Abstract
In 1977 Peter Kramer performed the first CAVH (continuous arteriovenous hemofiltration) treatment in Gottingen, Germany. CAVH soon became a reliable alternative to hemo- or peritoneal dialysis in critically ill patients. The limitations of CAVH spurred new research and the discovery of new treatments such as CVVH and CVVHD (continuous veno-venous hemofiltration and continuous veno-venous hemodialysis). The alliance with industry led to development of new specialized equipment with improved accuracy and performance in delivering continuous renal replacement therapies (CRRTs). Machines and filters have progressively undergone a series of technological steps, reaching a high level of sophistication. The evolution of technology has continued, leading to the development and clinical application of new membranes, new techniques and new treatment modalities. With the progress of technology, the entire field of critical care nephrology moved forward, expanding the areas of application of extracorporeal therapies to cardiac, liver and pulmonary support. A great deal of research made extracorporeal therapies an interesting option for the treatment of sepsis and intoxication and the additional use of sorbents was explored. With the progress in understanding the pathophysiology of acute kidney injury (AKI), new guidelines were developed, driving indications, modalities of prescription, monitoring techniques and quality assurance programs. Information technology and precision medicine have recently contributed to further evolution of CRRT, with the possibility of collecting data in large databases and evaluating policies and practice patterns. This is likely to ultimately result in improved patient care. CRRTs are 40 years old today, but they are still young and full of potential for further evolution.
Introduction
Exactly forty years ago Peter Kramer treated the first patient with continuous arterio-venous hemofiltration (CAVH) in the intensive care unit (ICU) of Gottingen, Germany (1). Acute renal failure was mostly treated with peritoneal dialysis or haemodialysis, but in critically ill patients, these modalities were often contraindicated or precluded due to severe cardiovascular instability. CAVH was well tolerated and easy to institute in ICUs where haemodialysis was not routinely performed. In the year following the first description of CAVH, a significant evolution of acute renal replacement therapy (ARRT) and related technology occurred, leading to the modern techniques of continuous renal replacement therapy (CRRT). In this process of evolution, the centre of Vicenza has always been leading the edge and it has significantly contributed to the birth of a new speciality called Critical Care Nephrology. In Figure 1 we report the history timeline of CRRT as seen from our point of observation. It may be incomplete or partial, but certainly every single minute of this history has been personally lived with passion and dedication.
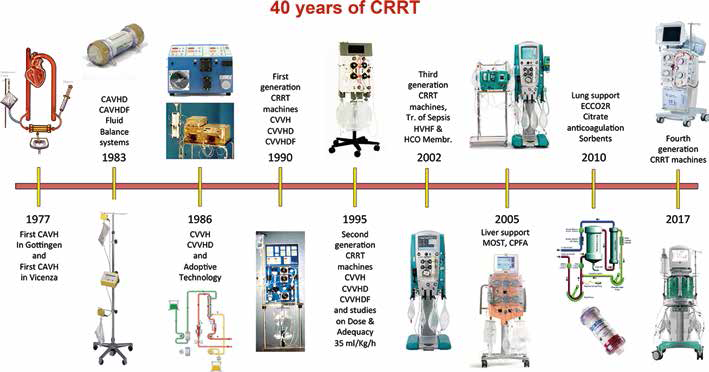
Timetable of the forty years of evolution in CRRT.
The Era of CAVH
The introduction of CAVH made it possible to perform renal replacement therapy even in ICUs not fully equipped or trained for hemodialysis. Our first patient was treated in Vicenza with the original Amicon 20 diafilter in 1978. We further refined our understanding and knowledge of CAVH by collaborating with Juan Bosch at the Mount Sinai Hospital in New York (2). In the late 1970s we extended the use of the technique used in adults to neonates with the use of specific Minifilters (Fig. 2) (3, 4). We developed specific filters with reduced flow resistance, suitable for operating in arterio-venous mode that are today preserved in our Vicen- za Dialysis Museum. In spite of several advantages, CAVH soon demonstrated limitations in efficiency and frequent complications due the need for arterial cannulation. This spurred new research for alternative techniques.
