Abstract
Background
Anthracyclines are among the most active drugs against breast cancer, but can exert cardiotoxic effects eventually resulting in congestive heart failure (CHF). Identifying breast cancer patients at high risk of developing cardiotoxicity after anthracycline therapy would be of value in guiding the use of these agents.
Aims
We determined whether polymorphisms in the renin-angiotensin-aldosterone system (RAAS) and in the glutathione S-transferase (GST) family of phase II detoxification enzymes might be useful predictors of left ventricular ejection fraction (LVEF) kinetics and risk of developing CHF. We sought correlations between the development of cardiotoxicity and gene polymorphisms in 48 patients with early breast cancer treated with adjuvant anthracycline chemotherapy.
Methods
We analyzed the following polymorphisms: p.Met235Thr and p.Thr174Met in angiotensinogen (AGT), Ins/Del in angiotensin-converting enzyme (ACE), A1166C in angiotensin II type-1 receptor (AGTR1A), c.-344T>C in aldosterone synthase (CYP11B2), p.Ile105Val in GSTP1. Additionally, we analyzed the presence or absence of the GSTT1 and GSTP1 genes. A LVEF <50% was detected at least once during the 3 years of follow-up period in 13 out of 48 patients (27.1%).
Conclusion
RAAS gene polymorphisms were not significantly associated with the development of cardiotoxicity. GSTM1 may be useful as a biomarker of higher risk of cardiotoxicity, as demonstrated in our cohort of patients (p=0.147).
Keywords
Introduction
Anthracyclines are effective and widely used agents for the treatment of breast cancer in both adjuvant and metastatic settings (1). Notwithstanding their advantages, their use involves a significant risk of cardiotoxicity that may culminate in congestive heart failure (CHF) syndrome (2). This risk is related to the cumulative dose of anthracycline (3, 4). Moreover, doxorubicin-related CHF risk is schedule-dependent, with lower incidence with schedules based on one administration per week compared to one administration every 3 weeks (3). The development of CHF is more common within the first year after completion of therapy, but is sometimes seen after a longer time interval. A key player in the pathophysiology of CHF is the renin-angiotensin-aldosterone system (RAAS) (5), the main component of which is the angiotensin-converting enzyme (ACE) that converts angiotensinogen (AGT) into angiotensin II. Angiotensin II is the major ligand of the angiotensin II type-1 receptor (AGTR1A), which regulates intravascular volume and blood pressure (6). Finally, in the zona glomerulosa of the adrenal cortex, angiotensin II stimulates the production of aldosterone (7).
Functional polymorphisms in ACE, AGT, and AGTR1 affect expression or function of the encoded protein, and have been associated with stroke, coronary heart disease, renal and vascular dysfunctions, and diabetes (8). Most studies have focused on a number of well-known single-nucleotide polymorphisms (SNPs), including: p.Met235Thr and p.Thr174Met SNPs within exon 2 of the AGT gene, an insertion/deletion (Ins/Del) polymorphism involving 287 base-pairs (bp) in intron 16 of the ACE gene, and the transversion A1166C SNP in the 3′-untranslated part of the AGTR1A gene (9, 10). Another RAAS polymorphic gene encodes the aldosterone synthase (CYP11B2 or ALDOS), a member of the cytochrome P450 family with steroid 11b-hydroxylase, 18-hydroxylase, and 18-oxidase activities required for aldosterone biosynthesis (11). A common polymorphism exists in the promoter region of the CYP11B2 gene at position c.-344C>T, which overlaps with the putative binding site for the steroidogenic transcription factor SF-1 (12). Increasing evidence has suggested an important role for drug-metabolizing enzymes such as the glutathione S-transferase (GST) family in determining inter-individual variations in the response to chemotherapeutic agents (13, 14). GSTP1, GSTM1 and GSTT1 genes encode the most important phase II detoxifying proteins involved in the conjugation of substrates that are toxic to cancer cells, including chemotherapeutic agents used in breast cancer treatment such as anthracyclines (15–17). We investigated whether the status of polymorphisms in RAAS and GST enzymes influences the kinetics of the plasma levels of troponin I, brain natriuretic peptide (BNP) and other blood markers during the course of anthracycline chemotherapy, following up on our previous works (18, 19). Our primary goal was to test whether patients developing anthracycline cardiotoxicity have a different RAAS and GST enzyme genetic makeup in comparison to treated patients who did not manifest an impairment in cardiac function.
