Abstract
Purpose
Dialysis is a highly quantitative therapy involving large volumes of both clinical and technical data. While automated data collection has been implemented for chronic dialysis, this has not been done for acute kidney injury patients treated with continuous renal replacement therapy (CRRT).
Methods
After a brief review of the fundamental aspects of electronic medical records (EMRs), a new tool designed to provide clinicians with individualized CRRT treatment data is analyzed, with emphasis on its quality assurance capabilities.
Results
The first platform addressing the problem of data collection and management with current CRRT machines (Sharesource system; Baxter Healthcare) is described. The system provides connectivity for the Prismaflex CRRT machine and enables both EMR connectivity and therapy analytics with 2 basic components: the connect module and the report module.
Conclusions
The enormous amount of data in CRRT should be collected and analyzed to enable adequate clinical decisions. Current CRRT technology presents significant limitations with consequent lack of rigorous analysis of technical data and relevant feedback. From a quality assurance perspective, these limitations preclude any systematic assessment of prescription and delivery trends that may be adversely affecting clinical outcomes. A detailed assessment of current practice limitations is provided together with several possible ways to address such limitations by a new technical tool.
Keywords
Introduction
Acute kidney injury (AKI) is a complex syndrome occurring with high frequency in critically ill patients admitted to intensive care departments. Severe clinical derangements are involved in the syndrome, including acid-base disturbances, uremic intoxication, electrolyte disorders and fluid overload. The technology involved in kidney and other organ support by extracorporeal therapies is different from that utilized in chronic hemodialysis with a significant attempt to maintain hemodynamic stability and slow gentle correction of homeostatic derangements. This is often managed by prescribing and delivering continuous renal replacement therapies (CRRT).
Dialysis in general is a highly quantitative therapy. However, while in chronic dialysis automated data collection and reporting have been implemented in the last decade, this has not been done so far for acute renal replacement therapies and in particular for patients treated with CRRT.
In intensive care patients, clinicians must efficiently manage large volumes of both clinical and technical data. This is especially true for AKI patients receiving CRRT. Furthermore, because the clinical status of these patients is constantly changing, this therapy continuously generates technical data that must be evaluated rapidly so that any needed changes in therapy prescription can be made. The manner in which this technical information is assessed and used to make clinical decisions potentially impacts patient outcomes, not only on an immediate basis (e.g., occurrence of hypotension) but also in the intermediate term (e.g., timely discharge from the ICU) and long term (e.g., recovery of renal function, survival). The need for efficient processing of data in the management of critically ill AKI patients was highlighted recently during an Acute Dialysis Quality Initiative (ADQI) conference focused on the implications of “big data” for this population (1).
The reality of current CRRT technology, however, is that significant limitations in meeting the substantial clinical information needs for this patient population exist. Current machines do not allow for automated data collection and even fail to use standardized terminology allowing comparisons across different institutions. Of note, the latter problem has recently been the focus of a collaborative meeting and consensus standards are forthcoming (2). As a consequence, rigorous and ongoing analysis of technical data and the ability to interpret this information in the context of the clinical status of the patient are difficult. Moreover, from a quality assurance perspective, these limitations in current CRRT technology preclude any systematic assessment of prescription and delivery trends that may be adversely affecting clinical outcomes. In summary, a new approach to information management in CRRT is needed.
In this report, a detailed assessment of the limitations of current clinical practice with regard to CRRT-related data analytics is provided. Subsequently, we provide several ways in which these current limitations can be addressed. After a brief review of the fundamental aspects of electronic medical records (EMRs), a new tool designed to provide clinicians with individualized CRRT treatment data is discussed, with emphasis on its quality assurance capabilities.
Data Management in CRRT Patients: Current Limitations
In the most recent ADQI consensus conference, the participants rightly identified the need for AKI/CRRT patient management to align with the current focus on personalized medicine. In this regard, the ADQI participants proposed the term “precision CRRT” to highlight the need for technology to be applied on an individualized basis (3). Moreover, the participants stressed that management of the critically ill AKI patient is a care continuum requiring different technology applications (4).