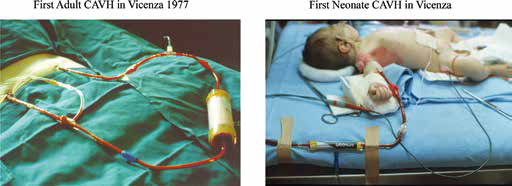
Vicenza, 1978: First CAVH treatment in an adult critically ill patient and first CAVH treatment in the world of a neonate with acute renal failure.
The Discovery of CAVHD and CAVHDF
A remarkable increase in treatment efficiency and urea removal was achieved with the addition of diffusion. New filters with 2 ports in the dialysate/filtrate compartment allowed the use of counter-current flow of dialysate, giving birth to continuous arteriovenous hemodiafiltration or hemodialysis (CAVHDF or CAVHD) (5). CAVHD- CAVHDF made it possible to treat hyper-catabolic patients, simply by increasing dialysate the flow rate up to 1.5 or 2 L/h. In the case of excessive ultrafiltration, fluid losses were partially or completely replaced, allowing accurate fluid balance control. The problem of arterial cannulation was still the main drawback of the technique.
Fluid Balance Control
Manual control of ultrafiltration was initially achieved by positioning the filtrate bag at different heights, thus modifying the negative (suction) pressure generated by the filtrate column. Delivery of replacement fluid was initially regulated manually and later new systems were designed to provide automatic fluid balance (6). The systems operated by gravity, using scales and electronic clamps, although peristaltic pumps soon replaced these simple mechanisms with more advanced equipment.
CVVH, CVVHD and CVVHDF
Because arterial-venous techniques were associated with significant complications related to arterial cannulation and low blood flow in the extracorporeal circulation, continuous veno-venous hemofiltration (CVVH) or hemodialysis (CVVHD) hemodiafiltration (CVVHDF) took over thanks to the availability of double lumen venous catheters and machines with a peristaltic blood pump (7–10). These changes improved the safety and performance of extracorporeal treatments in the ICU, allowing the use of high blood flows (>150 mL/min) and large dialyzers (>1.0 m2). This resulted in better control of hyper-catabolic states and other severe pathophysiological derangements. Initially veno-venous circuits took advantage of already existing technology, often creating a combination of devices (blood pump, UF pump, reinfusion pump, anticoagulation, etc.) that were not integrated and, therefore, unable to communicate and operate together, with the possibility of amplifying errors and complications. Adoptive technology allowed significant advances to be made, but it soon proved inadequate when a clear need for integrated and safer devices became evident in clinical practice.
Integrated Technology
In Vicenza we started the process of integration of blood and dialysate filtrate pumps in a single machine and pushed the industry of the field toward new research to create dedicated devices for CRRT With this effort, companies mostly involved in chronic hemodialysis began to collect components from chronic machines and to integrate them into machines for CRRT (11, 12). Using this approach, several companies made new equipment: B. Braun moved from TRIO (3 separate pumps) to the compact ECU Carex machine (Fig. 1) designed by the engineer, Mr. Frigato, in Mirandola. Baxter moved to the integrated BM 25; and Hospal generated the DM 32 from the blood module of the Monitral machine. Medica built an integrated version of the Equapump and Equaline, while in Germany, Fresenius Medical Care commercializsed the DM08 with 1 blood pump and 2 pumps for dialysate or fluid balance (13–15). Bellco utilized the blood module of the Multimat chronic machine to create the Multimat B Acute version, and Gambro adjusted a special version of the AK 10 module for continuous dialysis. All these machines derived from the technology generated for chronic hemodialysis, but they represented the seeds for the upcoming generation of specific CRRT equipment (16, 17).