Materials and Methods
Patients
Patients with early breast cancer appropriate for adjuvant chemotherapy were included in a prospective evaluation of cardiac function and have been described elsewhere (18, 19; Tab. I).
Summary of The Characteristics at Baseline (T0) of the 48 Patients
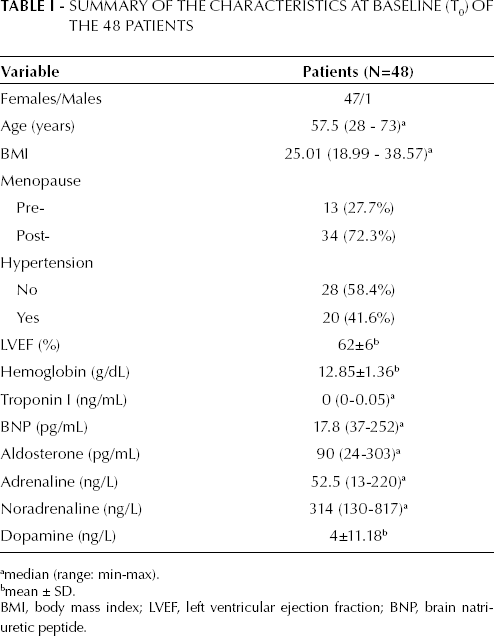
BMI, body mass index; LVEF, left ventricular ejection fraction; BNP, brain natriuretic peptide.
median (range: min-max).
mean ± SD.
Chemotherapy consisted of 6 cycles of FEC (cyclophosphamide 600 mg/m2, epirubicin 90 mg/m2 and fluorouracil 600 mg/m2) given intravenously on day 1 every 21 days. Loco-regional radiotherapy was delivered at the end of the chemotherapy plan, if clinically indicated. All patients that had hypertension (20/48; 41.6%) were treated with the standard anti-hypertensive therapy.
Specimen characteristics
For DNA genotyping analysis, a 5 mL venous blood sample was drawn into EDTA-containing tubes and stored at -20°C prior to DNA extraction using the EZ1 DNA blood kit (QIAGEN, Hilden, Germany). For analysis of blood biomarkers either serum, plasma or complete blood samples were collected by venopuncture and, depending on specific (typical) markers, stored at -20°C or analyzed immediately (19).
Assay methods
We examined the AGT p.Met235Thr (rs699), AGT p.Thr174Met (rs4762), ACE Ins/Del (rs4340), CYP11B2 c.-344T>C (rs1799998), AGTR1A A1166C (rs5186), GSTT1/GSTT1 “null”, GSTM1/GSTM1 “null”, GSTP1 p.Ile105Val (rs1695) and TP53 p.Pro72Arg (rs1042522) SNPs. Allelic discrimination was performed for AGT p.Met235Thr (c.803 T>C) and AGTR1A (c.∗86A>C A1166C) SNPs, using the Applera SNP-Assay C_1985481_20 and C_3187716_10, respectively. Reaction solutions were prepared by adding 12.5 μL of TaqMan Gene Expression Master Mix (Applied Biosystems Foster City, CA, USA), 1.25 μL of the TaqMan Pre-Designed SNP Genotyping Assay specific to each polymorphism, and 9.25 μL of distilled water to 100 ng of DNA sample. Amplification and analysis were done according to the manufacturer's protocol in 96-well plates in the ABI PRISM 7000 Sequence Detection System. Amplifications for ACE Ins/Del, AGT p.The174Met (c.620 C>T), CYP11B2 (c.-344C>T), GSTP1 p.Ile105Val (c.313 A>G), GSTT1/GSTT1 “null”, GSTM1/GSTM1 “null” and TP53 p.Pro72Arg were performed with 0.5 units of AmpliTaq Gold DNA polymerase (Applied Biosystems), in a 10 μL volume reaction mixture, containing 0.75 μL of a 5 μM solution of both primers, 200 μM deoxynucleotide triphosphate (Invitrogen Carlsbad, CA, USA), 1.5 mM MgCl2 and 50 ng of DNA sample. Amplification conditions included a 10 minutes “hot start” at 96°C, followed by 35 cycles at 96°C for 30 seconds, 30 seconds at the specific annealing temperature for each fragment, and 40 seconds at 72°C, with a final 10 minutes extension at 72°C. All reactions were carried out in AB Applied Biosystems model 9700 thermal cyclers. Amplicons for AGT p.Thr174Met, CYP11B2 (c.-344 T>C), and GSTP1 p.Ile105Val were sequenced on both strands with a 3130 Avant Genetic Analyzer (AB Applied Biosystems) using 3.1 BigDye (Applera) terminator sequencing chemistry. The amplified products of ACE Ins/Del, GSTT1/GSTT1 “null” and GSTM1/GSTM1 “null” were separated on 2% ethidium bromide-stained agarose gels and visualized using a UV trans-illuminator. For GSTM1 and GSTT1, the presence of the wild-type or null alleles was assessed by co-amplifying a fragment of a constitutive gene (TP53) as a positive control. Primer sequences and annealing temperatures for PCR reactions are available as Supplementary Information.
