Abstract
The stem cell marker ALDH1 has been of particular interest to scientists since it has been successfully used as a marker to isolate cancer stem cells from breast cancers. However, little is known, especially in Chinese breast cancer patients, on whether ALDH1 enrichment is prevalent in certain subtypes of breast cancer. In this study, we performed flow cytometry and immunohistochemistry to measure the expression of ALDH1 in 10 breast cancer cell lines and in a set of tissue microarrays consisting of 101 breast cancer tissues from the Chinese population. The 101 breast cancer tissues included 4 cancer subtypes defined on bases of their ER, PR, and HER2 statuses: triple-negative (25 cases), luminal A (33 cases), luminal B (16 cases) and HER2-overexpressing (HER2-OE, 27 cases). We found that ALDH1 was expressed in 25 of the 101 cases of breast cancer tissues. When the analysis was stratified, we found that the expression of ALDH1 varied significantly among the 4 subtypes, with a higher expression in triple-negative breast cancer (TNBC, p=0.003) than in the other 3 subtypes. In a series of breast cancer cell lines, we also confirmed that ALDH1 activity was mainly found in TNBC cell lines compared with non-TNBC ones (15.6%±2.45% vs 5.5%±2.58%, p=0.026). These data support the concept that the expression of ALDH1 is higher in TNBC than non-TNBC, which may be clinically meaningful for a better understanding of the poor prognosis of TNBC patients.
Introduction
Human breast cancer is a very complex and heterogeneous disease presenting with highly variable clinical responses to therapy and prognoses. One of the reasons explaining this phenomenon is the existence of cancer stem cells characterized by self-renewal capacities, multi-differentiation potential, and a strong potential of tumorigenicity; these cells are also believed to contribute to tumor metastasis and therapeutic resistance (1–3). Hence, it is of great importance to identify cancer stem cells and clarify their mechanisms of action in breast cancer. Al-Hajj and colleagues were the first group demonstrating that human breast cancers contain a cell population with stem cell properties expressing the cell surface markers CD44+/CD24- (4).
Aldehyde dehydrogenase (ALDH)1 has been reported to be involved in alcohol metabolism (5, 6). The first study linking ALDH1 to cancer showed that ALDH1 was correlated to cancer chemoresistance (7, 8). Until 2007, after a report by Ginestier et al, it was believed that ALDH1 could be a better marker for breast cancer stem cells because of the higher tumorigenic capacity of ALDH1+ tumor cells than that of CD44+/CD24- tumor cells (9). After this, other studies reported that ALDH activity was altered in the sera of patients with different types of cancer, such as pancreatic, colorectal, gastric, liver, and breast cancer (10–15). Recently, the ALDEFLUOR assay and a number of ALDH1-specific antibodies have been successfully developed to identify and isolate cancer stem cells in mammary gland and breast cancers (9). It has also been found that ALDH1+ breast cancer cells play a significant role in the therapeutic resistance and poor prognosis of breast cancers (9, 16–22). These findings opened up a new field of research on breast cancer stem cells and facilitated the translation of cancer stem cell concepts into clinical practice.
Another reason explaining the heterogeneity of breast cancer is the existence of molecular subtypes defined by characteristic molecular signatures determining different prognosis and responses to therapies (23–25). Four subtypes of breast cancer were first identified by Perou through clustering analysis (23). After this, many more independent studies supported the existence of breast cancer intrinsic subtypes (24, 25). Recently, many clinical studies widely used immunohistochemical biomarkers, such as ER, PR, and HER2, as surrogate biomarkers of genes to identify different cancer subtypes. Currently, it is unclear how breast cancer stem cells are linked to distinct subtypes of breast cancer. Despite the fact that some reports suggested that cancer stem cells were enriched in some subtypes of breast cancer, in which ALDH1 correlated with specific clinicopathological parameters (21, 26–28), the issue is still not fully clarified. These data mainly came from the Caucasian population, and can hardly be applied to all ethnicities worldwide. Moreover, there are some discrepancies among the reports of different groups. Thus it is important to further confirm this hypothesis in different populations. In particular, our work has been conducted in the Chinese population.
We analyzed the expression of ALDH1 in breast cancer tissues from the Chinese population by immunohistochemical staining of tissue microarrays, summarizing the clinicopathological features of ALDH1+ breast cancers, as well as the ALDH1 expression profiles in different subtypes of Chinese breast cancers. Eventually, these results were confirmed in breast cancer cell lines including various subtypes.