“Dynamic CRRT”: Data Management Considerations
Unfortunately, current clinical practice regarding patients treated with CRRT is far from individualized from an information management perspective, in part due to technical limitations of CRRT machines. As opposed to the automated, real-time data capture that characterizes many interventions in the ICU (5), CRRT machine data are collected and analyzed manually. This is a laborious, time-consuming process that frequently delays necessary treatment interventions. Moreover, in the stressful, often chaotic environment of an ICU, manual recording may lead to errors, potentially impacting patient outcomes in an adverse way (6). Finally, the valuable nursing time consumed by this inefficient process comes at the expense of direct patient care.
This inefficiency inherent in current CRRT practice is a major barrier to the achievement of precision CRRT. In keeping with a personalized medicine approach, the ADQI group has also highlighted recently the dynamic nature of CRRT, characterized by the need for frequent assessment and calibration of the prescription on an individualized basis (7). In addition to clinical assessment of the patient, important technical components of the dynamic CRRT analysis include solute clearance, delivered/prescribed dose, effective treatment time, solute control indicators, and circuit/filter pressure trends. Of note, these parameters are included in a battery of quality metrics recently proposed for CRRT (8). The term “dynamic” implies that nearly real-time CRRT machine data need to be available for closure of the feedback loop shown in Figure 1. While any prescription changes needed to accomplish this have to be made “manually” by the clinical team at present, it is hoped that such changes can be made automatically by the CRRT machine in the future.
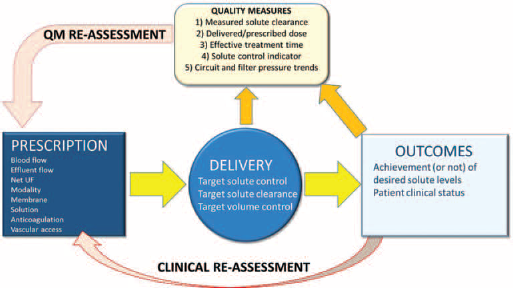
Concept of dynamic CRRT. A therapy prescription/delivery feedback loop incorporating treatment quality metrics is shown. Reprinted with permission from ADQI.net. “QM” refers to quality metric and “UF” refers to ultrafiltration.
A dynamic CRRT program also implies the ability to use information technology beyond the real-time phase for longer-term purposes. At present, CRRT machine data are not routinely stored in an accessible warehouse, rendering impossible the systematic generation of reports for review by the clinical team. As such, clinicians are unable to assess historical trends easily on a facility-level basis, especially those related to the basic quality metrics mentioned above, or to use these data for quality assurance purposes.
Lack of Connectivity between Clinical and CRRT Machine Data
As mentioned, the ICU team is faced with the daunting task of continuously evaluating not only the barrage of technical data produced by the CRRT machine but also the therapy's effect on the clinical status of the patient. On an instantaneous basis, clinical indices that have the potential to be influenced significantly by CRRT are related to the hemodynamic, respiratory, and electrolyte/acid-base status of the patient (9, 10). An integral aspect of modern critical care is hemodynamic monitoring involving the use of both invasive and noninvasive techniques (11). However, unlike the technical data generated by a CRRT device, the parameters generated during hemodynamic monitoring are typically incorporated into an ICU's clinical data system (12). The ICU team has full access to the hemodynamic data in real-time, allowing rapid clinical decisions to be made (e.g., dosage changes in vasoactive medications). Similar real-time data acquisition tools also allow ICU staff to make timely interventions in response to parameters related to mechanical ventilation, such as tidal volume and peak airway pressure. Additional parameters relevant to CRRT, such as serum electrolytes and arterial blood gases, also can be fed automatically to an ICU EMR, providing the clinical team access to a wide array of clinical information in a single repository.
The vast data pool located in an EMR permits a clinical snapshot at any given moment for a particular patient and the possibility to evaluate the clinical impact of different ICU interventions in a rapid manner. Moreover, the integrated and comprehensive nature of the EMR data creates the opportunity to establish a robust quality assurance program by providing quality metrics established for a given ICU (13). As mentioned above, these advantages are predicated upon connectivity between different sources of information and the EMR. Unfortunately, this connectivity, specifically between the CRRT machine and the EMR, has not existed traditionally.
In the discussion below, we propose several ways in which the above limitations with respect to CRRT machine data management can be addressed.
CRRT Information Management: “charting” a New Course
The access of clinicians to a comprehensive CRRT treatment database may improve their prescription and delivery of therapy at a given institution in several ways. First, for a specific patient with a difficult clinical course, (repeated filter clotting after only a few hours of circuit use, for example), relevant parameters (such as filter and access pressures) can be compared not only to historical norms but also to previous patients with a similar clinical course. The insights provided by such an analysis may facilitate the root cause analysis and expedite the necessary interventions.