The blood biomarker assays have been previously described elsewhere (19).
Study design
All patients underwent radical surgery i.e. mastectomy or breast-conserving treatment, having no evidence of distant metastases and adequate bone marrow, renal and hepatic function. Before starting chemotherapy (t0), all patients underwent a cardiac assessment consisting of ECG, LVEF measurement at rest by multigated radionuclide angiocardiography (MUGA Scan), and measurement of the following plasma molecules: troponin I, brain natriuretic peptide (BNP), aldosterone, adrenaline, noradrenaline, dopamine, endothelin 1, big endothelin, and tumor necrosis factor-alpha (TNFα) (19). The presence of coronary artery disease, valvular heart disease and pre-treatment of left ventricular dysfunction were considered exclusion criteria. Arterial blood pressure was measured by sphygmomanometer after a few minutes of rest in a sitting position, and hypertension was defined as systolic blood pressure (SBP) ≥140 mmHg, or/and diastolic blood pressure (DBP) ≥90 mmHg. All the above measurements were repeated at 1 month (t1), 1 year (t2), 2 (t3), and 3 (t4) years after the end of the chemotherapy plan, together with a complete clinical examination performed by a cardiologist.
A cardiotoxic event was defined as the development of overt CHF (grade III) or a decline of left ventricular ejection fraction (LEVF) below 50% (grade II) at any time point during the 3-year follow-up according to the National Cancer Institute Common Toxicity Criteria (version 2.0) commonly used. In 7 cases (14.6%) LVEF could not be measured at each time point (t0-t4) during the follow-up period (18, 19). An informed consent was obtained from each patient enrolled in the study.
Statistical analysis methods
The association between RAAS and GST polymorphisms and anthracycline cardiotoxicity was analyzed using the Fisher's exact test or Pearson's χ 2 test when appropriate. All analyses were performed with Stata/SE 11.1. Hardy-Weinberg equilibrium was evaluated using the χ 2 test. A p value <0.05 was considered statistically significant.
Results
Data
In our cohort of 48 patients (47 females, 1 male) a value of LVEF <50% was detected at least once during the t1-t4 interval in 13 out of 48 patients (27.1%) and overt CHF was diagnosed in 2 of these 13. Patients with at least one LVEF <50% determination (grade II of toxicity criteria) or clinical evidence of CHF (grade III of toxicity criteria) during the entire follow-up period were classified as group 1, while the absence of left ventricular systolic dysfunction was determined in 35 (72.9%) subjects (group 2). Age, body mass index (BMI), basal hemoglobin levels, white blood cell and platelet counts, basal plasma or serum levels of troponin I, BNP, adrenaline, noradrenaline, dopamine, endothelin, big endothelin, and TNFα were not significantly associated with cardiotoxicity. Instead, we found that diastolic blood pressure (DBP) and serum aldosterone (sAldo) levels at the beginning of anthracycline chemotherapy were significantly higher in group 1 (p=0.023 and p=0.032, respectively) (Tab. II).
