Abstract
BACKGROUND:
The new systemic inflammation response index (SIRI) constructed based on neutrophil, monocyte and lymphocyte counts in peripheral blood is considered to be related to the prognosis of a variety of tumours.
OBJECTIVE:
To evaluate the prognostic value of the SIRI in operable breast cancer patients and establish a nomogram to predict the survival of breast cancer patients.
METHODS:
A total of 949 patients with operable breast cancer were enrolled in the present study.
RESULTS:
The overall survival (OS) of breast cancer patients with SIRI
CONCLUSIONS:
The SIRI before and after surgery is closely related to the prognosis of breast cancer patients.
Keywords
Introduction
In the last 3 decades, breast cancer has gradually become one of the major causes of cancer in women, and its incidence rate has been increasing in different countries and regions in the world [1, 2]. Although the incidence rate of breast cancer in China is not as high as that in Europe and the United States, it has increased rapidly [3]. Breast cancer ranks first in morbidity rate among all female malignancies in large cities in China [3]. The survival rate of breast cancer patients has been greatly improved with comprehensive multidisciplinary treatment, but it remains a major cause of cancer-related death in women, second only to lung cancer [1]. Therefore, searching for predictive factors for the survival of breast cancer is critical to maintaining female health.
Currently, TNM stage, receptors (oestrogen receptor, progesterone receptor and human epidermal growth factor receptor 2), Ki-67 and tumour markers are recognized as predictive indexes for the prognosis of breast cancer patients. In recent years, it has been found that the prognosis of cancer patients depends on not only the characteristics of the tumour but also the patient’s response. Since Virchow discovered a large number of infiltrating lymphocytes around tumour tissues for the first time 150 years ago [4], a large number of studies have confirmed that inflammation plays an extremely important role at different stages of tumour occurrence and development [5, 6, 7]. The systemic inflammatory response in patients is associated with the poor prognosis of a variety of cancers [8, 9]. The systemic inflammation response index (SIRI), neutrophil to lymphocyte ratio (NLR), platelet to lymphocyte ratio (PLR) and monocyte lymphocyte ratio (MLR) have been shown to be closely related to the prognosis of breast cancer [10, 11, 12, 13, 14]. Recently, a SIRI constructed based on neutrophil, monocyte and lymphocyte counts in peripheral blood was considered to be able to predict the prognosis of pancreatic cancer [15], oesophageal cancer [9], adenocarcinoma of the oesophagogastric junction [16] and nasopharyngeal carcinoma [17]. Chen et al. and Hua et al. have reported that the pretreatment SIRI may be a reliable predictor of OS in breast cancer in recent years [18, 19]. In our study, the prognostic value of the SIRI before and after surgery in operable breast cancer patients was explored, a nomogram was constructed for such patients, and the accuracies of this model and the TNM stage in predicting prognosis were compared to guide clinical practice and develop individualized therapeutic regimens.
Material and methods
Patients
This study was approved by the Ethics Committee of the First Affiliated Hospital of Soochow University, the guidelines were prepared based on the Helsinki Declaration, and an informed consent form was signed by each patient. A retrospective analysis was performed for 949 patients with operable breast cancer meeting the inclusion criteria and treated in the above hospital between January 2009 and December 2012. All patients had complete clinical case data, pathological data, and routine blood test results before and 4 weeks after surgery. All patients were followed up by telephone or outpatient visits. The follow-up was performed once every 3 months within 2 years after the operation and then once every 6 months until October 2019. The median follow-up time was 102 months. The inclusion criteria were as follows: 1) patients with operable breast cancer newly treated in the First Affiliated Hospital of Soochow University, without undergoing chemotherapy, radiotherapy or endocrine therapy before specimen collection; 2) those pathologically diagnosed with breast cancer; 3) those with normal haematological, liver and kidney function and no serious immune system disease; and 4) those without distant metastasis. The exclusion criteria were as follows: 1) patients with incomplete follow-up data; 2) patients complicated with blood system diseases and other factors affecting routine results; and 3) patients with incomplete routine blood results within 1 week before treatment.
Clinical characteristics of the breast cancer patients
Clinical characteristics of the breast cancer patients
AJCC: American Joint Committee on Cancer.
Baseline characteristics for patients with SIRI
AJCC: American Joint Committee on Cancer; SIRI: Systemic Inflammation Response Index.
