Abstract
Purpose
Acute right ventricular failure is a life-threatening condition with poor prognosis. It occurs as a result of right ventricular infarction, postcardiac transplantation, or postimplantation of a left ventricular assist device. Temporary mechanical right ventricular support could be a reasonable treatment option. Therefore, we developed a novel percutaneously implantable device.
Methods
The PERKAT device consists of a self-expandable chamber covered with multiple inflow valves carrying foils. A flexible outlet tube with a pigtail tip is attached to the distal end. PERKAT is designed for percutaneous implantation through the femoral vein (18 French sheath). The chamber is expanded in the inferior vena cava while the outlet tube bypasses the right heart and the pigtail tip is lying in the pulmonary trunk. An IABP balloon is inserted into the chamber and connected to an IABP console. Balloon deflation generates blood flow from the vena cava into the chamber through the foil valves. During inflation blood is pumped through the tube into the pulmonary arteries.
Results
Conclusions
The novel percutaneously implantable right ventricular assist device offers emergency support of up to 3 l/min. Based on the successful
Keywords
Introduction
Right ventricular failure (RVF) is a clinically unsolved problem. Neither inotropic drugs nor the use of intra-aortic balloon counter pulsation (IABP) are capable of reducing the extremely high rate of morbidity and mortality. RVF due to cardiogenic shock after right ventricular (RV) infarction is associated with a 1-month mortality rate of up to 50% (1). RVF occurs as a consequence of left-sided heart failure (2), after heart transplantation (3), during circulatory support with left ventricular assist systems (4), due to pulmonary hypertension (5, 6), after cardiothoracic surgery (7), or as a result of acute pulmonary hypertension, e.g., after pulmonary embolism (8).
Current therapeutic concepts of RVF treatment include inotropic support to improve RV contractility, volume infusion to maintain RV preload, and pulmonary vasodilatation to reduce RV afterload (9). In cases of profound RVF, refractory to medical treatment, surgical options such as heart transplantation, right ventricle exclusion strategies (10, 11), or implantation of RV assist devices may help to diminish the substantial mortality rate. Surgically implantable assist devices possess the risk of infection (12), thromboembolic events (13), hemolysis, and disseminated intravascular coagulation (14).
However, there is currently a lack of rapid, implantable, percutaneous options for mechanical circulatory support in patients with RVF. Recently, emergency devices such as the Tandem Heart (Cardiac Assist, Pittsburgh, PA, USA) or the Impella RP (Abiomed Impella, Danvers, MA, USA) have been applied in first cases of acute mechanical support in the setting of RVF (15, 16). Nevertheless these systems have specific limitations.
Thus, we developed a pulsatile device for rapid percutaneous implantation specifically supporting the RV without the disadvantages of the continuous flow systems mentioned above. This novel device was named PERKAT (PERcutaneous KATheterpump).
Methods
Description of Device and Implantation Procedure
The PERKAT device consists of a nitinol stent cage that is covered by flexible membranes containing numerous foil valves forming together the foil valve concept (Fig. 1). The foil membranes consist of a specific thermoplastic polyurethane. Its durability has been tested in
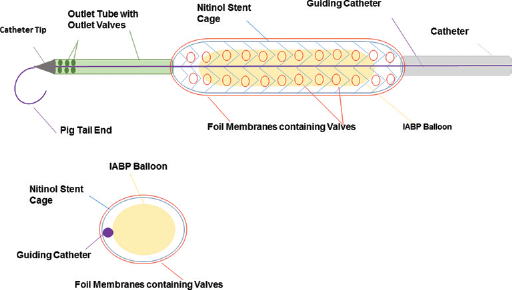
Longitudinal and cross section schematic drawing of PERKAT with inserted IABP balloon.

Unfolded device with standard IABP Balloon below.

