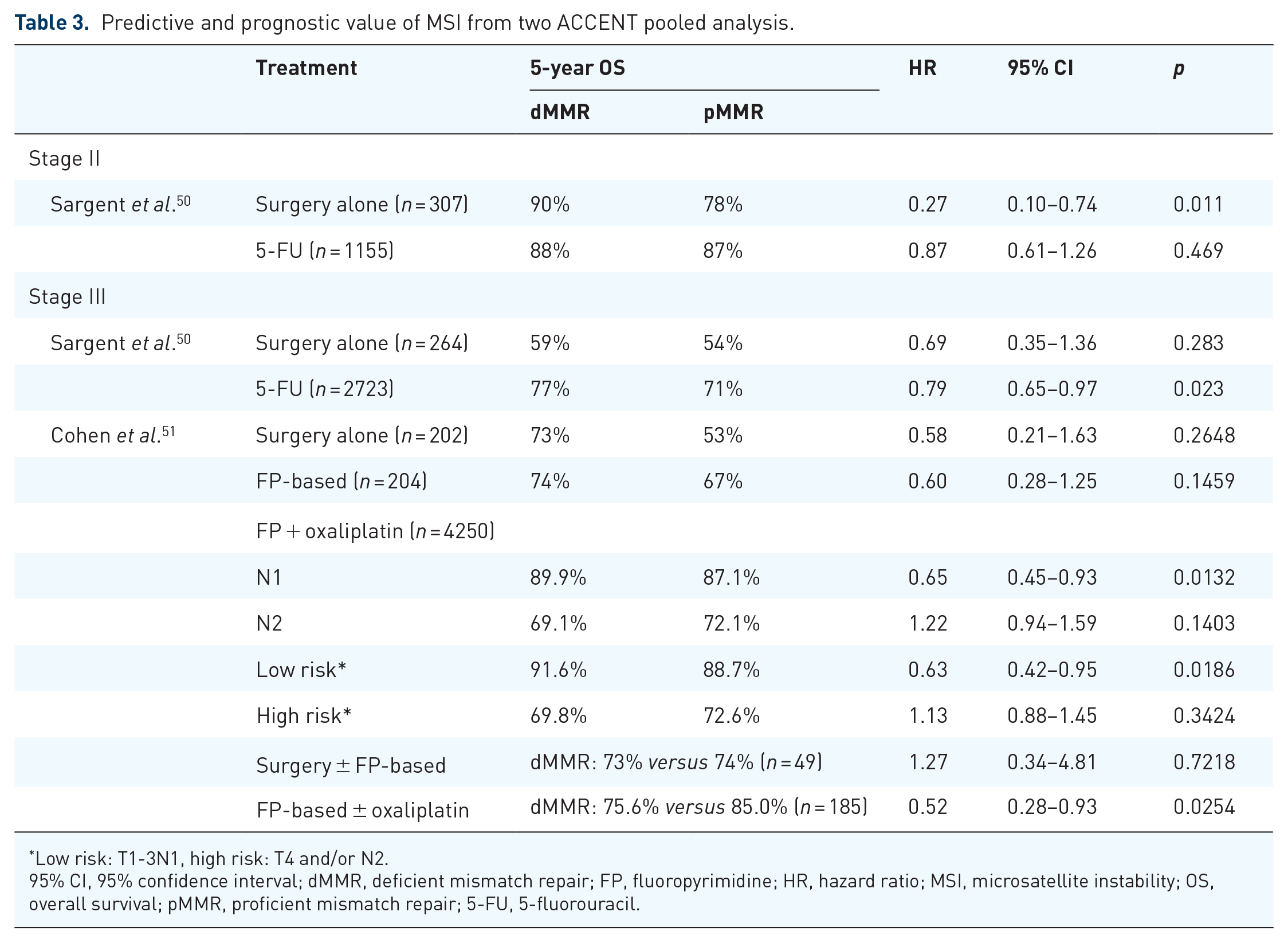
Low risk: T1-3N1, high risk: T4 and/or N2.
95% CI, 95% confidence interval; dMMR, deficient mismatch repair; FP, fluoropyrimidine; HR, hazard ratio; MSI, microsatellite instability; OS, overall survival; pMMR, proficient mismatch repair; 5-FU, 5-fluorouracil.