Second, a robust CRRT treatment database could provide trending data that suggest a gradual departure from a previously established best demonstrated practice. Based on the same example above, an increase in the incidence of early, frequent filter clotting may be found to be associated with very high negative access pressures. Such a finding provided by this trending analysis could prompt a reevaluation of institutional vascular access procedures, including placement and nursing practices.
Finally, in addition to assessments within a given institution, a comprehensive CRRT database would also allow comparisons between institutions. Based on the assumption that AKI/CRRT quality metrics will eventually be incorporated into clinical practice (as is the case in end-stage renal disease) (14), comprehensive data collection, processing, and analysis will become mandatory. Included among these data will most certainly be CRRT machine data, which will be used for benchmarking according to standards established at a local, regional, national, or international level. A related benefit would be the opportunity for institutions to participate in clinical trials, which could be conducted on a large scale as long as standard sets of data are collected. At this stage in the evolution of CRRT, clinical practice most likely will be influenced more significantly by pragmatic trials, such as registries, (15) than large, randomized studies. However, data capture in current clinical practice is not configured to conduct these sorts of pragmatic trials efficiently.
With respect to a roadmap for the future, we believe the following are the desired connectivity characteristics for CRRT machines:
Information from the CRRT machine should be provided to the clinician in real-time and should prominently feature consensus-based quality metrics.
CRRT machine data should be offered to the clinical team in the context of the patient's current clinical status. As such, the information package offered on a continuous basis will be a combination of machine data and clinical data (e.g., hemodynamic parameters, fluid balance, metabolic parameters).
Monitoring and data collection systems should be designed to assist clinicians in assessing whether clinical goals have been reached and to support them in making the necessary prescription changes to reach newly-instituted targets. These systems can be based on various connectivity platforms at the machine, local, or cloud-based level (Fig. 2).
Connectivity options for CRRT machines. Reprinted with permission from ADQI.net. CRRT machine: Machine and patient chip cards can be used to extract data manually from a terminal Local: Cable or wireless connectivity may permit the download of technical and clinical parameters from single or multiple machines. Moreover, this approach can be employed to analyze single-treatment data or statistics in multiple treatments. Cloud-based: Virtual registries could be created for the analysis of single treatments or facility performance in absolute terms or in relation to other units. This will allow differentiation between outlier patients and undesirable trends, the latter of which could be mitigated by a policy change.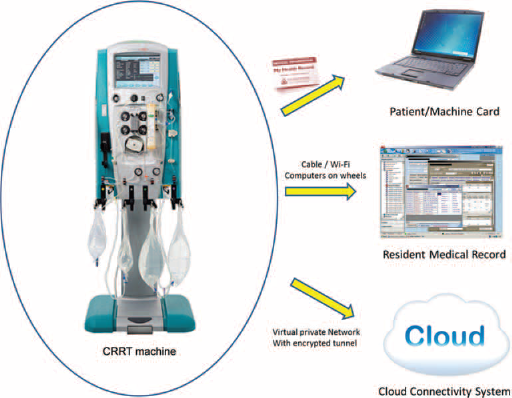
For the time being, active clinician intervention should be 1 component of a biofeedback loop that constantly responds to changes in a patient's clinical status (16). Each prescription change, suggested by the machine at the user interface, must either be authorized or implemented manually. In the future, experience in the chronic dialysis realm (17) can be leveraged to produce a fully automatic feedback loop for the CRRT machine (see below).
The CRRT machine data should include elements related to alarms and their management, especially when treatment delays occur.
In the future, the automated biofeedback mechanisms incorporated in current chronic dialysis machines should serve as a template for CRRT machines, including online tools to estimate delivered dose and fluid status. The ultimate goal for all biofeedback systems is to minimize the cumulative time that therapy is delivered outside of the prescribed target range.
