Abstract
A cost analysis model was developed to evaluate the 3-year cost of treating vision impairment associated with chronic diabetic macular oedema (DMO) with either a single fluocinolone acetonide (FAc) implant or with 14 ranibizumab injections in the National Health Service (NHS) in England. The model accounts for the overall direct cost of treatment in both pseudophakic and phakic eyes including the cost of the drugs, the cost of administering the drugs, the cost of monitoring the patient, the cost of additional interventions required, and the cost of managing adverse events.
The model indicates a considerable cost saving with the FAc implant irrespective of lens status even allowing for the additional cost of cataract extraction surgery in the majority of steroid-treated phakic patients. Using NHS list prices, the total treatment cost over a 3-year period for one eye is calculated to be £14,273 with multiple ranibizumab injections, £8205 with an FAc implant in a pseudophakic eye and £8932 with an FAc implant in a phakic eye – resulting in an overall cost saving with the implant of £6068 per pseudophakic eye and £5341 per phakic eye. The FAc implant remains the dominant treatment when up to 60% of patients receive the FAc implant and at least 40% of patients receive 14 ranibizumab injections.
The results presented here indicate that 3 years of treatment with a single FAc implant offers a considerable cost saving over 3 years of treatment with ranibizumab (assumed to comprise 14 injections) in both phakic and pseudophakic eyes.
Keywords
Introduction
Diabetic macular oedema (DMO) is a condition where blood vessels in the eye leak fluid or protein onto the central part of the retina (the macula) causing swelling (oedema). It is the most common eye condition in patients with diabetes and, without timely treatment, can lead to loss of vision. Indeed, in the absence of treatment, more than half of those who develop DMO will lose at least 2 lines of visual acuity within 2 years (1). Untreated DMO is one of the leading causes of blindness among individuals of working age in industrialised countries (2). Therefore, aside from the dramatic impact of sight loss on individuals and their families, blindness also imposes a large and costly burden on society because governments will likely incur costs associated with: registering individuals as blind; treating other potential medical sequelae such as depression and broken bones due to falls; and providing low vision aids and rehabilitation, tax allowances, social services, community care, and residential care (3, 4). Where blindness can be prevented through appropriate and timely intervention, a multitude of problems and costs can be avoided. In 2013, an evidence review on the cost of sight loss reported that even just the indirect costs associated with sight loss amounted to more than £5 billion annually in the UK (4). Directing healthcare resources toward preventing blindness can help patients to avoid one of the most feared medical conditions – and maintain their quality of life and economic productivity – while also helping to minimise the enormous but avoidable cost burden associated with helping them cope with such a life-changing chronic condition.
The fluocinolone acetonide (FAc) intravitreal implant (ILUVIEN®, Alimera Sciences Inc., Atlanta, Georgia, USA) is indicated for the treatment of vision impairment associated with chronic DMO that is insufficiently responsive to available therapies (5). The FAc implant is delivered as a single injection into the vitreous of the eye and its duration of action is unique for a DMO treatment – as one implant provides sustained intravitreal release of a micro-dose of the corticosteroid FAc (at a daily dose of 0.2 µg) over 3 years (6). This is in contrast to other therapies approved for DMO that have shorter durations of action and therefore require more frequent injections – including ranibizumab, which inhibits the angiogenic action of vascular endothelial growth factor (VEGF) and which is often injected monthly (7), as well as shorter-acting steroids, which are injected every 3-6 months (8).
Patient anxiety about intravitreal DMO injections is a significant problem – the INCITE survey revealed that 75% of patients are anxious before intravitreal injections, 46% found it hard to think of anything else, and 42% reported that fewer injections would be their most desired improvement to treatment (9). Although anxiety and other factors could negatively impact adherence and lead to missed appointments, a single injection assures optimum compliance over a 3-year period – ensuring continued intravitreal steroid release even if patients miss appointments. Fewer intravitreal injections also translates into greater convenience (for patients, carers and healthcare professionals (9)), fewer physician visits and less pressure on clinics. As some patients are injection phobic and will accept a single injection but refuse multiple injections, less frequent injections may also improve outcomes – as greater patient acceptability can mean the difference between being treated and not being treated. As lack of treatment can lead to blindness, it is imperative that treatments are not so unpalatable that patients avoid them.
