Abstract
Background
It is important to identify novel plasmatic biomarkers that can contribute to assessing the prognosis and outcome of breast cancer patients. Neuregulin-1 (NRG1) and galectin-3 (Gal-3) are proteins that are involved in breast cancer development and patient survival; therefore, we studied whether the serum concentration of these 2 proteins can be correlated to breast cancer progression.
Methods
Plasmatic NRG1 and Gal-3 were evaluated in 25 healthy controls and 50 breast cancer patients at baseline and at 3 and 6 months after treatment with anthracyclines and taxanes, with or without trastuzumab.
Results
NRG1 and Gal-3 were significantly more elevated in cancer patients than in healthy controls; furthermore, NRG1 and Gal-3 were significantly increased after chemotherapy and were predictive of mortality at 1 year.
Conclusions
Circulating NRG1 and Gal-3 can be additional biomarkers indicative of prognosis and outcomes for breast cancer patients.
Introduction
Neuregulins (NRGs) are proteins of the epidermal growth factor (EGF) family that bind human epidermal growth factor receptor 2 (HER2), HER3 and HER4. These growth factors play an important role in the development and progression of many types of human cancer, in particular of breast tumors, acting as mitogenic factors (1, 2). NRGs are produced by microvascular endothelial cells and can influence the growth rate of malignant cells through paracrine stimulation (3).
Galectin 3 (Gal-3) is a member of the β-galactoside-binding lectin family and is a pleiotropic protein involved in many biological processes such as cell adhesion, migration, growth and apoptosis (4). Gal-3 plays an important role in neoplastic progression and is highly expressed by triple-negative breast cancer, for which it has been proposed as a potential therapeutic target (5).
In recent years, studies have been undertaken to understand the clinical significance of several circulating molecules in cancer patients, to evaluate whether they relate to each other and whether they can be correlated with prognosis and outcomes. For this purpose, our group recently demonstrated that plasmatic N-terminal pro-brain natriuretic peptide is capable of identifying breast cancer patients undergoing chemotherapy at risk of cardiotoxicity, and that the same biomarker is capable of giving a prognosis regarding patient outcomes (6). In the present study, we prospectively measured circulating NRG1 and Gal-3 in breast cancer patients before and after chemotherapy, to examine whether these biomarkers can be correlated with patient survival and consequently be used as additional indicators of patient outcomes.
Patients and Methods
We performed a prospective study of 50 breast cancer patients, 25 of whom were HER2-negative and 25 HER2-positive, undergoing adjuvant chemotherapy, and of 25 healthy controls. Patients were enrolled on the basis of the following criteria: (a) they were undergoing treatment with anthracyclines, taxanes and trastuzumab; (b) had filled in a questionnaire for the evaluation of the functional validity of the Eastern Cooperative Oncology Group (ECOG) performance status (PS), with a score between 0 and 2; (c) had normal indices of liver (bilirubin <1.5 mg/dL) and renal function (creatinine <1.5 mg/dL).
The chemotherapy was based on HER2 expression: HER2-negative patients underwent treatment with epirubicin (90 mg/m2 every 21 days) and cyclophosphamide (600 mg/m2 every 21 days × 4 cycles) (EC) + paclitaxel (80 mg/m2 every 7 days × 12 cycles); HER2-positive patients underwent EC + paclitaxel + trastuzumab (8 mg/kg loading dose, followed by 6 mg/kg every 21 days) followed by trastuzumab alone (1 year total).
Plasma concentrations of NRG1, Gal-3, N-terminal pro-brain natriuretic peptide (NT-ProBNP), I troponin (Tn I) and creatine kinase-myocardial and brain (CK-MB) were evaluated before chemotherapy (TO), and NRG1 and Gal-3 also at 3 months (T1) and 6 months (T2) after treatment in cancer patients. In parallel, plasma from healthy controls was evaluated for NRG1 and Gal-3. Venous blood (5 mL) was drawn into a lithium heparin tube, and within 30 minutes, the sample was centrifuged at 3000 rpm for 10 minutes to obtain the plasma. The supernatant was then transferred to an Eppendorf tube and stored at −80°C until measurements were performed. The following kits were utilized to evaluate Gal-3: VIDAS (bioMérieux); NRG1: Abcam® Human ELISA Kit ab100614; Tn-I: Roche Elecsys kit; CK-MB: Roche Elecsys kit; NT-ProBNP: Roche Diagnostic GmbH kit (Roche, Mannheim, Germany).