Electronic Medical Record (EMR): The Basics
Electronic medical record (EMR) is the common term for a hospital's data management system, which may capture data across an entire hospital or from only a specific hospital care area (e.g., the ICU) or function (e.g., the clinical chemistry laboratory). An ICU's EMR may receive data from numerous sources, although a common mechanism by which data are provided to the EMR remains manual input. In the ICU, substantial amounts of time may be spent by the care team in entering vital signs, input/output parameters, and other routinely collected data. On the other hand, some ICUs have EMRs allowing automatic electronic data transfer from medical devices and other systems. However, as noted already, this latter capability has traditionally not included CRRT machines. Nevertheless, the potential uses of EMR data for the management of AKI patients have recently been highlighted (18).
The term “connectivity” encompasses the myriad of requirements for transmitting data to the EMR. These information technology requirements are substantial and specify not only the necessary infrastructure but also the competence, neither which may exist at many institutions. Health Level 7 (“HL7”) is the connectivity platform (“language”) upon which EMRs have been standardized (19). A detailed analysis of HL7 is beyond the scope of this paper but Wikipedia provides the following description:
HL7 International specifies a number of flexible standards, guidelines, and methodologies by which various healthcare systems can communicate with each other. Such guidelines or data standards are a set of rules that allow information to be shared and processed in a uniform and consistent manner. These data standards are meant to allow healthcare organizations to easily share clinical information. Theoretically, this ability to exchange information should help to minimize the tendency for medical care to be geographically isolated and highly variable.
It should be noted that this definition refers to the common scenario in which a given healthcare institution may have multiple EMRs (“systems”) for clinical data collection. Within HL7, the ADT (Admission, Discharge, and Transfer) system is the most commonly used approach and is comprised of 51 different types of messages that frequently originate from “trigger events” (20). This messaging system is critical in maintaining efficient patient flow.
However, ADT messaging may not be the ideal communication language for medical devices. The manner in which data from medical devices are provided to EMRs is particularly complex, as they communicate with a vastly more diverse and often incompatible range of languages. Many devices have a proprietary non-HL7 format which requires translation into HL7. In addition, the complexity of requiring all medical devices to employ HL7 solution messaging may exceed what is appropriate for a device that is very simple from an end-user perspective. For example, the need for a caregiver to process ADT messages from an EMR to get electronic “permission” to take a single measurement with a simple, one-button blood pressure meter might be unwieldy.
Even with agreement on HL7 messaging, the data provided by a particular medical device are unique (and sometimes proprietary) – a CRRT machine and a hemodynamic monitor provide vastly different information, for example. As such, each device must have a “destination” in the EMR requiring a mapping of data from that device. Finally, while the capability for automated electronic data transfer may improve efficiency and reduce the possibility of entry errors, an additional consideration is the ability to use the transferred data to evaluate treatment efficacy and efficiency. As discussed below, a system designed to obtain this additional clinical and operational value has recently been developed for a CRRT device.
Overview of a New CRRT Connectivity Platform
The first platform addressing the above limitations of current CRRT machines is the Sharesource system (Baxter Healthcare), which provides connectivity for the Prismaflex device (Baxter). This system enables both EMR connectivity and therapy analytics with a flexible, tiered approach and has 2 basic components: a connect module and a report module. The connect module capabilities are reported in Table I.
Connect module capabilities

The report module comes in 2 forms, based on the institution's EMR capabilities and its clinical goals. One level contains only data collected from the CRRT machine, including basic prescription parameters (e.g., flow rates, filter type, modality), filter life span information, treatment downtime (21), prescribed and delivered dose (22), fluid removal parameters and alarm management information (Fig. 3A). Each month can be seen at a glance (e.g., May, shown in Fig. 3B).
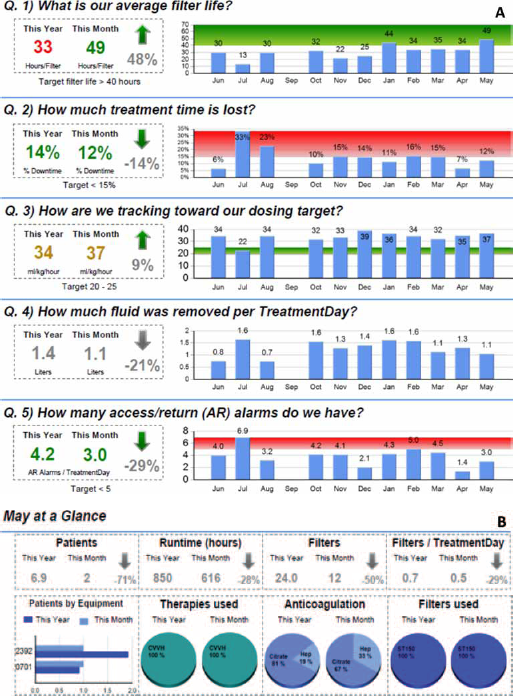
(A) Example of report module providing CRRT machine data for clinical review; (B) 1 month of data at a glance.