Picture showing close-up view of the foil valves.
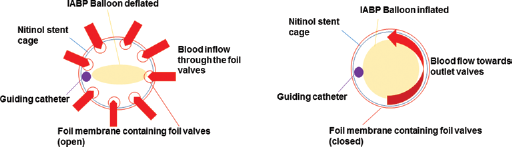
Cross-sectional scheme of PERKAT during IABP balloon deflation (left) and inflation (right).
The PERKAT device should be implanted using fluoroscopy guidance. The nitinol stent cage has to be placed in the inferior vena cava, while the flexible outlet tube bypasses the right atrium and ventricle and the pigtail tip is placed in the pulmonary trunk (Fig. 5).
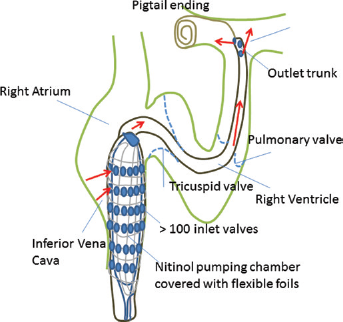
Positioning of PERKAT.
In Vitro Testing
The PERKAT device was placed in the lower reservoir with prespecified water levels simulating the preload of the inferior vena cava (Fig. 6). The flexible outflow tube of the PERKAT device was mounted via a custom-made connector to the higher reservoir for accurate fitting of the outflow tube and to prevent any leakage. This higher reservoir representing the pulmonary trunk was positioned at 2 defined levels above the first water reservoir to simulate various afterload situations (30 cm = 22 mmHg and 60 cm = 44 mmHg). This was chosen to simulate several degrees of pulmonary hypertension in RHF. The PERKAT device was connected to a standard intra-aortic balloon pump (IABP) console (Arrow KAAT II Plus; Teleflex, Morrisville, NC, USA) via the helium drive line. After heating up the water in the lower reservoir to 37°C, the IABP drive unit was started. After the initiation process the experimental setup was ready. The purpose was to measure the total pumped weight of the medium (water) via a scale in a specific period of time (60 s). We measured the flow while the PERKAT system pumped at a constant beats per min (bpm) rate, thus avoiding start and stop phases of the IABP drive unit and to obtain the true flow rate associated with a specific bpm rate. Afterwards the flow rate of the PERKAT system was calculated precisely in liters per min taking into account the density and temperature of the medium. We tested the device in this setting with 2 different standard IABP balloons (Maquet 40 mL-Sensation Plus, Maquet and Arrow 30 mL, Teleflex) and with varying inflation/deflation rates of 80, 90, 100, and 110 bpm.

Experimental setup for
Results
Summary of flow rates are listed in Table I. The flow rates were higher when using the 40-mL IABP balloon in comparison to the 30-mL IABP balloon in the 22-mmHg afterload experimental setup. Flow rates were nearly identical with both balloons with inflation/deflation rates of 90 bpm or 100 bpm when applying 44 mmHg afterload. In contrast, an inflation/deflation rate of 80 bpm led to higher flow rates with the 40-mL balloon, while at 110 bpm the performance of the 30 mL balloon was slightly higher. Increasing the afterload to 44 mmHg resulted in lower flow rates for both balloons in comparison to an afterload of 22 mmHg. We found a linear increase in flow rate with higher inflation/deflation rates at an afterload setting of 22 mmHg. The maximum flow rate reached 3.11 l/min using the 40-mL balloon in the 22-mmHg afterload setting at 110 bpm.

Flow rate dependency from inflation/deflation rate, pre-specified afterload, and balloon size.
Discussion
For the first time, we report the technical details of PERKAT, a novel catheter-based device for pulsatile mechanical RV support. Our initial,
PERKAT consists of a nitinol stent covered by special foils containing numerous valves. The novel foil valve concept of PERKAT and the interaction with the already established IABP balloon and conventional drive unit allows easy handling and rapid implantation. We do not expect any problems during implementation process in the clinical routine, since the IABP systems are well established in catheterization laboratories worldwide. Furthermore, percutaneous application of the circulatory support device without the need for open-heart surgery provides the opportunity for early intervention in the cascade of refractory right ventricular failure. Only a few other RV assist devices for percutaneous implantation have recently reported their first use in humans, such as the Impella RP (16) and the TANDEM Heart RV system (15). In contrast to these RV assist devices, the PERKAT system can be implanted through an 18-F sheath while the TANDEM Heart system requires two 21-F cannulas (17) and the Impella RP needs a 23-F access (18). The PERKAT system is completely percutaneously implantable using the femoral vein without the need for an additional arterial access. Since it only uses 1 femoral vein for implantation, the other groin can be used for catheterization, hemodynamic monitoring, or additional implantation of a left heart assist device like the Impella CP. The small diameter and use of a standard sheath allows rapid implantation of the PERKAT system. Thus, early reversal of acute RVF may help to prevent possible multiorgan failure due to venous overload. Percutaneous implantation reduces the risk of bleeding complications in comparison to surgically implantable devices (17). Assist devices based on a microaxial approach like the Impella RP may generate significant hemolysis (19). Kapur et al described elevated sodium levels and decreased hemoglobin and platelet counts after implantation of continuous flow devices (20). We plan to further evaluate this issue in animal experiments.
Study Limitations
The data obtained in
Conclusions
We have described the development and design of a novel, percutaneously implantable, and rapidly deployable device for temporary right heart support with sufficient
Footnotes
Conflict of interest: Prof. Dr. M. W. Ferrari is cofounder and a shareholder in Novapump. None of the other authors have any financial interest in the devices described in this paper.
Meeting presentation: This manuscript was presented in poster form at the 81st annual meeting of the German Society of Cardiology (DGK) in Mannheim on April 11, 2015.
