Abstract
Background:
The standard recommendation for neoadjuvant therapy for human epidermal growth factor receptor-2 (HER2)-positive breast cancer patients is trastuzumab in combination with chemotherapy, but there is no current standard recommendation for appropriate chemotherapy regimens. This meta-analysis evaluated the efficacy and cardiac safety of the concurrent use of anti-HER2 targeted drugs and anthracycline-based neoadjuvant chemotherapy (NAC) for HER2-positive breast cancers.
Methods:
The pooled odds ratio (OR) rate for pathologic complete response (pCR), the pooled hazard ratio (HR) of overall survival (OS), and the left ventricular ejection fraction (LVEF) decline events were all calculated. Differences in efficacy, prognosis, and cardiac safety were compared between patients receiving an anthracycline-containing regimen (AB) and those treated with non-anthracycline-based (nAB) NAC.
Results:
A total of 1366 patients in 4 prospective and 3 retrospective studies were included in the meta-analysis. The pooled OR for pCR rate was 0.73 with a 95% confidence interval (CI) of 0.43 to 1.24 (P = .246). Subgroup analysis of low tumor burden cases showed no improvement in pCR rate for patients in the AB group compared with nAB, with the pooled OR rate being 0.73 with a 95% CI of 0.37 to 1.44 (P= .357). The 3-year OS rate was 95.63% and 95.54% in the AB and nAB groups, respectively, with no statistical difference (P= .157). There was a significant increase in the rate of LVEF decline of 19.07% in the AB group compared with 13.33% for the nAB group, with an HR of 1.62 and a 95% CI of 1.11 to 2.36 (P = .013).
Conclusions:
The addition of anthracyclines did not improve pCR rates and survival after neoadjuvant and the increased cardiotoxicity of anthracyclines further limited their application. This study showed that it was feasible to use anti-HER2 drugs without anthracyclines in neoadjuvant therapy for HER2-positive breast cancer patients.
Introduction
Breast cancer is the most common malignancy in women worldwide and one of the leading causes of cancer-related deaths. 1 About 20% of these patients have concomitant amplification of human epidermal growth factor receptor-2 (HER2) also known as ErbB2, leading to increased malignancy, and in the absence of treatment, HER2 overexpression is associated with a poorer prognosis.2,3 Trastuzumab was first used in 1998 in combination in chemotherapy regimens for metastatic breast cancer, which led to a fundamental change in the treatment of HER2-positive breast cancer. 2 The use of anti-HER2 targeted drugs has become widespread, leading to a significant increase in the overall survival (OS) of patients with HER2-positive breast cancer. There are currently various targeted drug options available for HER2-positive breast cancer, including large-molecule monoclonal antibodies such as trastuzumab and patuximab4,5 and small-molecule tyrosine kinase inhibitors such as lapatinib 6 and pyrotinib, 6 as well as antibody-drug conjugates such as T-DM1 and T-DXd (DS-8201).7,8 These new drugs have demonstrated remarkable efficacy in the treatment of postoperative adjuvant intensive or advanced breast cancer, but trastuzumab remains the preferred choice for neoadjuvant treatment of HER2-positive breast cancer from a medical perspective. While the anti-HER2 targeted drugs may have a concomitant increased risk of cardiotoxicity, 9 trastuzumab has excellent efficacy, so the National Comprehensive Cancer Network guidelines still recommend that patients with HER2-positive breast cancer receive single- or dual-target therapy of trastuzumab combined with pertuzumab in combination with adjuvant chemotherapy, when contraindications are excluded.