Materials and Methods
Tissue samples
Tissue microarrays (BR1503, BR962, and T088) were purchased from Xi'an Ailina Biotechnology Co., Ltd., which also provided the data regarding the clinicopathological parameters. All human tissues were collected according to protocols approved by our institutional review board. For all tissues, the diagnosis was reconfirmed by 2 pathologists. Since the HER2 status was not confirmed by fluorescence in situ hybridization (FISH), all the patients who resulted HER2 single-positive or double-positive by immunohistochemistry were excluded from this study. Finally, a total of 101 breast cancer tissues with detailed information were included. The median age of the patients from whom tissues were collected was 49 years (range 28-83 years). Stages of disease at the time of diagnosis ranged from stage 0 to stage B. ER, PR, and HER2 status were assessed by immunohistochemistry. The 101 breast cancer tissues were divided into 4 subtypes according to their ER, PR, and HER2 status: triple-negative breast cancer (TNBC) (ER-/PR-/HER2-; 25 cases), luminal A type (ER+ and/or PR+ and HER2-; 33 cases), luminal B type (ER+ and/or PR+ and HER2 3+; 16 cases), and HER2-overexpressing (HER2-OE) (ER-/PR-/HER2 3+; 27 cases).
Main reagents used in immunohistochemistry
The rabbit polyclonal primary antibody against ALDH1A1 (ab51028; 1:200) was obtained from Abcam plc. (Cambridge, Massachusetts, USA). We then bought the commercially available second-generation general-purpose two-step detection system (Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd., PV9000), and the hematoxylin solution (Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd., ZLI-9039).
Immunohistochemistry staining procedure
After heating at 60°C for 2h, the slides were deparaffinized in xylene and rehydrated in decreasing ethanol dilutions. Tris-EDTA pH 9.0 retrieval buffer was used for heat-induced antigen retrieval. After the buffer was cooled to room temperature, endogenous peroxidase activity was blocked with a buffer containing 3% H2O2 peroxide. Slides were incubated with the ALDH1A1 primary antibody at 4°C overnight. After being incubated with goat anti-rabbit/mouse IgG polymer (Beijing Zhongshan Golden Bridge Biotechnology Co., Ltd., China, K103324A) for 30 minutes at room temperature, the slides were visualized with diaminobenzidine (DAB), then lightly counterstained with hematoxylin, and finally mounted.
Evaluation of the staining
Two pathologists independently evaluated the stainings. The ALDH1 protein was mainly localized in the cytoplasm with a diffuse brownish-yellow distribution. Results were evaluated in terms of percentage (P) and intensity (I) of positive cells, as previously described (19, 29). The quick score (Q; Q = P × I) was divided into 4 grades: Q=0 (negative, -), 0< Q ≤120 (weakly positive, +), 120< Q ≤210 (moderately positive, 2+), 210< Q ≤300 (strongly positive, 3+). By taking the Q value of 120 as the cutoff, the ALDH1+ tumors were divided into the high-expression group (Q >120) and low-expression group (0< Q ≤ 120). In accordance with the related article (19), a tumor presenting at least 1 ALDH1+ cancer cell was considered as an ALDH1+ tumor. The cutoff value was 1% for ER and PR, and 10% for HER2 (30, 31). HER2 status was divided into 2 groups as previously described (30).
Cell culture
The human breast cancer cell lines MCF-7, MDA-MB-231, CAL-51, MDA-MB-435s, MDA-MB-436, BT-20, T47D, ZR-75-1, MDA-MB-453, and BT-474 were obtained from the Beijing Union Medical College Cell Resource Center or from collections developed at the State Key Laboratory of Molecular Oncology. We classified the 10 cell lines into 2 groups according to the data in the literature: TNBC cell lines (ER-/PR-/HER2-; MDA-MB-231, CAL-51, MDA-MB-435s, MDA-MB-436, BT-20) and non-TNBC cell lines (ER+ and/or PR+ and/or HER2+; T47D, BT-474, ZR-75-1, MCF-7, MDA-MB-453) (32–34). These breast cancer cell lines were cultured in specific culture conditions as described by the American type culture collection.