Treating DMO with a single FAc implant not only reduces the number of times that a patient will suffer injection anxiety but also helps to minimise the risk of injection-related adverse events – as the risk with a single injection that lasts 3 years is likely to be lower than the risk with multiple injections of a shorter-acting product. Although the frequency of intravitreal injections is often lower for steroids than anti-VEGF products, intravitreal injections of triamcinolone or dexamethasone (short-acting steroids used in DMO though triamcinolone is not licensed in Europe) appear to be associated with a relatively higher incidence of endophthalmitis per injection – 0.13% versus 0.019% with anti-VEGF products (10). Endophthalmitis is an inflammation of the interior of the eye and is a possible complication with all intravitreal injections. It is important to minimise the risk as it can threaten vision. The use of the FAc implant would be anticipated to lower the risk by substantially reducing the number of intravitreal injections required over a 3-year period.
FAc implants are licensed regardless of lens status, so can be used in both phakic eyes (eyes still containing the natural lens) and pseudophakic eyes (eyes where the natural lens has been replaced with an intraocular lens) (5). However, for historical cost reasons, NICE (National Institute for Health and Care Excellence) guidance for Technology Appraisal 301 has so far recommended that their use in England is limited to pseudophakic eyes only (11). As a result, using FAc implants in phakic eyes with DMO is subject to individual funding requests and approval from commissioning groups. NICE guidance excludes phakic eyes, not for reasons of clinical effectiveness [3-year clinical outcomes in patients with DMO are similar regardless of lens status (12, 13)] but because of the perceived disadvantage and additional cost of cataract extraction surgery (as cataracts commonly develop or worsen in phakic eyes after months of intravitreal exposure to any steroid). Nevertheless, optimal treatment of DMO in phakic eyes remains an important unmet need (12) and the overriding clinical priority should be to preserve vision regardless of whether it increases or accelerates the potential need for cataract surgery. Diabetes itself increases the risk of cataracts even without the additional influence of steroid treatment (14, 15) and cataract surgery does not appear to negatively affect long-term outcomes (13). Thus, clinically, it is not rational to avoid steroid treatment because of cataract concerns when sight is threatened by DMO.
In phakic eyes already in need of cataract surgery, steroid treatment may also be the optimal clinical choice. Ideally, DMO should be well controlled before a cataract is extracted (16) and pre-treatment with a steroid can be valuable in achieving such control and minimising the risk of surgery worsening the DMO (17). For this reason, ophthalmologists would often prefer to inject an FAc implant
Importantly, although the NICE guidance does not recommend use of the implant in phakic eyes for cost reasons, it does remind physicians that “the application of the recommendations in this guidance are at the discretion of health professionals and their individual patients and do not override the responsibility of healthcare professionals to make decisions appropriate to the circumstances of the individual patient” (11).
The ophthalmology department at the Royal Hallamshire Hospital (Sheffield, UK) was the first centre in England to explore the cost of treating both phakic and pseudophakic eyes with the FAc implant and to evaluate the appropriateness of the NICE guidance that limits approval of the cost of this treatment to pseudophakic eyes (thereby excluding phakic eyes). We performed a cost analysis to evaluate the overall 3-year cost for one National Health Service (NHS) trust in England of treating phakic eyes and pseudophakic eyes with a single FAc implant compared with multiple ranibizumab injections (the current first-line intravitreal anti-VEGF therapy for DMO and the established standard of care).
Methods
This analysis assessed the cost of treating DMO in both phakic and pseudophakic eyes with either a single FAc implant or multiple ranibizumab injections. As the FAc implant has a lifespan of 3 years, the cost analysis was for a 3-year period and data indicate that patients receiving ranibizumab treatment for 3 years will receive 14 ranibizumab injections over this time (18). Thus, the cost of treating eyes with a single FAc implant was compared with that of 14 ranibizumab injections.
Total costs involved were defined as the sum of: the cost of treatment (i.e., drug costs, injection administration costs, and follow-up costs associated with top-up ranibizumab injections if this is required over the 3-year period for any FAc implant-treated patients); the cost of monitoring (i.e., follow-up appointments); and the cost of adverse event management (i.e., drug treatment costs, surgical treatment costs, and additional appointment costs) (Tab. I).