Characteristics of the Study Population Shown According to the 2 Cohorts of Patients
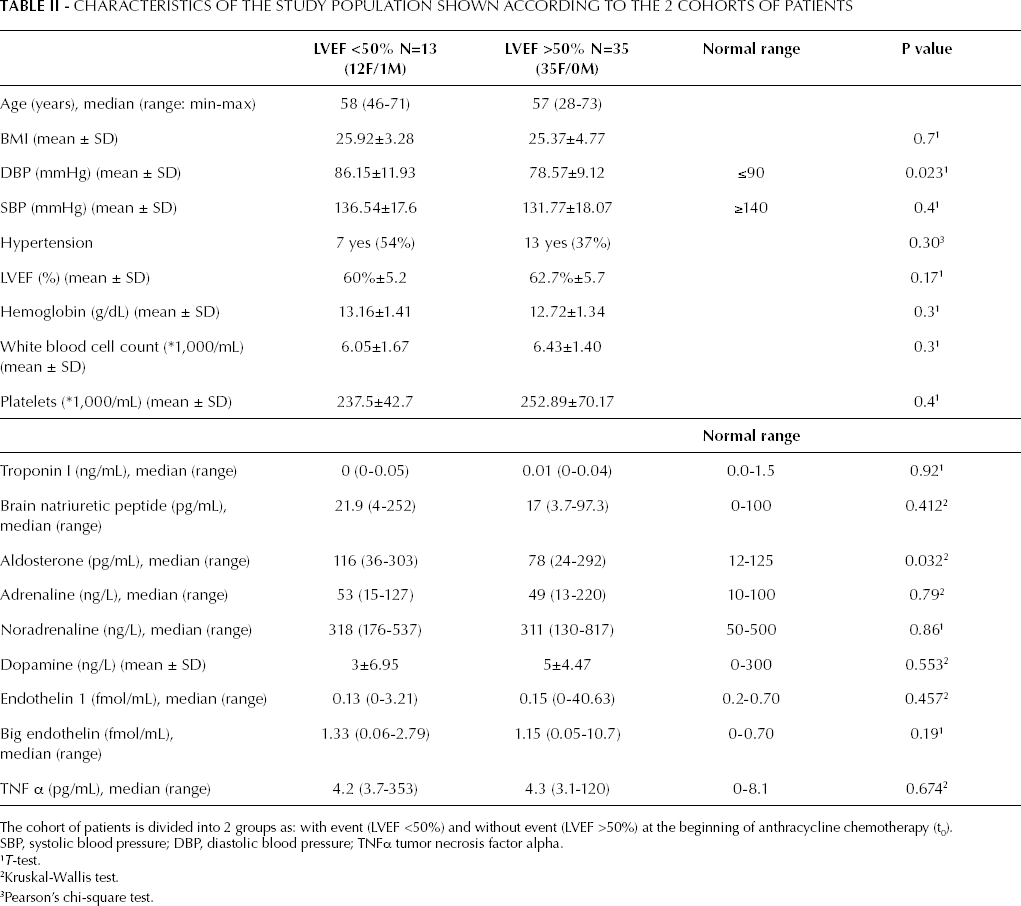
SBP, systolic blood pressure; DBP, diastolic blood pressure; TNFα tumor necrosis factor alpha.
The cohort of patients is divided into 2 groups as: with event (LVEF <50%) and without event (LVEF >50%) at the beginning of anthracycline chemotherapy (t0).
T-test.
Kruskal-Wallis test.
Pearson's chi-square test.
Hypertension was diagnosed in 6 (54%) and 13 (37%) patients, respectively in groups 1 and 2 (p=0.54 Pearson's chi-square test). All patients with high blood pressure were treated with the aim of obtaining a systolic blood pressure <140 mmHg and a diastolic pressure <90 mmHg.