Aldefluor Assay
We used the ALDEFLUOR kit (STEMCELL Technologies Inc., Canada) to analyze the cell population with high ALDH enzymatic activity, according to the manufacturer's instructions. Cells were incubated in ALDEFLUOR assay buffer containing ALDH substrate (BAAA, BODIPY®-aminoacetaldehyde, 1 μmol/L per 1×10 6 cells) for 45 minutes at 37°C. As negative control, a sample of cells was incubated under identical conditions, with 50 mmol/L of diethylaminobenzaldehyde (DEAB), which is a specific ALDH inhibitor. After centrifugation at 250 g for 5 minutes at 4°C, cells were resuspended in ALDEFLUOR assay buffer before being assayed by flow cytometry (BD; Becton, Dickinson and Company). Flow cytometry data were analyzed by BD FACSDiva software V6.1.3 (BD Biosciences). Results were quantified in 3 independent experiments and were used to calculate mean and standard error.
Mammosphere culture
Mammospheres were cultured as reported by Dontu et al (35). Single cells were plated in ultra-low attachment 6-well plates (Corning) or plates coated with 1% agarose in PBS at a density of 20,000 viable cells/mL and 5,000 cells/mL in subsequent passages. Cells were grown in a serum-free mammosphere culture medium (MammoCult, StemCell Technologies) supplemented with MammoCult Proliferation Supplements. Mammospheres were collected after 7-10 days and dissociated both mechanically and enzymatically in Accutase (Millipore, SCR005) for 10 minutes.
Statistical analysis
The SPSS software version 16.0 was used for all statistical analyses. The correlations of the ALDH1 levels with different subtypes of breast cancers and other clinicopathological parameters were assessed by the standard chi-square test or Fisher's exact test. Comparison of the ALDH1 levels between the 2 groups of breast cancer cell lines was performed by the t-test. All tests were two-sided and a p<0.05 was considered statistically significant.
Results
ALDH1 expression was mainly found in ER-negative breast cancer, especially in TNBC
ALDH1 expression rates were 44.0%, 6.1%, 18.8%, and 33.3% in TNBC, luminal A type, luminal B type, and HER2-OE types, respectively (Tab. I). ALDH1+ cancer cells with diffuse brownish-yellow staining were scattered in the cytoplasm in some of the tumor tissues (Fig. 1). The difference in ALDH1 expression among the 4 subtypes was statistically significant (p=0.003). When comparing the ALDH1 levels between TNBC and non-TNBC (luminal A type, luminal B type, and HER2-OE type), the expression of ALDH1 was significantly higher in TNBC (44.0% vs 18.4%; p=0.016).
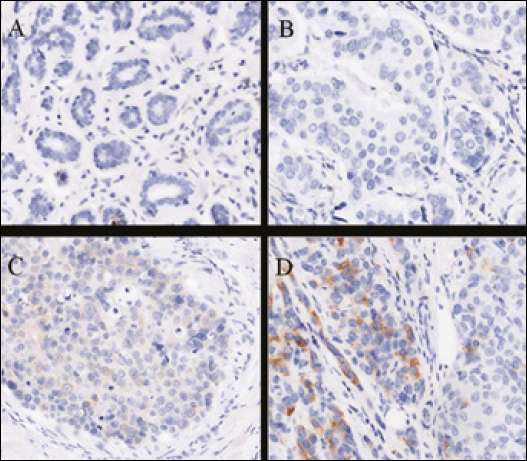
Expression of ALDH1 in normal breast tissue and breast cancer tissues. (×200).The ALDH1 protein was mainly located in the cytoplasm with a diffuse brownish-yellow distribution. A, ALDH1 was negative in normal breast tissue; B, ALDH1 was negative in breast cancer tissue; C-D, low to high expression of ALDH1 in breast cancer tissue according to the quick score.
More ALDH1+ cells were found in ER-negative breast cancers than in the ER-positive ones (36.2% vs 9.3%, p=0.002) (Tab. I). In 58 patients with ER-negative cancer, the rate of ALDH1 expression was still higher in TNBC (11/25, 44.0%) than non-TNBC (10/33, 30.3%), although the difference was not statistically significant due to the limited sample size. However, we did not find any correlation between ALDH1 expression and age, histopathology, tumor size, tumor grade, lymph node status, TNM stage, PR and HER2 status (p>0.05). Taken together, these data indicate that ALDH1 is mainly expressed in ER-negative breast cancer, especially in TNBC.