Breakdown of cost calculations included in the model. The costs for ranibizumab treatment are the same in phakic and pseudophakic eyes
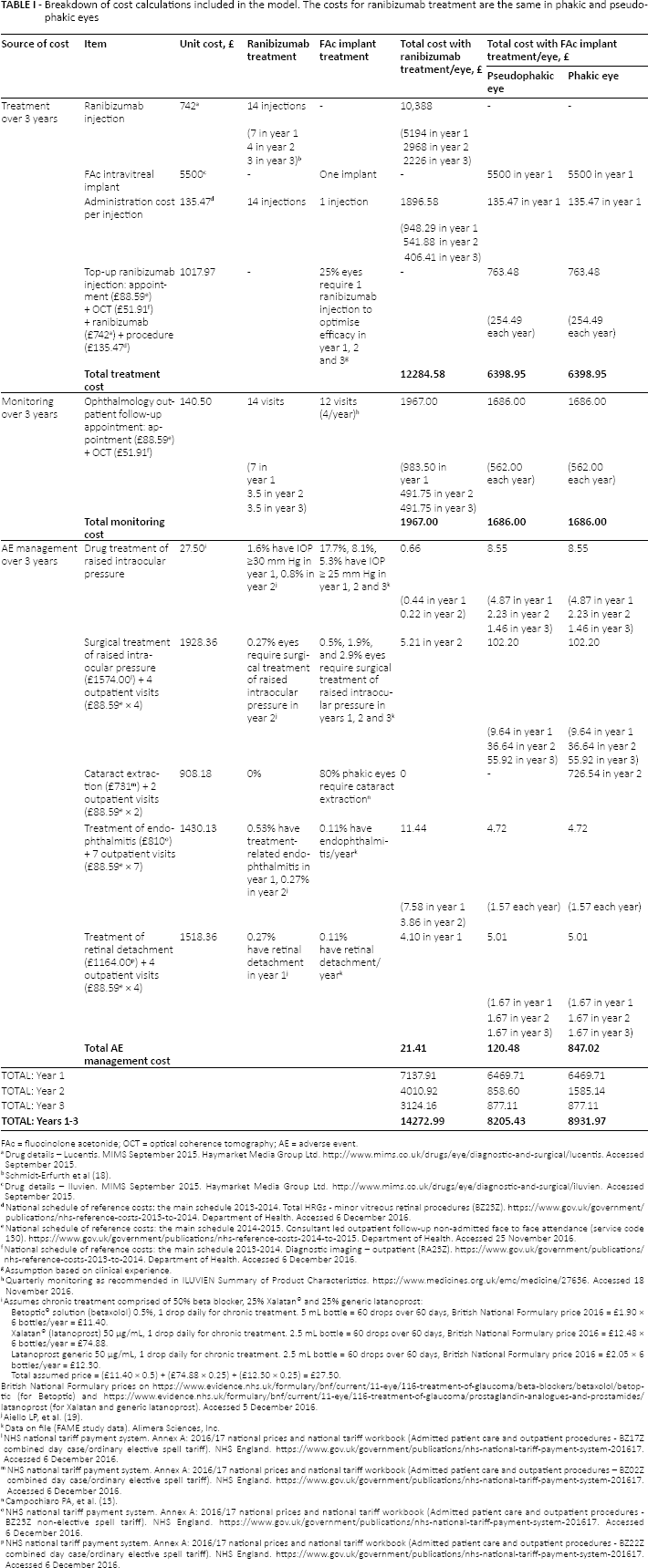
FAc = fluocinolone acetonide; OCT = optical coherence tomography; AE = adverse event.
Drug details – Lucentis. MIMS September 2015. Haymarket Media Group Ltd. http://www.mims.co.uk/drugs/eye/diagnostic-and-surgical/lucentis. Accessed September 2015.
Schmidt-Erfurth et al (18).
Drug details – Iluvien. MIMS September 2015. Haymarket Media Group Ltd. http://www.mims.co.uk/drugs/eye/diagnostic-and-surgical/iluvien. Accessed September 2015.
National schedule of reference costs: the main schedule 2013-2014. Total HRGs - minor vitreous retinal procedures (BZ23Z). https://www.gov.uk/government/publications/nhs-reference-costs-2013-to-2014. Department of Health. Accessed 6 December 2016.
National schedule of reference costs: the main schedule 2014-2015. Consultant led outpatient follow-up non-admitted face to face attendance (service code 130). https://www.gov.uk/government/publications/nhs-reference-costs-2014-to-2015. Department of Health. Accessed 25 November 2016.
National schedule of reference costs: the main schedule 2013-2014. Diagnostic imaging – outpatient (RA23Z). https://www.gov.uk/government/publications/nhs-reference-costs-2013-to-2014. Department of Health. Accessed 6 December 2016.
Assumption based on clinical experience.
Quarterly monitoring as recommended in ILUVIEN Summary of Product Characteristics. https://www.medicines.org.uk/emc/medicine/27636. Accessed 18 November 2016.
Assumes chronic treatment comprised of 50% beta blocker, 25% Xalatan® and 25% generic latanoprost:
Betoptic® solution (betaxolol) 0.5%, 1 drop daily for chronic treatment. 5 mL bottle = 60 drops over 60 days, British National Formulary price 2016 = £1.90 × 6 bottles/year = £11.40.