Blood was collected from patients within 3 days before the operation and 4 weeks after the operation. The peripheral blood results were recorded with a routine blood analysis recorder. According to previous research [20], the sensitivity and specificity were estimated using the Youden index to obtain the optimal cut-off values as follows: SIRI (SIRI
Statistical analysis
Analysis of variance and the Pearson chi-square test were used to assess correlations between variables. The Kaplan-Meier method was used to compare the OS in different groups, and the log-rank test was used to estimate the difference in survival rates. Multivariate Cox regression analysis was performed to determine whether the SIRI is an independent prognostic indicator of OS in patients with breast cancer via backward stepwise selection. A nomogram was constructed based on the results of multivariate analysis. According to the Akaike information criterion (AIC), the final model was subjected to backward stepwise selection, and the discriminative ability of the model was evaluated by calculating the consistency index (C-index). Bootstrap sampling (1000 times) was adopted for verification of the nomogram and calibration evaluation, and ROC curves were used to compare the sensitivity and specificity of the survival predictions. In addition, property score matching (PSM) analysis was required due to the imbalance of baseline characteristics. The variables age, grade, ER, PR, Her2, Breast Subtype, T stage, N stage and AJCC stage were used for matching. PSM was
Relationship between NLR, PLR or MLR and clinicopathological characteristics of breast cancer patients
Relationship between NLR, PLR or MLR and clinicopathological characteristics of breast cancer patients
AJCC: American Joint Committee on Cancer; SIRI: Systemic Inflammation Response Index; NLR: neutrophil lymphocyte ratio; PLR: platelet lymphocyte ratio; MLR: monocyte lymphocyte ratio.
Univariate and multivariate cox regression analyses for overall survival in breast cancer patients
AJCC: American Joint Committee on Cancer; SIRI: Systemic Inflammation Response Index; PLR: platelet lymphocyte ratio; NLR: neutrophil lymphocyte ratio; MLR: monocyte lymphocyte ratio; HR: hazard ratio; CI: confidence interval; Ref: reference.
performed using the nearest neighbour matching algorithm, and the maximum tolerance difference between propensity scores was less than 30% of the propensity score standardized difference. The matched standardized difference of all variables is less than 0.2, indicating that it meets the matching requirements. All statistical calculations were performed using SPSS 23.0 and R language 3.6.2 software. Unless otherwise specified,
Clinical characteristics of the patients
A total of 949 patients were included in this study, and they were all women with a ranging from 24–89 (median 54). The 5-year and 10-year survival rates of the patients were 86.9% and 73.2%, respectively. The clinicopathological characteristics of the patients are shown in Table 1.
Kaplan-Meier survival curves for patients stratified based on (A) SIRI, (B) NLR, (C) PLR and (D) MLR in operable breast cancer patients. Predictive ability of the SIRI in operable breast cancer patients was compared with PLR, NLR and MLR by ROC curves in 5-years (E) and 10-years (F).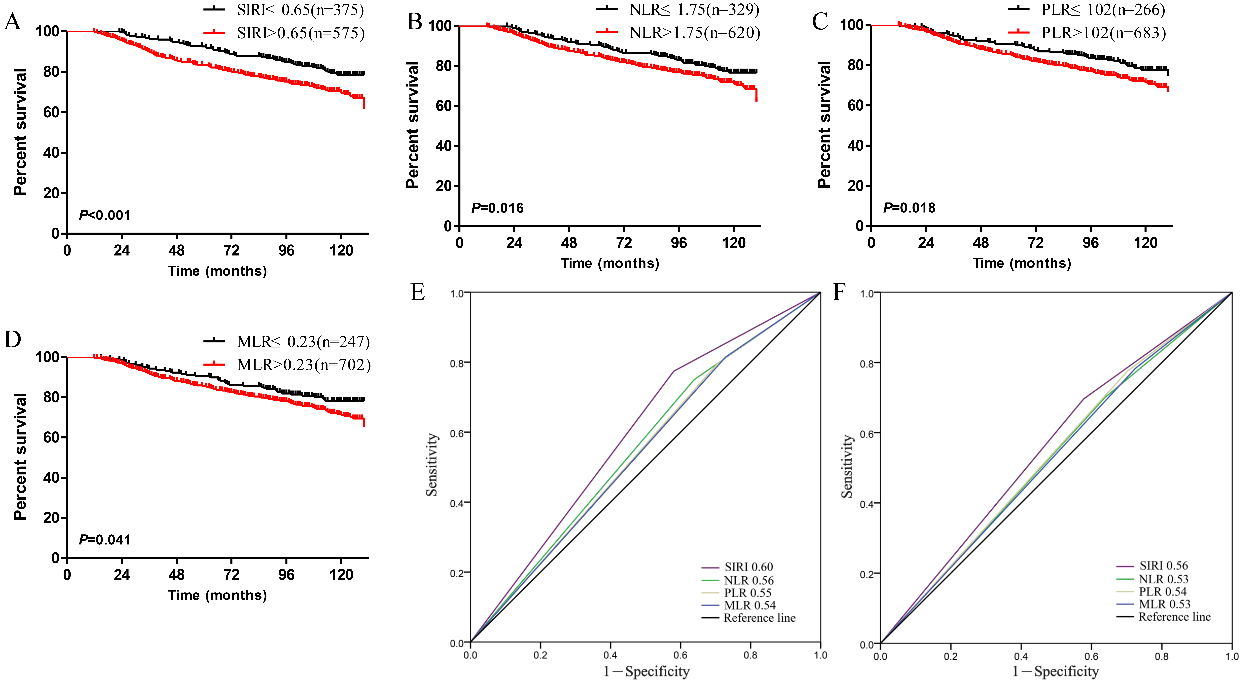
Kaplan-Meier survival curves for patients stratified based on SIRI after propensity matching.
The associations between the SIRI, NLR, PLR and MLR and the clinicopathological characteristics of breast cancer patients are shown in Tables 2 and 3. The SIRI was significantly correlated with the grade, N stage and TNM stage of breast cancer (
Evaluation of the nomogram integrating SIRI in operable breast cancer. (The risk factors involved in nomogram comprises grade, TNM staging, and SIRI. Draw a line up each variable axis, based on the value of multiple variables of a single patient, to determine the corresponding score for each variable value. The sum of these scores are matched to the total score axis, and then draw a line down to the survival axis, so as to determine the 5-year or 10-year survival rate).