TNBC cell lines showed a higher ALDH1 activity than non-TNBC cells
To determine whether ALDH1 enrichment in TNBC was a common feature of breast cancer cell lines, we detected ALDH1 activity with the ALDEFLUOR assay in a panel of TNBC and non-TNBC cell lines. Our results showed that ALDH1 was expressed at different levels in the various cell lines, with values ranging from 0.2% to 23.4% (mean and standard error shown are in Fig. 2). The percentage of ALDEFLUOR-positive subpopulations in the TNBC cell lines was higher than that in the non-TNBC group (15.6%±2.45% vs 5.5%±2.58%, p=0.026), indicating that higher levels of ALDH1 activity were mainly found in TNBC cell lines. This result adds further evidence for future studies concerning ALDH1 activity in the TNBC cell model.
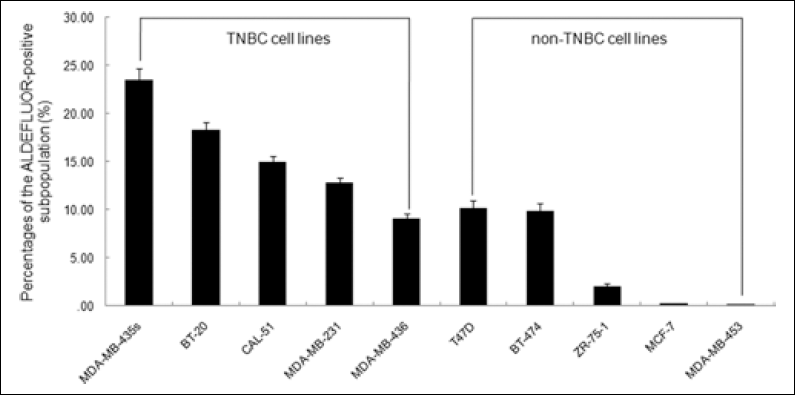
Percentages of the ALDEFLUOR-positive subpopulations in a panel of breast cancer cell lines. TNBC cell lines: MDA-MB-231, CAL-51, MDA-MB-435s, MDA-MB-436, BT-20; non-TNBC cell lines: T47D, BT-474, ZR-75-1, MCF-7, MDA-MB-453.
The ALDH1+ cell population was enriched in mammospheres cultured from the BT-20 cell line
The formation of mammospheres is acharacteristic of breast cancer stem cells in culture. We observed that a small fraction of the BT-20 cells started to form mammospheres 3 days after passage in culture, and grew into spheres larger than 50 μm after 7-10 days. A higher percentage of ALDEFLOUR-positive cells was detected in mammospheres compared with the parental cells (30.2% vs. 18.2%). This observation confirms that the ALDH1 population has cancer stem cell properties.
Discussion
Cancer is now considered as a genetic disease involving multiple genes and signaling pathways. The molecular signature of a specific cancer represents the basis of its clinical and biological behavior. This also applies to breast cancer. Genetic or epigenetic patterns of breast cancer, including ethnical factors, are to be considered when exploring the clinicopathological features of breast cancer. The subdivision of breast cancer into the abovementioned 4 subsets reflects our deeper understanding of breast cancer.
ALDH1 has been indicated to be a cancer stem cell marker in some cancer types, such as adenoid cystic carcinoma (36). But contrasting results have been published in other cancer types, such as malignant melanoma and prostate cancer (37, 38). Therefore, the relevance of ALDH1 in different cancers should be evaluated for each single cancer type.
Keeping in mind that cancer stem cells may be the source of disease development and progression, the main purpose of the present study was to clarify the distribution of ALDH1+ breast cancer cells, which are those with cancer stem cell potentials, in different subtypes of breast cancer in the Chinese population. We found a higher expression level of ALDH1 in ER-negative breast cancers, especially in TNBC. This finding is partly consistent with those of other studies (9, 26, 39), and it suggests that TNBC includes more cells with stem cell potential. The enrichment of ALDH1+ cells is consistent with the clinical behavior of this breast cancer subtype.