Xalatan® (latanoprost) 50 µg/mL, 1 drop daily for chronic treatment. 2.5 mL bottle = 60 drops over 60 days, British National Formulary price 2016 = £12.48 × 6 bottles/year = £74.88.
Latanoprost generic 50 µg/mL, 1 drop daily for chronic treatment. 2.5 mL bottle = 60 drops over 60 days, British National Formulary price 2016 = £2.05 × 6 bottles/year = £12.30.
Total assumed price = (£11.40 × 0.5) + (£74.88 × 0.25) + (£12.30 × 0.25) = £27.50.
British National Formulary prices on https://www.evidence.nhs.uk/formulary/bnf/current/11-eye/116-treatment-of-glaucoma/beta-blockers/betaxolol/betoptic (for Betoptic) and https://www.evidence.nhs.uk/formulary/bnf/current/11-eye/116-treatment-of-glaucoma/prostaglandin-analogues-and-prostamides/latanoprost (for Xalatan and generic latanoprost). Accessed 5 December 2016.
Aiello LP,
Data on file (FAME study data). Alimera Sciences, Inc.
NHS national tariff payment system. Annex A: 2016/17 national prices and national tariff workbook (Admitted patient care and outpatient procedures - BZ17Z combined day case/ordinary elective spell tariff). NHS England. https://www.gov.uk/government/publications/nhs-national-tariff-payment-system-201617. Accessed 6 December 2016.
NHS national tariff payment system. Annex A: 2016/17 national prices and national tariff workbook (Admitted patient care and outpatient procedures – BZ02Z combined day case/ordinary elective spell tariff). NHS England. https://www.gov.uk/government/publications/nhs-national-tariff-payment-system-201617. Accessed 6 December 2016.
Campochiaro PA,
NHS national tariff payment system. Annex A: 2016/17 national prices and national tariff workbook (Admitted patient care and outpatient procedures - BZ23Z non-elective spell tariff). NHS England. https://www.gov.uk/government/publications/nhs-national-tariff-payment-system-201617. Accessed 6 December 2016.
NHS national tariff payment system. Annex A: 2016/17 national prices and national tariff workbook (Admitted patient care and outpatient procedures - BZ22Z combined day case/ordinary elective spell tariff). NHS England. https://www.gov.uk/government/publications/nhs-national-tariff-payment-system-201617. Accessed 6 December 2016.
The assumptions used in the cost analysis were derived from published data or real-life clinical experience at the Royal Hallamshire Hospital. The analysis is limited to direct costs typically incurred by the English NHS (except, as further discussed later, it was necessary to use NHS list prices for key drug costs rather than the patient access scheme price available to the NHS as such information is confidential (11)). The analysis was developed as part of a budget impact model and costs are not discounted. The most recent data for costs that were available at the time of writing were used so, as detailed in the footnotes to Table I costs are derived from more than one year. Only direct costs are considered. The analysis does not reflect indirect costs such as those associated with travel or time taken off work by patients and carers to attend appointments. Thus, societal and opportunity costs, and their effect on the overall economic burden, are not calculated.
Key assumptions for ranibizumab treatment
For eyes receiving ranibizumab treatment, the model assumes patients will have 14 ranibizumab injections, and 14 follow-up visits, over a 3-year period (Tab. I). This is based on the mean of 14 ranibizumab injections over a 3-year treatment period reported in the RESTORE extension study (18), which is similar to treatment patterns in real UK clinical practice.
Thus, the total cost of 3 years of ranibizumab treatment is the sum of the cost of purchasing and administering 14 ranibizumab injections, the cost of monitoring the patient (14 follow-up appointments, each with optical coherence tomography [OCT] retinal imaging), and the cost of managing any adverse events (Tab. I). Data from a large multicentre randomised clinical trial (19) indicate that, in any year, up to 1.6% of patients have an increase in intraocular pressure (IOP) ≥30 mm Hg that requires treatment with eye drops, and less than 1% have surgery to reduce raised IOP or develop endophthalmitis or retinal detachment. The cost of managing these is included according to the propensity of each so that, for example, if the cost of eye drop treatment of raised IOP is £27.50 and this is required in 1.6% of patients in the 1st year, it is assumed that the cost per eye is £0.44 (i.e., 1.6% of £27.50) in the 1st year on average (Tab. I).
Key assumptions for FAc implant treatment
The model assumes that a single FAc implant is given and that, as recommended (5), patients have quarterly monitoring visits for 3 years (so 12 monitoring visits in total). Based on clinical experience, it is also assumed that 25% of eyes will receive one top-up ranibizumab injection each year to maintain optimal efficacy.