Analysis and presentation
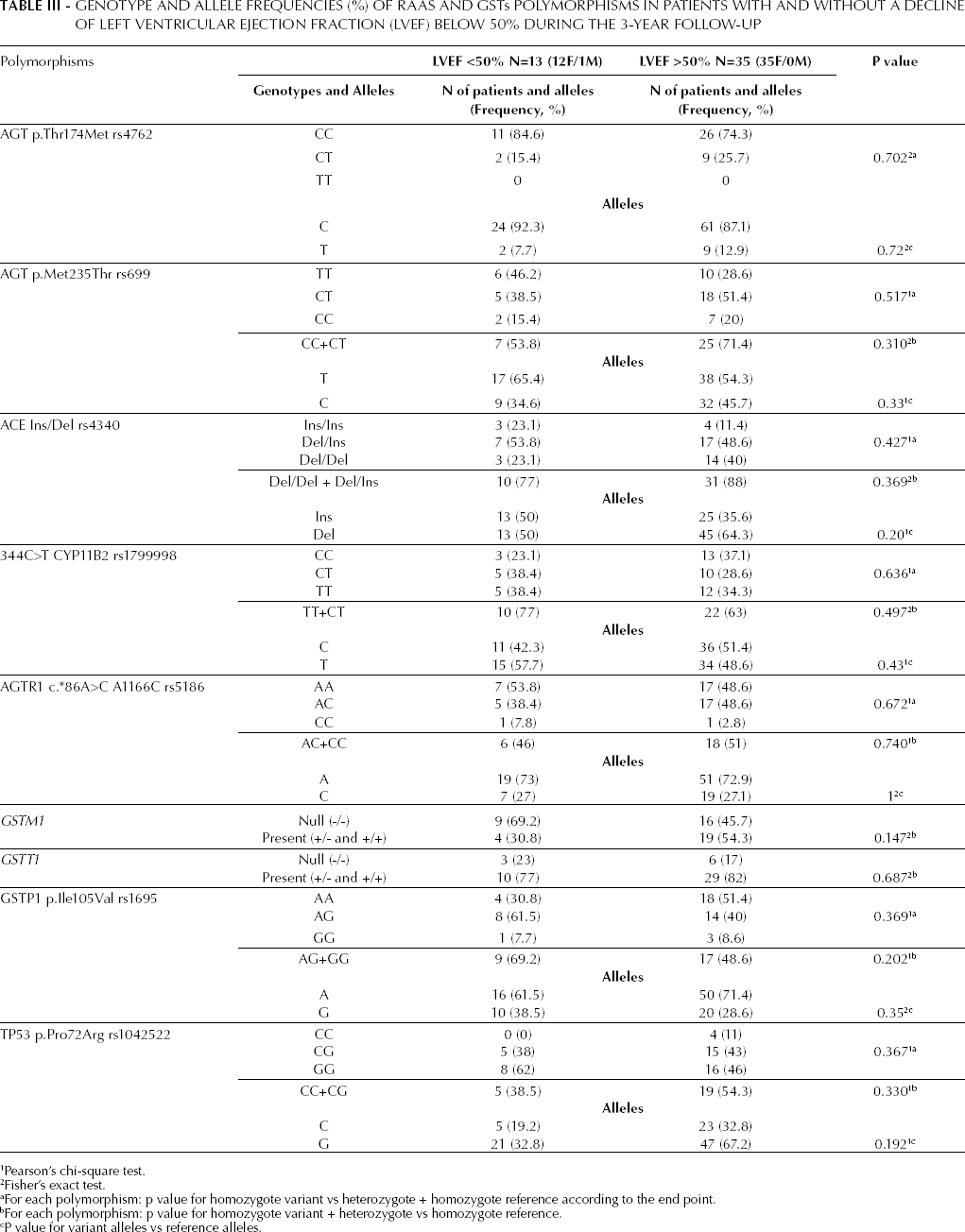
The genotypic makeup and allele frequencies of the patients included in this study are summarized in Table III.
Genotype and Allele Frequencies (%) of RAAS and GSTs Polymorphisms in Patients with and without a Decline of Left Ventricular Ejection Fraction (LVEF) below 50% during the 3-year follow-up
Pearson's chi-square test.
Fisher's exact test.
For each polymorphism: p value for homozygote variant vs heterozygote + homozygote reference according to the end point.
For each polymorphism: p value for homozygote variant + heterozygote vs homozygote reference.
P value for variant alleles vs reference alleles.
The observed genotype distributions were consistent with the Hardy-Weinberg equilibrium in all SNPs analyzed, except for c.-344C>T CYP11B2 rs1799998 (allele frequency T/C = 0.51/0.49, expected genotype frequen cies % TT/TC/CC = 26/50/24, observed 35/31/34, chi-square expected vs observed, p<0.01). The allelic frequencies of all analyzed SNPs did not significantly deviate from those reported for Caucasian individuals in the NCBI dbSNP Short Genetics Variations database (http://www.ncbi.nlm.nih.gov/projects/SNP). Overall allelic frequencies and genotype distributions did not significantly differ between group 1 and 2. However, we observed a trend towards a higher risk of cardiotoxicity in patients negative for the GSTM1 gene (p=0.147).