The PRISMA® Revolution
With the progress in understanding the pathophysiology of AKI and its clinical implications, the targets for renal replacement therapy became clearer (18–25). The indications for CRRT were defined and the practice of CRRT was better identified in the first edition of the book
CRRT Dose Quantification
Started in 1995, in Vicenza we concluded the first randomized trial on treatment dose in the year 2000 (26). We underlined the importance of a minimum quantity (dose) of treatment and we indicated 35 mL/kg per hour as the optimal target according to the trial result. This target, with a minimum of 80% delivery of the prescribed dose, began the golden standard for adequate CRRT and improved clinical outcomes. Subsequent studies have demonstrated that lower doses can be equally safe and successful in treating the critically ill patient (27–33), although effective delivery often differs significantly from the prescription (34–37). The new machines developed by industry with the possibility of moving toward more sophisticated techniques such as coupled plasma filtration-adsorption (CPFA) and high-volume hemofiltration (HVHF) fulfilled these new performance requirements (–41). Improved usability and user-friendly interfaces with systematic instructions made CRRT machines popular and widely used in every ICU (17).
CRRT in Sepsis
The original observation that higher doses of treatment could be beneficial in patients with sepsis introduced the rationale for HVHF and CPFA with the intent to remove chemical mediators (42–45). HVHF or CPFA showed potent immunomodulatory effects in sepsis. Since a cytokine network that is synergistic, redundant, autocatalytic and self-augmenting characterizes sepsis and systemic inflammatory response syndrome, it is unlikely to achieve the control of such a nonlinear system by simple blockade or elimination of some specific mediators. Thus, we hypothesized that the unselective nature of mediator removal by extracorporeal therapies could be a potential advantage, capable of reconstituting the immune-homeostasis of the septic patient, as suggested by the ‘peak concentration’ hypothesis (42). Higher volumes and complex techniques were made possible by the third generation of CRRT machines such as the Prismaflex (Gambro), the Equasmart (Medica), The Lynda (Bellco), The Multifiltrate (Fresenius Medical Care), The Acquarius (Edwards Life Sciences) and others.
From renal replacement to multiple organ support therapy
The effect of different modalities of CRRT on length of stay and recovery of renal function in the general population is still under evaluation, since the case mix changes in every study and the population treated is not homogeneous. Further research is needed in this field, although it has become evident that Precision CRRT (personalized prescription) should be adopted in order to optimize results in single patients, even in the absence of the documented benefits of 1 specific technology for the general population. Adequate technological support becomes mandatory to fulfil all these expectations and new machines have been upgraded with specific circuits to support organs other than the kidney. The possibility of supporting or partially replacing the function of organs such as the liver, the lung and the heart with modified extracorporeal circuits, make multiple organ support therapy (MOST) a reality in the critical care setting. CRRT machines become platforms for multiple organ support via extracorporeal therapies carried out with specific biomaterials and devices (45, 46).
Sorbent Therapies
Hemoperfusion has been available for detoxification purposes since the second part of the last century. Zeolites and charcoal were the most common sorbents, although soon a new generation of more hemocompatible materials became available. In recent years, cartridges with polymixin-B-coated polystyrenic fibres designed for the adsorption of endotoxin have been used for the treatment of sepsis. This therapy seems particularly beneficial in patients with post-surgery abdominal septic shock (47, 48). New sorbents, derived from the original experience carried out by our group in the early years of the new century, are today appearing as a new option for the treatment of sepsis (49–).
Latest Generation of CRRT Machines
The latest generation of machines available on the market today, which represent the evolution of the past 4 decades of research and development is shown in Figure 3. Specific machines have now been designed to permit safe and reliable performance of the therapy. These new devices are equipped with a user-friendly interface that allows for easy performance and monitoring. The apparent complexity of the circuit is made simple by a self-loading circuit or a cartridge that includes the filter and the blood and dialysate lines. Priming is performed automatically by the machine and pre- or post-dilution (reinfusion of substitution fluid before or after the filter) can easily be performed by changing the position of the reinfusion line. These new machines permit all CRRT techniques to be performed by programming the flows and the total amounts of fluid to be exchanged or circulated as a counter-current dialysate throughout the session.