Thus, the total cost of 3 years of FAc implant treatment is the sum of the cost of purchasing and administering one FAc implant and any top-up ranibizumab injections required to optimise efficacy (it is assumed that 25% of eyes will require one top-up injection each year), the cost of monitoring the patient (12 follow-up appointments, each with OCT retinal imaging), and the cost of managing any adverse events (Tab. I). As reported in phase III clinical trial data (13), it is assumed that 80% of phakic eyes will require cataract surgery in year 2. Also, in both pseudophakic and phakic eyes, it is assumed that in any one year up to 17.7% of patients will have an increase in IOP ≥25 mm Hg that will require treatment with eye drops, less than 3% will require surgery to reduce raised IOP, and less than 1% will develop endophthalmitis or retinal detachment. The assumed incidence of each of these is based on those observed in large multicentre randomised clinical trials (20) and the cost of managing the adverse effects is included according to the propensity of each (Tab. I).
These cost assumptions were used to calculate the average cost of treating a single pseudophakic eye and a single phakic eye over 3 years. These values were then extrapolated to larger populations to allow an estimation of the costs to the local NHS trust of treating patients seen at the Royal Hallamshire Hospital (in total, 73 pseudophakic eyes and 30 phakic eyes). The costs per eye were also extrapolated further to provide an estimate of costs for the DMO population in England.
Results
The cost assumptions in the model, and overall calculated costs per eye, are detailed in Table I. An interactive model is also available from Alimera Sciences that can be tailored to reflect local centre treatment pathways.
Cost over 3 years per eye
As shown in Table I and Figure 1 the anticipated total treatment cost per eye over 3 years of treatment (including product cost, monitoring, additional therapies required and management of adverse events) is £14,273 with ranibizumab, £8205 with FAc in pseudophakic eyes and £8932 with FAc in phakic eyes. Savings arise every year with the implant relative to ranibizumab and are greatest in years 2 and 3 – in years 1, 2 and 3 the anticipated saving per pseudophakic eye is £668, £3152 and £2247, respectively, and the anticipated saving per phakic eye is £668, £2426 and £2247, respectively (Tab. I).
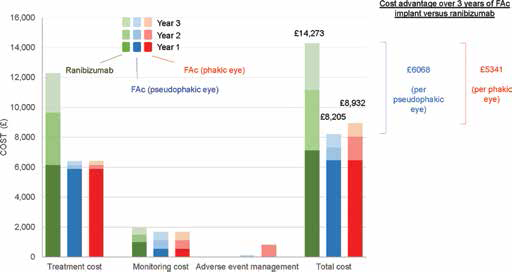
Total cost over 3 years of treating a single eye with either multiple ranibizumab injections or a single fluocinolone acetonide (FAc) implant.
Cost over 3 years per cohort of 73 pseudophakic eyes at Royal Hallamshire Hospital
Using the model, the anticipated overall treatment cost for a series of 73 pseudophakic eyes treated at the Royal Hallamshire Hospital would be £1,041,928 with 14 injections of ranibizumab (73 × £14,272.99) or £598,996 with a single FAc implant (73 × £8205.43). Thus, if the primary treatment in all 73 pseudophakic eyes was the FAc implant rather than 14 injections of ranibizumab, the saving would be £442,932 (in favour of the FAc implant).
A breakpoint analysis illustrating the impact on cost of treating different proportions of patients with either ranibizumab or the FAc implant is shown in Table II. This analysis shows the FAc implant has a cost advantage up to the point where 60% of the patients are treated with the FAc implant and the remaining patients are treated with ranibizumab (i.e., the total group costs associated with treating up to 60% of patients with the FAc implant are lower than the total group costs associated with treating the remaining 40% of patients with ranibizumab). The relative cost advantage is lost when more than 70% of patients are treated with the FAc implant (i.e., the cost of treating more than 70% of patients with the FAc implant is greater than the cost of treating 30% of patients with ranibizumab).
Breakpoint analysis to assess the cost of treating varying proportions of DMO patients with a pseudophakic lens (n = 73) with multiple ranibizumab injections and a single FAc implant
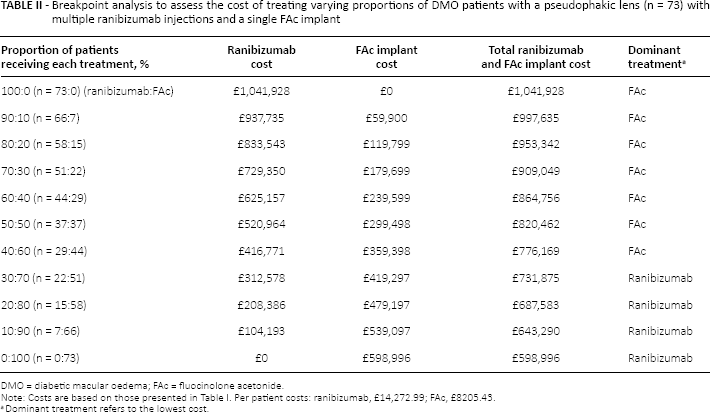
DMO = diabetic macular oedema; FAc = fluocinolone acetonide.