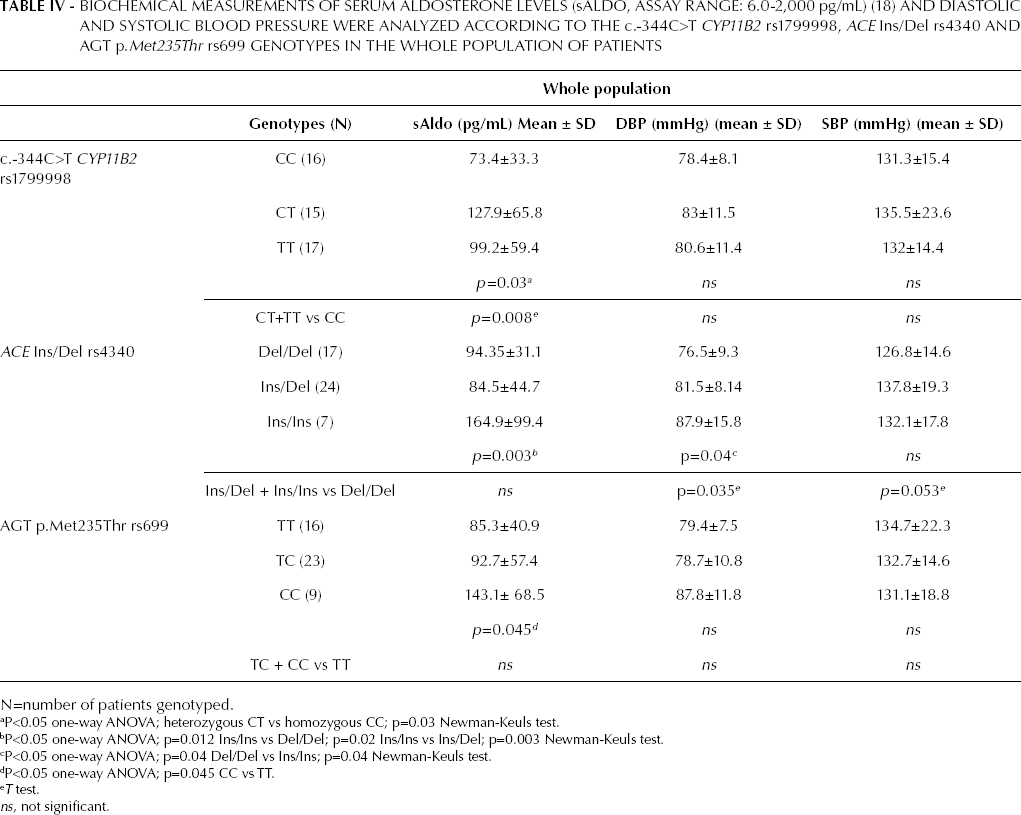
Although we were unable to assess the association of individual RAAS and GSTs gene polymorphisms with the risk of cardiotoxicity, due to the small size of our cohort, a correlation with blood biomarkers was evaluated considering all patients together. The analysis of the sAldo levels at the beginning of the therapy (t0) demonstrated a significant difference (p=0.03) for the 3 genotypic conditions of CYP11B2 SNP c.-344C>T (rs1799998), with the highest levels for the CT genotype subset, intermediate levels for the TT subset and the lowest level for the CC homozygote subsets (p=0.03).
We also observed an association between the ACE Ins-allele SNP (rs4340) and higher values of DBP as well as sAldo levels (p=0.04 and p=0.003, respectively). Finally, we found an association between the AGT p.Met235Thr SNP (rs699) and sAldo levels, being higher in the CC genotype subset than in the CT or TT subsets (p=0.045) (Tab. IV).
Biochemical Measurements of Serum Aldosterone Levels (sALDO, Assay Range: 6.0-2,000 pg/mL) (18) and Diastolic and Systolic Blood Pressure were Analyzed According to the c.-344C>T CYP11B2 rs1799998, ACE Ins/Del rs4340 and AGT p.Met235Thr rs699 Genotypes in the whole Population of Patients
N=number of patients genotyped.