A more comprehensive and clinically useful version of the report module combines CRRT machine data with patient data from the EMR, providing the potential to assess the timing of CRRT initiation in relation to KDIGO AKI stage in a relative short-term (23), the influence of CRRT on a variety of laboratory and physiologic parameters, the calculation of the degree of fluid overload and efficacy of fluid management with CRRT (24), and the correlation of CRRT efficacy with other ICU interventions, such as ventilator duration and vasopressor requirements. Furthermore, statistical data about the treated population can be analyzed upon demand (Fig. 4A). Again, each month can be seen at a glance (Fig. 4B). In the longer term, a second-order report will also allow for historical trending, especially in the context of established quality metrics.
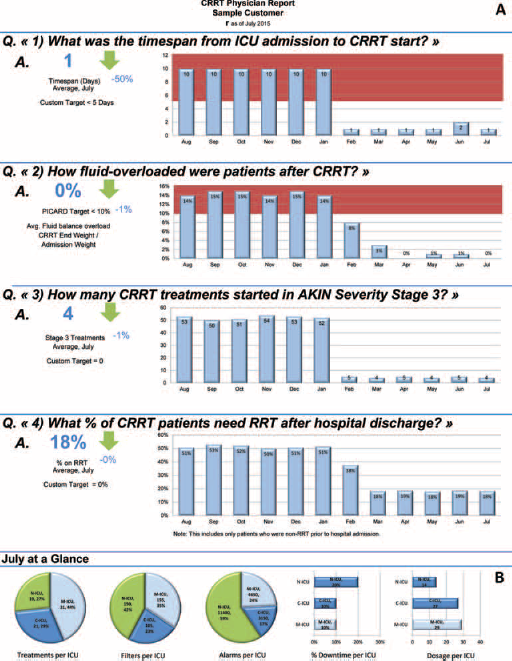
(A) Example of report module in which CRRT machine data are combined with patient data from the electronic medical record; (B) 1 month of data at a glance.
For institutions without the requisite IT infrastructure and expertise, the simple connect module can generate a report using an available data card available for use with the CRRT machine. Without any IT support at all, ICU staff can collect data monthly by copying files from the data card located in the back of the device to produce the report with an ordinary desktop or laptop PC. On the other hand, institutions with more advanced IT capabilities can utilize a secure server housed at their institution to receive CRRT machine data from both the connect and report modules. These are combined to allow electronic transfer to the EMR (paperless/automated charting) and monthly reports for the institution without the need to copy data card files manually. Finally, institutions with the most sophisticated IT capabilities have the ability to generate all of the above in addition to reports providing clinical context for more advanced insights.
Conclusions
The ICU is a complicated clinical care environment and efficient data management is essential for the optimal management of critically ill AKI patients. This is especially the case for patients treated with CRRT, for whom the volume of data generated and processed is prodigious. In this report, we have discussed the historical limitations of CRRT machines from a data management perspective and have proposed a set of attributes which can potentially help guide the development of future CRRT devices with improved connectivity capabilities. A recently developed CRRT data management system offering clinicians the opportunity to improve clinical care and outcomes has been discussed. The system does not work as a remote console of the dialysis machine but rather as a data collection system that allows outliers, average performance measures and effective adherence to prescribed values to be identified.
Some limitations of the system include limited connectivity with different brands of CRRT machines and the need for integration with the EMR to achieve combined patient and machine data. Nevertheless, this may represent an advancement in the management of CRRT. Every center can decide on the desired target performance along with the relevant quality assurance and continuous quality improvement policies. In the event of outliers, adaptive correction measures can be implemented to improve the prescription in that treatment or individual. If the team observes a repeated deviation from the desired target in multiple patients, a change in policy is likely to be requested. Physicians and nurses can therefore utilize the system as a tool to improve quality of care.
Footnotes
Financial support: Dr. Clark received consulting fees from Baxter Healthcare.
Conflict of interest: Dr. Clark owns Baxter stock.