Our study shows that the frequency of ALDH1-positivity in breast cancer in the Chinese population is 25/101 (24.8%). This result is similar to the data reported by the groups of Ginestier (9) and Morimoto (39), but lower than those from Nalwoga who reported a positivity rate of 48% in breast cancer in the African population. Besides the discrepancy in the evaluation system, the ethnical variation may play a role in these differences. Therefore, the expression of ALDH1 in breast cancer patients still needs to be further investigated. We found that, in the Chinese population, the majority of breast cancers had a low frequency of ALDH1+ cancer cells, which is in accordance with the concept that cancer stem cells exist in the bulk tumor at a very low percentage. But the fact that ALDH1 was found to be expressed in 17/25 TNBC cases indicated that cancer stem cells may be a common feature in this subtype of breast cancer.
As confirmed also by other studies (9, 26, 39), our finding that the expression of ALDH1 is correlated with the ER-status (p=0.002), suggests that ER- breast cancer cells may contain more stem-cell-like cells and may originate from those breast stem cells which are more likely to have the ER- and ALDH1+ phenotype (40, 41). Furthermore, higher levels of ALDH1 were found in TNBC compared with non-TNBC (p=0.016), thus indicating the presence of a higher proportion of cancer stem cells in this subtype. In accordance with our data, a recent study showed the elevated expression of ALDH1 in BRCA1-related breast cancers, of which 80%-90% are TNBC (42). The enrichment in the ALDH1 population, representing cancer stem cells, explained the biological and clinical behaviors of TNBC, characterized by invasiveness, relapse, and chemoresistance. It has been reported that a high expression of ALDH1 was associated with therapeutic resistance and poor prognosis (16, 18, 19, 21, 43). Thus, our study seems to explain a possible mechanism for the high risk of relapse and poor prognosis of TNBC. However, when comparing the expression rates of ALDH1 in the TNBC and HER2-OE subtypes, no significant difference was observed. This suggests that also the HER2-OE subtype includes more cancer stem cells, a feature that might be associated with the poor prognosis of patients with HER2-overexpression. The expression of ALDH1 in luminal A/B types breast cancers was significantly lower than that in TNBC; a finding that is compatible with the hypothesis that luminal A type breast cancer may originate from ER+ breast progenitor cells rather than from ER- breast stem cells. It is therefore reasonable to speculate that the observed differences in the frequency of ALDH1+ cells among distinct subtypes may indicate subtype-specific tumor origins (27, 39). However, the intrinsic mechanisms validating this hypothesis still need to be further investigated.
Moreover, similarly to what we observed in primary tumors, ALDH1 levels differed greatly between TNBC and non-TNBC breast cancer cell lines. The higher levels of ALDH1 activity found in the TNBC cell lines imply the presence of a higher proportion of cancer stem cells. Thus, our in vitro data also support the evidence from the tissue microarray analysis that ER- breast cancer cells were more likely to be ALDH1+. In addition, we showed a higher percentage of ALDEFLOUR-positive cells in mammospheres cultured from the BT-20 cell line, indicating that mammospheres are enriched with ALDH1+ stem cells in vitro. Hence, as also described in other studies (9, 44), mammospheres' culture was a suitable and practical method to enrich breast cancer stem cells in vitro.
TNBC represents a type of cancer with a relative lack of new treatment protocols. In fact, in TNBC there is no specific target for the currently widely used small molecular inhibitors or antibodies directed against ER or HER2, while these drugs produce a good response in other subtypes of breast cancer. The current study not only suggests that ALDH1+ cells may be a potential therapeutic target for TNBC, but also supports the development of therapies targeting cancer stem cells. In fact, it has been shown that the inhibition of bmi-1 in ALDH1+ cells suppresses tumorigenicity and enhances radiochemosensitivity in head and neck squamous cell cancer (45). Such strategies might be useful in the future management of patients with TNBC.
Conclusions
In conclusion, a higher expression of ALDH1 was observed in TNBC compared to non-TNBC in the Chinese population, suggesting an elevated cancer stem cell potential in TNBC. These findings seem to explain, at least in part, the early relapse and poor prognosis of this subtype of breast cancer. In this study the assessment of ALDH1 expression in different subtypes of breast cancer links the properties of breast cancer stem cells to the biological behaviors and clinical characteristics of the cancer subtypes, and possibly provides a theoretical basis for guiding the development of future therapies targeting breast cancer stem cells.
Authors' contributions
Conceived and designed the experiments: HL FM HQ BX. Performed the experiments: HL FM HW YF XZ. Analyzed the data: HL HQ BX. Contributed reagents/materials/analysis tools: HW CL. Wrote the paper: HL FM HQ BX.