Note: Costs are based on those presented in Table I. Per patient costs: ranibizumab, £14,272.99; FAc, £8205.43.
Dominant treatment refers to the lowest cost.
Cost over 3 years per cohort of 30 phakic eyes at Royal Hallamshire Hospital
At the Royal Hallamshire Hospital, an additional 30 eyes were suitable for FAc therapy but, as they were phakic, they did not fulfil current NICE guidance for funding. However, the model indicates that the anticipated overall cost of treating these 30 eyes would be £428,190 with 14 injections of ranibizumab (30 × £14,272.99), or £267,959 with a single FAc implant (30 × £8931.97). Thus, if the primary treatment in all 30 phakic eyes was an FAc implant rather than 14 injections of ranibizumab, the saving would be £160,231 (in favour of the FAc implant).
A breakpoint analysis illustrating the impact on cost of treating different proportions of patients with either ranibizumab or the FAc implant is shown in Table III. As in the previous breakpoint analysis, this also shows that the FAc implant has a cost advantage up to the point where 60% of the patients are treated with the FAc implant and the remaining patients are treated with ranibizumab (i.e., the total group costs associated with treating up to 60% of patients with the FAc implant are lower than the total group costs associated with treating the remaining 40% of patients with ranibizumab). Again, the relative cost advantage is lost when more than 70% of patients are treated with the FAc implant (i.e., the cost of treating more than 70% of patients with the FAc implant is greater than the cost of treating 30% of patients with ranibizumab).
Breakpoint analysis to assess the cost of treating varying proportions of DMO patients with a phakic lens (n = 30) with multiple ranibizumab injections and a single FAc implant
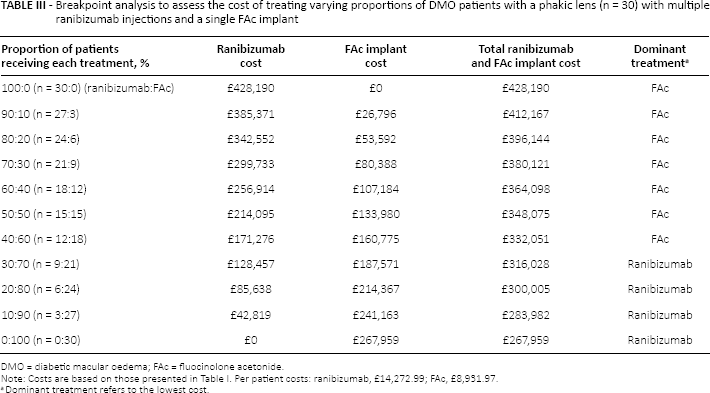
DMO = diabetic macular oedema; FAc = fluocinolone acetonide.
Note: Costs are based on those presented in Table I. Per patient costs: ranibizumab, £14,272.99; FAc, £8,931.97.
Dominant treatment refers to the lowest cost.
Cost over 3 years across the DMO population of England
Extrapolating the cost per eye calculations to a larger population may have inherent inaccuracies, as each treating centre may have a different DMO treatment pathway. However, using the assumptions already described, it is possible to estimate the costs for patients with DMO across the entire population of England.
First, the number of people who could face this treatment choice (i.e., who fit the criteria to receive ranibizumab and the FAc implant) is calculated. For example, England has a population of 46,335,291 individuals aged ≥17 years (21), among whom it is estimated that 6.55% have diabetes mellitus (21), with 2.66% of these having both DMO and visual impairment (22), 26% of these having a central field thickness ≥400 µm (which determines eligibility for ranibizumab funding in the UK) (23), 90% of these having chronic DMO (24), and 39.7% of these having an insufficient response to available therapies (25). This equates to 7500 individuals potentially eligible for ranibizumab treatment who are also potentially eligible for FAc treatment.
If it is assumed that 68% (5100) of these individuals are phakic and 32% (2400) are pseudophakic (as reported in a large ranibizumab trial (19)), then the model shows that the 3-year cost of treating one eye of all 7500 patients would be £107,047,420 with 14 injections of ranibizumab (7500 × £14,272.99) compared to £65,246,079 with a single FAc implant (£19,693,032 for the pseudophakic eyes [2400 × £8205.43] plus £45,553,047 for the phakic eyes [5100 × £8931.97]). The potential saving with the FAc implant is therefore £41.8 million.