(A) The 5-year survival rate of breast cancer patients predicted by nomogram is highly consistent with the actual observed values. (B) The 10-year survival rate of breast cancer patients predicted by nomogram is highly consistent with the actual observed values. (C) The ability of ROC analysis nomogram to predict the 5-year survival rate of the breast cancer patients, the nomogram has a larger AUC than TNM staging. (D) The ability of ROC analysis nomogram to predict the 10-year survival rate of the breast cancer patients, the nomogram has a larger AUC than TNM staging.
(A) Different changes of SIRI before and after the breast cancer radical operation have significant effects on survival. (B) The OS of SIRI decreasing by more than 75% (HR 
The AIC values were used to find the optimal model via backward stepwise selection. The independent prognostic markers identified (grade, TNM stage and SIRI) were included in the nomogram to predict the 5- and 10-year survival of breast cancer patients (Fig. 3). In order to make the nomogram easier to understand, a Supplementary Fig. 1 has been added to explain how to use the nomogram by giving an example of breast cancer case. The C-index of the nomogram was significantly higher than that of the TNM stage (
Prognostic value of dynamic changes in SIRI after surgery
Breast cancer patients, including baseline and 4-week postoperative SIRI data, were enrolled in the analysis (
Discussion
Rudolf Virchow, a German pathologist, first discovered massive white blood cell infiltration around tumour tissues 150 years ago, suggesting a correlation between inflammation and tumours [4]. Inflammatory cells are attracted by chemokines secreted by tumor cells to migrate around the tumour cells, and then secrete certain cytokines, inducing the normal cells near tumour cells to develop malignant phenotypes. These cytokines can accelerate the cell cycle, which is beneficial to tumour survival and angiogenesis. Moreover, the inflammatory microenvironment also promotes tumour growth, progression and metastasis in other ways, such as disrupting the immune response, inhibiting apoptosis, and changing the response of tumour cells to chemotherapy drugs [6]. According to multiple studies, inflammatory factors can effectively predict the prognosis of tumour patients [8, 9, 21, 22]. Recent studies have shown that a combination of cell ratios, including NLR, PLR, and MLR, has a better ability to predict the prognosis of tumour patients than single cells [11]. NLR, PLR and MLR have been confirmed to be closely related to the prognosis of breast cancer patients [10, 11]. Recently, the SIRI has been proven to be an independent predictor of OS in patients with pancreatic cancer before and after surgery [15]. Its predictive ability is stronger than that of other inflammatory indexes, and it is associated with chemotherapy resistance, higher serum inflammatory cytokine/chemokine levels, and shorter survival outcomes. Geng et al. found that the SIRI is closely related to pathological grade, tumour size and TNM stage and is an independent predictor of oesophageal cancer patients; moreover, a low SIRI is significantly related to survival [9]. Li et al. found that the SIRI is closely related to age, tumour size, TNM stage, and lymphatic and peripheral nerve infiltration and is an independent predictor of gastric cancer patients [23]. Before us, Chen et al. indicated pretreatment SIRI with the advantage of repeatable, convenient, and non-invasive is a useful prognostic indicator for breast cancer patients who received neoadjuvant chemotherapy, and Hua et al. reported that the preoperative SIRI can predict the prognosis of operable breast cancer [18, 19]. These