Evolving treatment landscape in the neoadjuvant setting for CRC
Colon cancer and rectal cancer are distinct entities when it comes to neoadjuvant therapy due to obvious anatomical considerations. Until recently, there has been little interest in neoadjuvant therapy for colon cancer because of the low rates of local recurrence and lower rates of surgical morbidity compared to rectal cancer. Nevertheless, surgery is still associated with complications and quality-of-life deterioration, 66 especially in elderly or frail patients. The role of neoadjuvant chemotherapy with 6 weeks of FOLFOX (fluorouracil, leucovorin, and oxaliplatin) or CAPOX (capecitabine and oxaliplatin) administered preoperatively for locally advanced colon cancer was investigated in the FOXTROT phase III trial, 67 a total of 6 months of chemotherapy was planned for both arms. Results showed halving of the rate of incomplete resection (5% versus 10%) and decreased 2-year recurrence rate (16.8% versus 21.2%, HR: 0.72, 95% CI: 0.54–0.98, p = 0.037) on updated analysis. Concerns about limitations with clinical staging and the potential for overtreatment with oxaliplatin, especially in frail or elderly patients, have limited clinical applicability and further investigation is ongoing. 68
Rectal cancer is typically managed with multimodality therapy including radiation, surgery and chemotherapy. Research over the past few years has focused on decreasing long-term treatment morbidity without compromising efficacy. Recommendations for systemic chemotherapy in rectal cancer were traditionally extrapolated from colon cancer with the goal of increasing long-term survival, systemic therapy is now also regarded as an additional tool to maximize downstaging for consideration of organ-sparing nonoperative management. Radiation and resection of the rectum are associated with significant long-term complications and disabilities69–71 that could potentially be avoided for a subset of patients with earlier use of effective systemic therapy.72,73 cCR in rectal cancer is increasingly recognized as a surrogate marker for pCR and DFS74–77 regardless of the strategy used to obtain clinical response. This is highly relevant when considering neoadjuvant strategies for dMMR CRC cancer given their unique response (or lack of response) to radiation, chemotherapy, and immunotherapy. Neoadjuvant therapy in CRC has two main goals: to maximize tumor downstaging to facilitate or avoid resection and to minimize the risks of locoregional and distant recurrence.
Patients with dMMR CRC appear to have decreased sensitivity to chemotherapy but not to chemoradiation. A systematic review and meta-analysis of nine studies including 5877 patients showed no association between dMMR status and pCR after chemoradiation (10.1% versus 6.6% for dMMR versus pMMR, odds ratio: 1.38, 95% CI: 0.7–2.72). 78 In a retrospective analysis of 50 patients with dMMR rectal cancer treated with neoadjuvant therapy, 93% of patients (13/16) experienced tumor downstaging after chemoradiation but 29% (6/21) had disease progression while receiving neoadjuvant chemotherapy with fluorouracil and oxaliplatin. 53 Similarly, response to 6-week neoadjuvant FOLFOX or CAPOX in the FOXTROT trial was poor with only 7% (8/115) of dMMR patients achieving moderate or greater histological tumor regression. The data suggest incorporating chemotherapy as part of a total neoadjuvant approach may not provide significant downstaging over radiation or chemoradiation alone, and patients should be followed closely given the risk of progressive disease. It is worth emphasizing that patients with stage III dMMR CRC still qualify for oxaliplatin-based chemotherapy per usual recommendations based on improved OS, as discussed previously.
