Abstract
Since implantable left ventricular assist devices (LVAD) with smaller configurations became available for bridge-to-transplant or even destination therapy in patients with end-stage heart failure, an increasing number of patients with these devices are receiving home medical management. However, these patients may be anxious about potential complications such as pump failure, thromboembolism, and infections that may occur during home management. To provide a sense of security during home management of patients with LVAD and to establish an ideal shared-care system, we developed a patient-centered cloud-based home management system for patients with LVAD. In this case report, we describe this system and report a trial of it in a 64-year-old patient with an LVAD.
Introduction
It is well recognized that home-managed patients with left ventricular assist devices (LVAD) tend to be anxious about potential complications that may lead to serious morbidities or even mortality (1–3). To reassure patients and properly treat such complications, it is essential to detect them at the earliest stage and to share information between patients and care providers. Therefore, we developed a cloud-based home medical management information sharing system called
Case Report
The patient was a 64-year-old male with end-stage heart failure due to a dilated form of hypertrophic cardiomyopathy. He underwent implantation of a HeartMate II LVAD (St. Jude Medical, St. Paul, MN, USA), in January 2014 at Kyoto University Hospital in Japan. His postoperative course was uneventful and he was discharged from hospital after learning how to use the
Information Sharing System
The
LVAD@home
The data items for
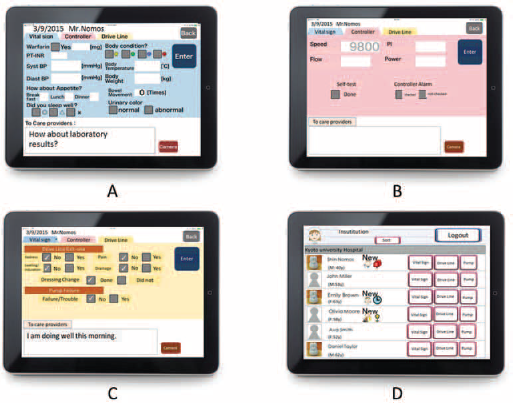
Browsing and input screens of
Communication security
Security of this system is assured by the following. The applications are only available from the Uniform Resource Locator (URL) for exclusive use of a device for which an IC Card ID consisting of 19 digits unique to that particular iPad has been registered. All communication is encrypted using Secure Socket Layer (SSL) technology. For recording and browsing of data, client authentication is used in addition to an ID and password, making it impossible to communicate with the server using a device other than the one that has been approved.
Data analysis
We analyzed the data derived from 305 consecutive days during the study period. The patient sent data of vital signs or pump parameters for 278 days, and photographs of his DLES for 175 days. In addition, the patient entered free text messages 145 times. During the study period, the attending physicians replied to the patient's text messages 72 times. Among 145 free text messages from the patient, the most frequent questions and reports were regarding the DLES (48%), followed by inquiries regarding medications and physical conditions (17%), pump parameters (10%), and minor items such as expression of thanks, greetings and so on (21%). The responses sent from the attending physicians to the patient included advice regarding the DLES (46%), followed by medications and physical conditions (15%), and pump parameters (10%) (Fig. 2).
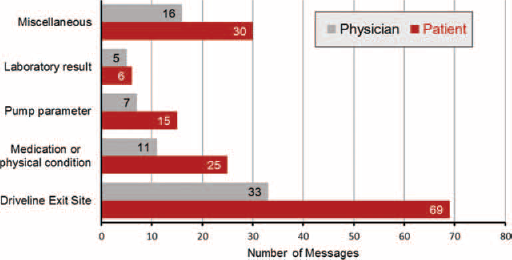
Free text message or question content analysis. The most frequent questions and reports sent by free text messages concerned the DLES in both the patient and the physicians.
We identified 2 useful instances that contributed to early detection of signs of DLES infection. In one instance, a physician started to suspect DLES infection based on the pictures sent by the patient, then asked the patient to visit the clinic earlier than his next planned appointment (Fig. 3A). At the clinic, a culture of the DLES was analyzed and he was diagnosed with methicillin-sensitive Staphylococcus aureus infection. The patient was successfully treated with IV antibacterial agents without developing a pump pocket infection or systemic bacteremia. The patient continued to send progress reports with pictures of the DLES thereafter, and did not require further treatment. In another instance, a physician identified abnormal hemorrhage at the DLES (Fig. 3B) and sent a message asking the patient to come to the clinic. The patient underwent a hemostasis procedure (Fig. 3C). The patient continued to report his DLES bleeding status and the description of the effusion together with pictures (Fig. 3D).