studies have reported the prognostic value of the pretreatment SIRI in breast cancer patients. These studies only assessed the prognostic value of the SIRI before pretreatment, but the value of the SIRI in postoperative research has not been reported. In this study, the SIRI was identified as a new independent prognostic marker of operable breast cancer patients. ROC analysis showed that the SIRI was better in predicting OS than the other inflammatory indexes (NLR, PLR, and MLR). Grade, SIRI, and TNM stage were integrated into a nomogram for breast cancer patients. The accuracy of this nomogram in predicting the survival of breast cancer patients is significantly higher than that of the TNM staging system. Changes in the SIRI after surgery are also significantly related to the prognosis of breast cancer patients. SIRI is easily detectable, with low cost and reproducibility. Therefore, it is believed that the SIRI and SIRI-based prognostic alignment are good indexes and tools for predicting the survival of breast cancer patients.
Systemic inflammatory indexes can be used to predict patient survival and may be related to a number of underlying mechanisms. Neutrophils infiltrate the tumour microenvironment, leading to angiogenesis in tumour tissue and promoting tumour cell proliferation, migration, invasion, metastasis, and even immune escape [24, 25]. The main reason is that the excessive release of alkaline phosphatase from neutrophils causes tissue damage, thus destroying the surrounding tissues of the tumour and helping the tumour spread and proliferate [24]. At the same time, the excessive release of alkaline phosphatase degrades immune globulin, complement fragments and receptors, blocks TNF-mediated apoptotic pathways and promotes tumour cell proliferation [24]. In addition, neutrophils can also release neutrophil elastase (NE), cathepsin G, MMP-8, etc. [24]. These proteases promote tumour cell migration by degrading ECM [26]. Neutrophils can also release proangiogenic factors such as MMP-9 and VEGF, promote angiogenesis in tumour tissues, and enhance tumour invasion [27]. Monocytes play an important role in the development and metastasis of tumours. Peripheral blood mononuclear cell-derived tumour-associated macrophages can inhibit the acquired immune response [28]. Lymphocytes mainly mediate cellular immune suppression or kill tumour cells, and they are important antitumour immune response cells in the body [29]. T lymphocytes infiltrating into tumours can kill tumour cells by releasing granzymes [30].
This study confirms the prognostic value of the SIRI in breast cancer, but there are still some limitations in this study. First, the evaluation of patients’ inflammation may have some deviations. SIRI can be affected by internal and external factors, including acute and chronic infection, lifestyle and glucocorticoids. Second, all subjects met the inclusion criteria, so there may be selection bias and unrecognized and uncontrolled biases. Third, the case data were from the same centre, and it was a retrospective study. Therefore, prospective, multicentre large-sample studies are still needed.
SIRI is a new prognostic marker and a potential marker for treatment response in patients undergoing radical mastectomy. The nomogram integrating SIRI can predict the survival time of breast cancer patients after surgery more objectively and reliably than AJCC-TNM stage and thus is helpful for screening high-risk breast cancer patients, developing reasonable therapeutic regimens and offering personalized consultations.
Footnotes
Acknowledgments
This work was supported by funding from the National Natural Science Foundation of China (81703162).
Conflict of interest
The authors declare that they have no competing interests.
Supplementary data
The use of the nomogram in a breast cancer patient. If there is a cancer patient with moderate histological grade, TNM stage II and a SIRI 