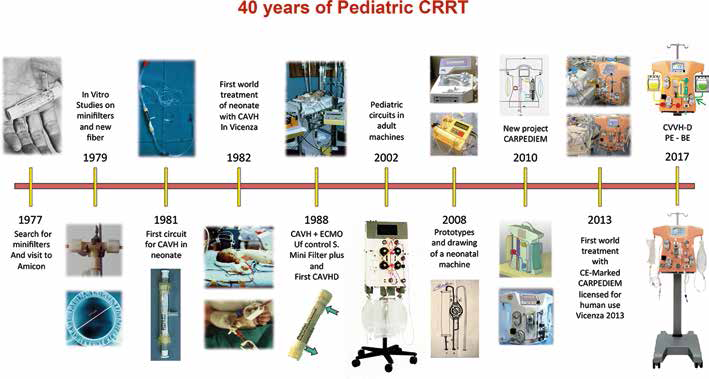
Timetable of the forty years of evolution in pediatric CRRT.
Precision CRRT
The Acute Disease Quality Initiative consensus group has stressed the importance of applying the principles of precision medicine to CRRT (54). The group has invited CRRT experts and manufacturers to utilize a standardized nomenclature and sign a document called “Charta of Vicenza” (55) agreeing to follow the recommendations proposed by 2 detailed publications (56, 57). This was a starting point to generate a harmonized terminology and to advance in a standardized environment for the generation of useful pragmatic trials. Today we have several examples of new machines that include software and terminology coherent with the recommended nomenclature.
The consensus group also focused on the identification of patients requiring CRRT and the correct timing of application, technological needs and expected advances, pointing out the desirable characteristics of new equipment, membranes and the importance of integration of information technology into the process of patient care and decision-making (58–61).
Information Technology and Connectivity
The ADQI group has recently recommended the use of modern information technology (IT) tools to improve practice and patient care. Such tools can be used for data acquisition and storage but also for treatment monitoring (62). Manual, authorized or automatic feedback technology is available today in chronic dialysis machines and has been advocated for CRRT machines as well. Integration of patient and machine signals through IT tools and connectivity with electronic health record (EMR) and data collection systems will be required to allow pragmatic trials and make big data registries available for analysis. Data can then be used for Quality Assurance (QA) and for Continuous Quality Improvement (CQI) purposes in the centre, in the region and even in multinational data collection studies.
Machine connectivity can be provided via different tools. Machine and patient chip-cards can be used to extract data from single treatments from the front-end terminal (CRRT machine). Cable or wireless connectivity may permit the download of technical and clinical parameters from single or multiple machines and to analyse single treatment data as well as trends or statistics in multiple treatments. Cloud-based connectivity could help clinicians to generate virtual registries and analyse the performance of single treatments or centres in absolute terms, or relative to other units. This may result in important feedback to clinicians, to either strictly control outliers, or to change policies and procedures in the event of repeatedly unsatisfactory results.
Data collected and stored in EMRs may be rapidly evaluated and managed by ad hoc designed electronic sniffers that can alert clinicians about dangerous trends or unwanted effects of CRRT. Solutions to the problem may be listed as suggestions or even automatically fed back into devices such as pumps and CRRT machines. The feedback may either require manual application of the necessary change by a nurse/physician, authorization of an operational change proposed by the system, or operate automatically.
Current and Future Perspectives
There are several areas of investigation where evolution in new therapeutic options and new devices is likely to occur. Different biomaterials and surface modifications have been introduced on semipermeable membranes to increase biocompatibility, to reduce thrombogenicity and the tendency toward fouling, and finally to modify sieving and adsorption properties. Vitamin E-coated membranes represent an example in which alpha-tocopherol has been covalently bound to a polysulfone membrane to reduce oxidant stress (63). These and other new membranes should be further investigated to elucidate the potential for improving the inflammatory pattern in sepsis.