If chemotherapy is not the optimal path to optimize local control and treatment de-escalation, ICIs are showing promising results for patients with dMMR CRC. In the NICHE phase II study, 79 32 patients with dMMR colon cancer (81% stage III, 67% sporadic) were treated with a single dose of the anti-CTLA4 ipilimumab and two doses of the anti-PD-1 nivolumab followed by surgery. The observed response rate was 100% with 69% (26/32) achieving pCR and 97% (31/32) achieving major pathological response (<10% viable tumor cells). Poor correlation was noted between radiological assessment and pathological response. All patients are alive and disease-free after a median follow-up of 25 months. In the PICC phase II trial, 80 34 patients with locally advanced dMMR colon cancer were randomized to toripalimab with or without celecoxib for 12 weeks followed by surgery and an additional 6 months of adjuvant therapy with the same regimen. pCR rate was 76.5% (26/34), 88% with toripalimab and celecoxib and 65% without celecoxib. All patients are alive and disease-free at a median follow-up of 14.9 months. Sequential use of five cycles of nivolumab after chemoradiation was investigated in the phase II VOLTAGE-A trial 81 for 37 patients with pMMR and 5 patients with dMMR locally advanced rectal cancer (LARC). The addition of nivolumab was safe, two patients had grade 3 immune-related toxicities, one myasthenia, and one interstitial nephritis. Two additional patients had grade 2 toxicity, one colitis, and one peripheral neuropathy. Three of the five patients (60%) with dMMR LARC achieved a pCR. Most recently and to much fanfare, the results from a single-center phase II study investigating the anti-PD-1 agent dostarlimab for 6 months for locally advanced dMMR rectal cancer were recently published in the New England Journal of Medicine. 82 Patients achieving a cCR (determined both radiographically and endoscopically) were to undergo a watch-and-wait strategy, while those with residual disease were to be treated with standard chemoradiation and surgery or nonsurgical management according to clinical response. At the time of publication, 12 patients have completed treatment with a minimal follow-up of 6 months, with a remarkable complete clinical response rate of 100% and no patient underwent chemoradiation or surgery. Responses were observed early with 81% of patients showing symptomatic improvement by week 9, and most patients achieving at least near complete response at the first assessment at 3 months.
It is not often in the field of medical oncology that we see response rates of 100%. While the follow-up is still limited, there is good reason to believe that these responses will be durable, extrapolating from data in the metastatic setting.83,84 For the first time, systemic therapy offers the potential for cure without the need for resection or radiation. The safety profile of ICI therapy compares favorably with the morbidity entailed by radiation or surgical resection. There are no new safety signals from the few trials previously mentioned, and the rate of severe or permanent toxicity from ICIs can be expected to be similar to what is observed in the metastatic setting or across tumor sites. Fatal toxicity rate from ICIs is 0.4–1.2%, the rate of chronic immune-related adverse events (irAEs) may be as high as 42.9%, mostly mild endocrinopathies (hypothyroidism) and rheumatological toxicities. 85 Chronic irAEs are still poorly defined and are the focus of ongoing research, severe permanent disabilities are fortunately rare (<5%). Although very promising, caution is still advised before ICIs become a standard of care in early-stage dMMR CRC: the conclusions are limited by the very small number of patients from a single center, and follow-up is still short. That said, clinicians are now facing a difficult dilemma where the usual standard of care designed for pMMR tumors is suboptimal but the emerging ICI alternative challenges 30 years of evidence-based medicine with available results based on a limited number of patients with minimal follow-up. In the metastatic setting, 29.4% of patients receiving first-line pembrolizumab had disease progression as best response in the landmark Keynote-177 trial. 84 Some of these events may be accounted for by radiological pseudoprogression 86 ; however, there is still a crossing of the survival curves within the first 6 months. No patient experienced primary resistance in the neoadjuvant setting but the small number of patients enrolled cannot provide a reliable estimate. How much evidence is required to safely change practice? At the very least, longer follow-up is needed and results will have to be reproduced in larger multi-center prospective cohorts. Even if randomized clinical trials may not be feasible or required to change standard of care in this case, further evidence is needed to determine the best treatment regimen, optimal duration of treatment, and to provide more information to weigh the risks and benefits of each approach.
Conclusion
In summary, dMMR GC and CRC are distinct entities with unique clinical characteristics and vulnerability to ICIs. Current standards of care were designed to optimize outcomes for pMMR cancers and are imperfectly suited for dMMR cancers. MMR ascertainment is recommended for all early-stage CRC and GC. ICIs demonstrate remarkable activity in the neoadjuvant setting with unprecedented rates of pCR, questioning the need for well-established multimodality approaches including surgery. Although it remains to be seen whether these current exciting results will translate into long-term benefits, neoadjuvant immunotherapy is feasible and offers tremendous promise for patients with dMMR cancers. These data are still considered preliminary – longer follow-up is needed and optimal duration and combination strategies remain to be determined. Hence, caution is advised before de-escalating standard of care treatments with proven survival benefit. Where available, enrollment to ongoing clinical trials is encouraged.