P<0.05 one-way ANOVA; heterozygous CT vs homozygous CC; p=0.03 Newman-Keuls test.
P<0.05 one-way ANOVA; p=0.012 Ins/Ins vs Del/Del; p=0.02 Ins/Ins vs Ins/Del; p=0.003 Newman-Keuls test.
P<0.05 one-way ANOVA; p=0.04 Del/Del vs Ins/Ins; p=0.04 Newman-Keuls test.
P<0.05 one-way ANOVA; p=0.045 CC vs TT.
T test.
ns, not significant.
This study failed to demonstrate a correlation between AGTR1 A1166C or AGT p.Thr174Met genotypes and aldosterone levels or blood pressure. Incidentally, when we analyzed the Pro72Arg (rs1042522) polymorphism on the TP53 gene, we found that patients with the G/C (Pro72Arg) (42%) or G/G (Arg72Arg) (50%) genotypes showed a trend in favor of a higher prevalence of hypertension than C/C patients (Pro72Pro) (8%) (p=0.180). When we compared all the G/C or G/G patients to C/C patients, the difference was borderline statistically significant (p=0.077). Lastly, we found significant or borderline significant differences in several biomarkers depending on the Pro72Arg polymorphism status. Interestingly, hemoglobin levels and LVEF seemed to drop and troponin I level seemed to rise faster during chemotherapy in patients carrying the Arg allele.
Discussion
Previous experiences demonstrated that measuring the rate of hemoglobin and troponin I change during anthracycline-based chemotherapy may be a powerful and inexpensive method to identify patients at high risk of developing CHF (18, 19). Following these studies, we here investigated the distribution of RAAS and GSTs polymorphisms in the same cohort of patients in order to find possible associations between genetic makeup and the risk of anthracycline cardiotoxicity in patients with early breast cancer. The RAAS has been shown to play a key role in the regulation of blood pressure, and several polymorphisms in genes belonging to this system have been variously associated with risk of hypertension and/or CHF. On the other hand, GST enzymes are involved in the detoxification of therapeutic drugs, such as anthracyclines, by conjugation with glutathione. It has been proposed that combinations of genetic polymorphisms in these systems may exhibit stronger associations with phenotypes related to cardiac toxicity. Unfortunately, due to the small size of our cohort of patients, we were unable to perform a conclusive multivariate analysis combining all polymorphisms in the RAAS and GST genes analyzed. Instead, we analyzed individual SNPs and compared their frequency in patients experiencing or not anthracycline cardiotoxicity, and we did not find significant differences. However, in agreement with previous reports, we were able to demonstrate a statistically significant association between several polymorphisms and biochemical parameters that might reflect the onset of adverse cardiac effects following therapy with anthracyclines. The correlation between the RAAS AGT p.Met235Thr polymorphism and cardiovascular diseases are under debate: in fact, while some reports concluded for a positive relationship in essential hypertension (20, 21) and myocardial infarction (22), another study did not confirm this statistical correlation (23), whereas few reports confirmed an association between this SNP and plasma angiotensinogen levels (24). The 235Thr allele has been associated with a stepwise increase in the level of circulating angiotensinogen (“gene dose-response”) (20, 21) and with an increase of blood pressure (25). In this study, we describe an association between the 235Thr allele and higher sAldo levels, but no association was found between this SNP and blood pressure. The ACE Ins/Del polymorphism is strongly associated with the levels of circulating ACE enzyme (26), and risk of hypertension (27). It has recently been reported that this association is influenced by gender, with the Ins-allele being correlated with hypertension in females (28, 29).
Our experience confirms these reports by showing a correlation between the ACE Ins-allele and both high blood pressure and sAldo levels. The AGTR1A A1166C polymorphism was found to be significantly associated with coronary disease in Caucasians (30), but not in Asians (31). The functional relevance of the 1166C allele of the AGTR1A gene is less understood: recently, it has been shown how this polymorphism attenuates binding of the micro-RNA 155, leading to lower receptor density in endothelial and vascular smooth muscle cells (25, 32, 33), and modified sAldo concentrations (34). In our study, we failed to detect any significant association of this allele with cardiac disease.