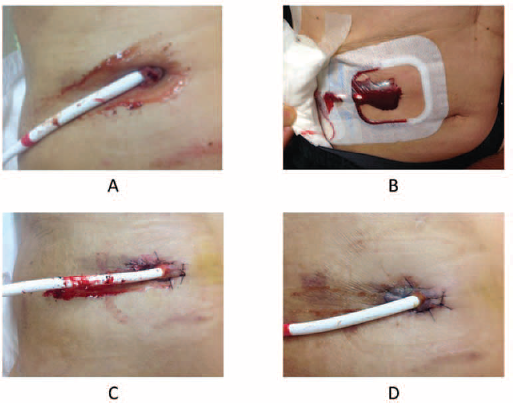
Sample photographs of the driveline exit site sent by the patient. (
Discussion
Since implantable LVADs were approved as bridge-to-transplant devices for patients with end-stage heart failure, the number of patients who receive LVADs has increased every year due to donor organ shortage. In addition, more and more elderly patients are now undergoing LVAD implantation as destination therapy (4). This has resulted in an increasing number of patients who receive home medical management (5). However, patients with LVAD are at risk of life-threatening complications such as pump infection, pump thrombosis, systemic thromboembolism, hemorrhage, and device failure (1–3). Therefore, daily follow-up is mandatory even in the context of home management, and early detection and treatment of such complications is very important to improve long-term survival and quality of life (6).
In normal home management after LVAD implantation, patients are typically trained before discharge to perform self-care and physicians confirm daily self-care through paper forms completed by the patients that are collected on outpatient visits once or twice a month. Although designated personnel, such as LVAD coordinators, can communicate with the patients and family as needed, this normal system does not allow for real-time information sharing between the heart team at an LVAD implant center and local healthcare providers, which is important for home management (7). To the best of our knowledge, this is a first report on a patient-centered, cloud-based, home management system for patients with LVADs.
Generally, the telephone and email are regarded as person-to-person communication methods (8), thus, patients sometimes hesitate to contact care providers directly for minor issues even during daytime and abnormalities tend to be left to the judgment of the patients and family. Obtaining advice regarding minor but potentially serious issues may result in a greater sense of security for patients under home management (9). Furthermore, unlike the telephone or internet videophone, information in the
Because this is a preliminary clinical study, at present, healthcare professionals are not required to review and respond to the data as a routine activity. Therefore, it is not clear where the responsibility lies when problems arise (e.g., an adverse event reported by a patient that was overlooked). These problems regarding remote monitoring and ultimate responsibility will need to be discussed in the future. In addition, although
Meanwhile, the costs of operating this system have not been considered. It is unclear whether the patients or care providers should cover the cost, or whether it should be covered by public funds. It is likely that reimbursing this kind of remote monitoring system may improve quality of care in LVAD patients, just as it has in patients with other devices such as implantable cardioverter defibrillators (11). Going forward, it will be necessary to demonstrate the utility of the system from the perspective of medical economics. Implementation of this system should reduce the number of hospitalizations and the burden on healthcare services in the community.
Since this study was limited to a single patient, the data might be biased. It will be necessary to increase the number of patients studied and examine the use of the device in many institutions under various conditions in the future.
Conclusions
A patient-centered, cloud-based, medical information sharing system for home management after LVAD implantation enables the sharing of daily information between patients and care providers.
Footnotes
Acknowledgment
The authors would like to thank Saori Sawada and Satoshi Sasayama for their application design, and also Takashi Igarashi for data acquisition support.
Financial support: This research was partly supported by The Center of Innovation Program from MEXT and JST, JSPS Grant-in-Aid for Scientific Research (C) (KAKENHI), and a grant from a Kyoto-Funding for innovation in Health-related R&D Fields.
Conflict of interest: None of the authors declare any conflict of interest. Meeting presentation: The Abstract of this manuscript was presented at the 42nd Congress of the European Society for Artificial Organs, Leuven, Belgium, September 2–5, 2015.