Discussion
The model indicates that treatment with a single FAc implant can achieve substantial cost savings relative to multiple injections of ranibizumab in each of the 3 years of the implant's anticipated lifespan (14 ranibizumab injections in total), even after allowing for the additional cost of cataract surgery in a large proportion of phakic eyes treated with the steroid. This cost advantage is largely attributable to the lower drug cost of a single FAc implant relative to 14 doses of ranibizumab (£5500 vs. £10,388). The number of physician visits required for treatment and monitoring is also lower for the FAc implant than for 14 injections of ranibizumab (1 vs. 14 visits for treatment and 12 vs. 14 visits for monitoring, respectively), which not only contributes further to the cost saving but also helps to relieve pressure for clinic appointments. This is of utmost importance because advances in ophthalmology treatment have resulted in a 30% increase in attendances at eye clinics over the last 5 years (26) – and delays in follow-up appointments may result in sight loss.
The analysis described here is limited to a comparison of 3 years of treatment with each drug because the FAc implant has a lifespan of 3 years. However, it is important to note that patients are likely to be switched to the FAc implant at varying times (i.e., not only after 3 years) and that nearly 40% of patients will respond sub-optimally to first-line treatment with ranibizumab (25) and may benefit from an earlier switch to the FAc implant. Data from the DRCR.net Protocol I study indicate that a patient's response to their first 3 ranibizumab injections predicts their response over the next 3 years. Thus, a patient who does not respond well to ranibizumab after 3 injections (does not improve by at least 5 Early Treatment Diabetic Retinopathy Study [ETDRS] letters) is likely to continue not responding well even if injections are continued for 3 years (25, 27). Patients not responding well to ranibizumab after 3 months of treatment (25) may benefit from the anti-inflammatory actions of steroid therapy (as DMO appears to gradually progress from a condition responsive to VEGF inhibition to one with a significant inflammatory component, with aqueous humour levels of IL-1β, IL-6, IL-8, MCP-1 and IP-10 increasing with the severity of DMO (28)). Clinically, it is important to make the switch to steroid therapy promptly as there is a window of opportunity to maximise steroid efficacy before the retina becomes permanently refractory to all treatments. However, the authorities do not define when a switch should occur and its timing is up to individual clinicians and therefore highly variable. Even after 3 years of treatment, patients may continue their existing treatment (i.e., continue with ranibizumab injections or receive another FAc implant) and a switch from ranibizumab to steroid treatment can still happen at any time. As a result, it is rational to analyse the costs over the lifespan of the FAc implant.
The model described here has two potential biases that may favour a relatively lower cost with ranibizumab. First, the incidence of cataract surgery is assumed to be 0% with ranibizumab even though clinical trial data report an incidence of 5.3% in year 1 and 7.2% in year 2 (19). However, because similar incidences were also reported in a control group (19), the cataracts are not likely to have been related to ranibizumab treatment but rather to the diabetes itself (14, 15). By the same token, however, it is also possible that some of the cataracts requiring surgery in the FAc implant-treated eyes may have developed anyway, and so may not have been related to the FAc implant treatment. If this is correct, then the assumed incidence of patients requiring cataract surgery after treatment with the FAc implant may be an overestimate.
The second potential source of bias is that the incidence of patients experiencing a raised IOP is defined as ≥30 mm Hg for ranibizumab and ≥25 mm Hg for the FAc implant when accounting for the cost of eye drops. Although this potentially underestimates the proportion of ranibizumab-treated patients who may be treated for raised IOP, any effect on cost is likely small given the relatively low cost of eye drops to treat raised IOP.
It should be appreciated that the model evaluates the cost of treatment in only one eye per patient and does not take into account any influences on costs of bilateral treatment (although another cost analysis has done this (29)) or re-treatment within the anticipated 3-year period. In the FAME trial, 24% of patients had a second FAc implant within the anticipated 3-year treatment period (13) but no real-world data on re-treatment are available currently.
The model does not reflect costs associated with travel to clinic appointments for patients and carers, nor any costs due to the anxiety often associated with intravitreal injections. Regardless, the model shows that the introduction of treatment with an FAc implant is less costly per eye than treatment with 14 injections of ranibizumab. This is qualitatively consistent with results from another economic evaluation (and, although the 3-year cost saving is quantitatively different in this other evaluation, this is at least partly due to methodological differences) (30).
The NHS list prices used in the model for the FAc implant and ranibizumab reflect the prices paid for the drugs in the event of private treatment. Both drugs are in fact available to the NHS through patient access schemes but, due to commercial confidentiality issues, these values could not be used in our calculations.