All patients provided informed consent, and the study was approved by the institutional ethics committee.
Statistical analysis
Statistical analysis was performed using R version 3.1.3 (2015-03-09; Smooth Sidewalk; R Foundation for Statistical Computing). All data were first analyzed for normality of distribution using the D'Agostino-Pearson test of normality. Continuous variables were expressed as means ± standard deviation (SD), unless otherwise specified, and the appropriate parametric test (t-test) was used to assess the significance of any differences between the 2 groups. Categorical variables were displayed as frequencies and compared using the Fischer test. For time-course comparisons, 1-way ANOVA was used. The receiver operating characteristic (ROC) curve was applied to evaluate the ability of the biomarkers to predict death at 1 year (6). Multiple regression analysis with stepwise variable selection was applied to assess independent correlations with death at 1 year. All statistical tests were 2-tailed, and a p value <0.05 was considered statistically significant.
Results
Our cohort was composed of 50 women with breast cancer (mean age 54 years, SD = 7.3) and 25 healthy controls (mean age 50 years, SD = 8.7); breast cancer patients were selected on the basis of HER2 expression: 25 negative and 25 positive.
Plasmatic NRG1 and Gal-3 were initially evaluated at baseline before chemotherapy (TO) and both were significantly higher in cancer patients than in healthy controls: NRG1 had a mean value of 1.95 ng/mL in cancer patients versus 0.52 ng/mL in controls (p<0.0001), Gal-3 had a mean value of 18.41 ng/mL in cancer patients and of 6.73 ng/mL in controls (p<0.0001) (Tab. I). A time course analysis of plasmatic NGR1 and Gal-3 was performed in cancer patients at 3 (T1) and 6 (T2) months after chemotherapy and showed that both NGR1 and Gal-3 were significantly (p<0.0001) increased at T1 and T2 in both groups of patients: HER2+ and HER2- (Tab. II). However, the values for either NGR1 or Gal-3 at T1 and T2 were similar, suggesting that the 2 molecules did not increase significantly at 6 months from treatment, as compared with the values for 3 months.
Comparison between cancer patients and healthy controls for neuregulin-1 (NRG1) and galectin-3 (Gal-3) levels at baseline (T0)
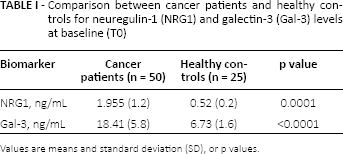
Values are means and standard deviation (SD), or p values.
Time course in cancer patients and HER2 groups
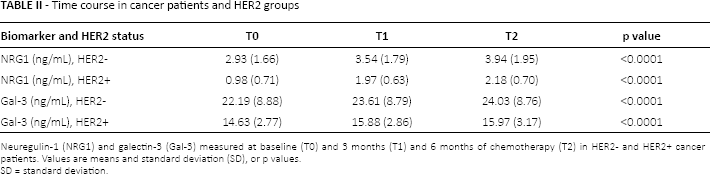
Neuregulin-1 (NRG1) and galectin-3 (Gal-3) measured at baseline (T0) and 3 months (T1) and 6 months of chemotherapy (T2) in HER2- and HER2+ cancer patients. Values are means and standard deviation (SD), or p values.
SD = standard deviation.
Comparing the HER2+ and HER2- groups, both NRG1 and Gal-3 were significantly (p<0.0001) higher in HER2- patients at every time of detection: TO, T1 and T2 (Tab. II).
To evaluate whether Gal-3 and NGR1 could be correlated with patient survival, their baseline plasmatic values were analyzed in a ROC curve for prediction of mortality at 1 year, together with the plasmatic values of NT-ProBNP, Tn-I and CK-MB, which we and other authors had previously demonstrated to be biomarkers of toxicity in breast cancer and, exclusively for NT-ProBNP, also a marker of prognosis (6, 7). We found that Gal-3 and NRG1, and also NT-ProBNP, but not Tn-I or CK-MB, were significantly correlated with the prediction of death at 1 year (Tab. III). Finally, the same biomarkers were evaluated in a multiple regression analysis for mortality prediction at 1 year and showed that baseline plasmatic Gal-3, body mass index (BMI) and HER2 expression were independent factors with respect to mortality, while CK-MB, NRG1, NT-ProBNP and Tn I were not independent factors with respect to mortality (Tab. IV).