The aldosterone synthase gene CYP11B2 is a major candidate gene for predisposition to hypertension and cardiovascular disease, due to the central role of aldosterone synthase in blood pressure and cardiac function regulation (35). Although several studies have shown a significant association between the c.-344C/T polymorphism and hypertension (36, 37), the functional effects of this SNP remain uncertain. It is believed that the T allele is associated with higher circulating levels of aldosterone and blood pressure, thus influencing the risk of arterial hypertension, cardiac fibrosis and, consequently, both diastolic dysfunction and ventricular remodeling evolution (38, 39). The prevalence of the c.-344C or c.-344T alleles is ethnicity and race-dependent. Genotype distributions in European analyses show heterogeneous data, but there is evidence that the c.-344C allele is more prevalent in the Italian population than the c.-344T allele (in dbSNP Short Genetic Variations HAPMAP-TSI: CC 25%; CT 58%; TT 16%). Although in our cohort of patients the c.-344C/T CYP11B2 SNP was not in Hardy-Weinberg equilibrium, we found that patients developing cardiotoxicity after therapy with anthracyclines had a lower proportion of the CC homozygous genotype (23%) than patients not experiencing cardiotoxicity (37%). In general, the high variability of these polymorphisms across various ethnic groups suggests that ethnicity can make a significant difference in the role of various genes in certain complex traits. The GSTP1, GSTM1 and GSTT1 genes, which belong to the GST family, encode the most important phase II detoxifying proteins involved in the conjugation of substrates that are toxic to cancer cells, including chemotherapeutic agents used in breast cancer treatment such as anthracyclines (40). Several polymorphisms in GST genes result in reduced or no activity of the enzymes. In particular, the GSTM1 and GSTT1 genes can harbor polymorphic deletions, while the polymorphism in GSTP1 (rs1695) results in the amino acid substitution p.Ile105Val, with the Val-containing enzyme having reduced activity. Homozygosis for the GSTM1-null and GSTT1-null genotypes has been found to confer risk for many cancers, including those of the breast (41–43). An improved survival has been observed in patients with GSTM1-null genotype and has been explained by a better response to chemotherapeutic agents related to more effective cell killing, which in turn is related to the absence of a protective effect of the GSTM1 allele (44). This finding is consistent with the biological hypothesis that higher levels of circulating active drug are associated with improved survival (45) and could be associated with increased toxicity, due to the possibility that null individuals are less able to detoxify carcinogens. In particular, the coordinated actions of a variety of different antioxidants and phase 2 enzymes ensure efficient detoxification of oxidative and electrophilic species as well as other reactive metabolites that contribute to the pathogenesis of cardiovascular disease (45). Of interest, anthracyclines cause free radical damage and lipid peroxidation. These events occur readily in cardiac tissue and are mainly responsible for the cardiotoxicity observed with these agents (46). In humans, reduced levels of antioxidants and phase 2 enzymes, like in GSTM1 null genotypes, expose cardiomyocytes to damage induced by oxidative/electrophilic species, like anthracyclines. For the GSTM1 and GSTT1 genes a null-genotype can be found in, respectively, about 50% and 20% of the North-Western European population, as also confirmed by our study. However, in our cohort we observed a trend towards a higher risk of cardiotoxicity for GSTM1-negative patients; although not reaching statistical significance, this trend suggests a possible role of GSTM1 in protecting from anthracycline-induced cardiac disease. This hypothesis deserves future investigation aimed at clarifying this association. Finally, we found no significant association between anthracycline cardiotoxicity and GSTP1 and GSTT1 polymorphisms.
The main limitation of this study is the relatively small size of our cohort, which might have masked some existing weak associations. Data regarding the aldosterone plasma level presented in Table II and IV should be interpreted with caution; in fact, many factors such as plasma renin activity, potassium level, sodium intake or use of antihypertensive drugs might influence this analysis even if they are not included in the scope of this clinical research.
In conclusion, our data indicate that RAAS SNPs are not correlated with anthracycline cardiotoxicity. In view of the impact of anthracycline cardiotoxicity on the quality of life and clinical management of breast cancer patients, it will be important to further continue this line of investigation in a larger cohort of patients, in order to conclusively assess the role of RAAS and GST genetic makeup in risk determination. We are confident that the methods presented in this study may represent a useful contribution towards this goal.
Footnotes
Acknowledgements
We thank Dr Tim Crook (University of Dundee, Scotland, UK) for the revision of the text and editing of the manuscript.