A possible opportunity cost for patients who discontinue anti-VEGF therapy and then switch to the FAc implant could be that anti-VEGF therapy may, in time, have elicited further clinical improvement if it had been continued for longer. However, although this might be the case in a small proportion of patients, there is no way to predict in which patients this might occur and, in the meantime, it could be at the cost of preserving existing vision adequately in most patients (25). There is a continuum in DMO, with earlier stages of the disease being more responsive to VEGF inhibition and later stages being more responsive to steroid treatment – which reflects an underlying change in the pathophysiology of the disease from a VEGF-driven disease to a pro-inflammatory disease. If steroid treatment is introduced before the disease progresses to a more pro-inflammatory stage, then anti-VEGF treatment could always be restarted and the presence of the steroid would not be expected to be harmful. In fact, as the DMO gradually progresses toward the pro-inflammatory stage, having a sustained-release steroid implant already in the eye could allow for the earliest possible mitigation of developing inflammation.
Phakic patients treated with the FAc implant are likely to need cataract surgery approximately 1-2 years after treatment (13). Nevertheless, many patients with DMO would eventually need cataract surgery even without FAc implant treatment, so the treatment appears to bring forward the need for surgery rather than necessarily increasing the need for surgery. Some areas of the UK have acceptable waiting times for cataract surgery (time from referral for surgery to undergoing surgery being within the 18-week target in the NHS Constitution) and any increase in the number of surgeries has not caused service capacity problems. Other areas have waiting times of more than 1 year (31) and additional surgeries may inevitably add some extra pressure. In practice, cataract surgery related to FAc implant treatment is likely to represent less than 1% of the current cataract surgery burden in England * and so should not pose a significant problem. In areas with long waiting lists, it may be wise to list patients for cataract surgery some time before their anticipated need, to ensure they receive treatment more promptly once they are in need of surgery.
Data suggest that up to 13,207 phakic patients could be eligible for FAc implant treatment in England [among 46,335,291 individuals aged ≥ 17 years in England (21), it is assumed that 6.55% have diabetes mellitus (21), 2.66% of these have DMO and visual impairment (22), 60.6% of these have chronic DMO (24), 39.7% of these are insufficiently responsive to ranibizumab (25) and 68% of these are phakic (19)]. However, only 23% may receive an FAc implant (32) and, among these, only 80% may require cataract surgery (13), meaning that 2,430 cataract surgeries might be related to FAc implant treatment in addition to the 372,437 other cataract surgeries already undertaken in England each year (33).
Conclusions
Comparing the costs involved over a 3-year period in treating visual impairment associated with chronic DMO with either 14 injections of ranibizumab or a single FAc implant shows that there is a considerable cost saving per eye associated with using the FAc implant. Importantly, this is irrespective of lens status and occurs even after allowing for the additional cost of cataract extraction surgery in the majority of phakic patients. Using NHS list prices, the overall direct cost of treatment per eye over a 3-year period is calculated to be £14,273 for 14 ranibizumab injections, £8205 for an FAc implant in a pseudophakic eye, and £8932 for an FAc implant in a phakic eye – resulting in an overall cost saving with the implant of £6068 per pseudophakic eye and £5341 per phakic eye. These values include the direct costs of the drugs and any additional interventions and the cost of administering the injections, monitoring the patients, and managing adverse events. Breakpoint analyses (in relation to 73 pseudophakic eyes and 30 phakic eyes at the Royal Hallamshire Hospital) show that, in both phakic and pseudophakic eyes, the FAc implant is the dominant treatment when up to 60% of patients receive the FAc implant (i.e., the total group costs associated with treating up to 60% of patients with the FAc implant are lower than the total group costs associated with treating the remaining 40% of patients with ranibizumab).
Once it is evident that an eye is insufficiently responsive to available therapies, there are both clinical and cost reasons for ensuring that treatment with an FAc implant is delivered promptly – and these reasons apply equally to phakic and pseudophakic eyes. Optimal treatment of DMO in phakic eyes remains an important unmet need. The overriding clinical priority should be to preserve vision – regardless of a patient's lens status and regardless of whether it brings forward the need for cataract surgery.
Footnotes
Financial support: Fahd Quhill has attended advisory boards and speaker engagements and has been remunerated for these by Alimera Sciences. Annette Beiderbeck is an employee of, and holds stock options in, Alimera Sciences. We are grateful to Alimera Sciences for funding this manuscript and to Gill Shears, PhD (Write on Target Ltd., UK) for writing assistance.
Conflict of interest: None of the authors has financial interest related to this study to disclose.