Receiver operating characteristic curve analysis for mortality at 1 year
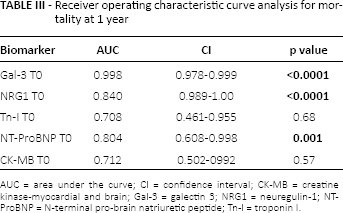
AUC = area under the curve; CI = confidence interval; CK-MB = creatine kinase-myocardial and brain; Gal-3 = galectin 3; NRG1 = neuregulin-1; NT-ProBNP = N-terminal pro-brain natriuretic peptide; Tn-I = troponin I.
Multiple regression analysis for mortality prediction at 1 year
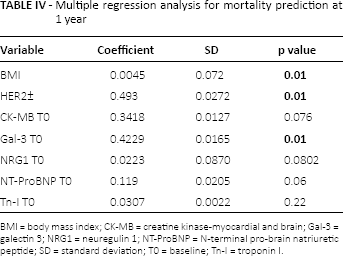
BMI = body mass index; CK-MB = creatine kinase-myocardial and brain; Gal-3 = galectin 3; NRG1 = neuregulin 1; NT-ProBNP = N-terminal pro-brain natriuretic peptide; SD = standard deviation; TO = baseline; Tn-I = troponin I.
Discussion
At present, therapy for breast cancer has reached a high level of quality, but it is necessary to be able to easily differentiate patients who need to be more aggressively treated, and this objective will be achievable whenever a more precise characterization of each single tumor in terms of prognosis and progression becomes feasible. So it is important that the identification of novel plasmatic molecules be added to that of established markers for the prediction of response to therapy and progression or relapse of breast cancer patients (8, 9). For this purpose, we recently described the fact that plasmatic NT-ProBNP can be used as a biomarker for early diagnosis of chemotherapy-related cardiotoxicity in breast cancer, and that this marker can also be predictive of patient outcomes (6).
In the present work, we found that baseline plasmatic values of NRG1 and Gal-3 were higher in breast cancer patients than in healthy controls and that these values were higher in HER- than HER2+ tumors, suggesting that the malignancy itself causes a dysregulation of these 2 circulating proteins. Furthermore, the data described are suggestive of the value of these 2 markers as prognostic indicators, because HER- patients have a worst outcome compared with HER2+ patients.
Further, we demonstrated that Gal-3 and NRG1 increased after 3 and 6 months of chemotherapy and that both markers were significantly correlated with the prediction of death at 1 year in our cohort of breast cancer patients. These findings are in accordance with a recent study that described increased Gal-3 at least 2 years after anthracycline treatment in pediatric patients (10) and with another study that showed an important correlation between plasmatic Gal-3 enhancement and toxicity in a cohort of 59 breast cancer survivors 1 year after anthracycline treatment (11). Furthermore, Gal-3 has been described as a promoter of metastasis in colon (12) and breast cancer (13).
Regarding neuregulins, their increased expression has been detected in several neoplasias, and their presence has been implicated in the development of resistance to treatments that target HER receptors (14). The data that we report for NGR1 are in accordance with a recent study where this plasmatic molecule was proposed as a potential prognostic marker of chemotherapy-induced cardiotoxicity (15). Our results are also in line with a recent study in which a monoclonal antibody that blocks NGR1-induced ErbB4 signaling was able to inhibit proliferation and tissue remodeling of breast cancer (16).
Despite the small size of our cohort and the requirement to validate the obtained results in a larger number of patients, we can speculate on the potential role of NRG1 and Gal-3 as additional parameters, which can be included in the larger panel of serum biomarkers utilized to better assess the prognosis and outcomes of breast cancer patients. The utilization of these novel markers is directed toward improving the management of breast cancer patients through the identification of appropriate and specific tools capable of more accurately defining the outcomes of the neoplastic disease. We are now tracking our cohort of patients over the years, measuring plasmatic NRG1 and Gal-3 during their follow-up, to confirm and clarify the value of these parameters as prognostic markers in breast cancer.
Footnotes
Financial support: None received.
Conflict of interest: The authors declare that they have no conflicts of interest.