Software integration should become a tool for new equipment to perform safer and more efficient CRRT treatments. The integration of bioimpedance and on-line haematocrit measurements may result in important effects on hemodynamic stability and treatment delivery (64). The integration of blood temperature monitoring may have the potential to control thermal energy balance and the patient's temperature. This could allow a specific energy balance (KJ/h) or a target temperature control to be achieved by adjusting the dialysate or replacement fluid temperature according to signals coming from temperature sensors placed on blood and dialysate lines. A significant heat loss can in fact occur when the extracorporeal circuit is exposed to room temperature (65).
Automated circuit pressure control with flow adjustment feedback could provide the best possible blood flow in presence of a malfunctioning catheter or could provide early warning of access malfunction, preventing inadequate treatment delivery.
On-line chemical sensors for acid-base and electrolytes may provide the basis for continuous control via biochemical feedback on dialysate and replacement fluid composition.
Miniaturized and Wearable Technology
There is a great deal of interest in applying nanotechnology, microfluidics and other emerging sciences to the field of renal replacement. In Vicenza we tested the first wearable system for ultrafiltration (66). For the original wearable belt, we mostly used components off the shelf, but we recently made new developments by creating a new design and applying newly conceived, dedicated technology (67–69). Wearable devices are mostly conceived for ambulatory care and out-of-hospital patients. Nevertheless, it seems that miniaturized systems may become useful even in the acute patient, allowing mobility, low-priming-volume extracorporeal circuits and better care at the bedside.
Specific Technology for Small Infants and Paediatric Applications
An interesting development in the process of machine and device miniaturization is the application of small-sized components for infants and children for whom the adult technology seems to be clearly inadequate. The requirement for specific technology in the field of paediatric CRRT emerged in the early 1980s (70). Over the years we personally contributed to a significant evolution of the field (71–74) and the historical milestones of paediatric CRRT are reported in Figure 3. Quite recently, based on the observation that every CRRT machine is conceived for adult patients and the use in neonates and small infants is precluded or off-label, we decided to undertake a special project for the creation of miniaturized equipment specifically designed for these small patients. We started from scratch and we moved, through artisanal design and rough assembly of prototypes, to an industrially designed machine called CARPEDIEM (Cardio Renal Pediatric Dialysis Emergency Machine). This evolution has captured new interest in the treatment of AKI in neonates and has allowed different techniques to be performed with the possibility of precisely adjusting the treatment dose and fluid balance in babies with body weights as low as 1.5 kg (75, 76) (Fig. 4). There is a consensus that such a specific technology will probably modify the outcome of AKI in neonates and small infants.
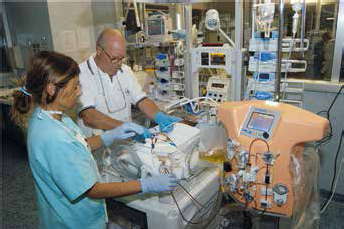
First CRRT treatment with CARPEDIEM (Cardio renal Pediatric Dialysis Emergency Machine).
Conclusions
Over the last 40 years, CRRT has been widely utilized for the management of AKI in critically ill patients (77–79). The Vicenza Centre has heavily contributed to important technological advances in machine design, to the expanded application of novel and complex modalities and, finally, to the optimization of extracorporeal therapies in the ICU, ultimately leading to improved patient outcomes.
We pushed the transition from CRRT as renal replacement therapy to the concept of MOST (Multiple Organ Support Therapy) (80), upgrading the extracorporeal circuit to provide support for patients with multiple organ dysfunctions. Information and communication technology together with big data collection represent the new frontier (81–85) for personalized prescriptions and measurement of results to achieve the desired level of precision CRRT (86).
I strongly recommend using the history of the last 40 years of CRRT not only for a simple celebration of an important anniversary, but also for building the future of our discipline with improved treatment of acute kidney injury and organ dysfunction in the critically ill patient.
Footnotes
Financial support: None.
Conflict of interest: None.