As highly heterogeneous diseases, different subtypes of breast cancer have different biological behaviors and chemosensitivities. The use of anthracyclines in early breast cancer was largely based on the Oxford review meta-analysis, which suggested a slight benefit of anthracycline-based therapy (AB) over non-anthracycline-based (nAB) therapy in terms of both improved prognosis and treatment toxicity. 10 Adjuvant chemotherapy based on anthracyclines such as doxorubicin and epirubicin improved disease-free survival (DFS) and OS in patients with early-stage breast cancer, regardless of estrogen receptor (ER), progesterone receptor (PR), and HER2 status. 11 The use of combination regimens of anthracyclines and anti-HER2 therapeutics has, however, been limited by the increased risk of irreversible cardiotoxicity, as cardiac insufficiency and arrhythmias are associated with anthracycline treatment.12,13
There is a growing awareness that long-term cardiotoxic effects may lead to increased cardiovascular morbidity and mortality. According to the 3-year follow-up data from the Breast Cancer International Research Group (BCIRG) 006 trial, a non-anthracycline combination regimen of docetaxel, carboplatin, and trastuzumab may be the best alternative to adjuvant therapy for early HER2-positive breast cancer. 11 Considering the high number of toxic side effects of anthracycline chemotherapy drugs, more studies in recent years have started to explore whether anthracycline chemotherapy drugs can be avoided without affecting the prognosis of HER2-positive breast cancer patients 13 and this debate has not yet been definitively concluded. This study will provide informative suggestions on the need for concurrent anti-HER2 drugs and anthracyclines during neoadjuvant therapy in HER2-positive breast cancer patients.
Methods
A systematic evaluation and meta-analysis were performed according to the preferred reporting items for systematic reviews and meta-analyses guidelines, 14 Supplementary File 1 shows this in more detail. Prior to conducting this study, it was registered in the International Prospective Register of Systematic Evaluations (PROSPERO). 15
Search strategy
The PubMed, Embase, and Web of Science databases and ASCO/EMSO/SANCS website were examined for relevant articles in any language from September 1, 1998, to December 1, 2022. References were searched using a combination of medical subject headings and free-text words, such as “anthracycline,” “epirubicin,” “breast neoplasms,” “breast cancer,” “breast carcinoma,” “neoadjuvant,” “preoperative,” “trastuzumab,” and “pertuzumab.” References of potentially eligible studies were also searched, and “similar articles” on PubMed were used to find eligible studies. Two investigators conducted reference searches independently, and a third investigator was consulted when disagreements arose. More details of the search strategy are provided in the Supplementary Information.
Eligibility criteria
Both prospective and retrospective studies were considered relevant and study criteria included any study involving a combination of anti-HER2 therapeutic agents and an anthracycline-based neoadjuvant regimen as the experimental group, with the presence of a control group without anthracyclines, the diagnosis of breast cancer had to be made by histopathological examination including needle aspiration and mass excision biopsies, patients included in the study were not recurrent or had a metastatic tumor, and only studies with a NewCastle-Ottawa Scale (NOS) score greater than 6 were included. The exclusion criteria included abstracts, letters, case reports, reviews, single-arm clinical studies, non-English language studies, and patients with non-HER2-positive breast cancer.
Data extraction and quality assessment
Two researchers independently extracted information on all eligible candidate studies using a spreadsheet. Full-text searching and reviewing were performed for those publications for which classification could not be completed by title and abstract. The items recorded for each publication included first author, study title, date of initial publication, country, design treatment regimen, number of cases, follow-up time, number of pCR events, and the number of prognostic events which included OS, DFS, and cardiotoxic events.
Study quality was assessed according to NOS. 16 This scale consists of 3 components including selection scored 0 to 4, comparability 0 to 2, and outcome 0 to 3 points. A NOS score greater than 6 was considered a high-quality study and was included in the meta-analysis.
Statistical analysis
The pooled odds ratio (OR), pooled hazard ratio (HR), and 95% confidence intervals (CIs) were extracted directly from each article or estimated using the method of Parmer et al. 17 An OR that exceeded one suggested a higher pCR rate in the AB group, and an HR less than one suggested a better prognosis or better cardiac safety in the AB group. Cochran Q test and Higgins I-squared analyses were used to assess the heterogeneity of the studies. Fixed effects using the Mantel-Haenszel method and random effects using the DerSimonia-Laird method were used as models to calculate the combined HR and 95% CI. A heterogeneity P < 0.10 or I 2 > 50% indicated the presence of significant heterogeneity. In the case of heterogeneity, a random-effects model was used instead of a fixed-effects model. Sensitivity analysis was performed by excluding heterogeneous studies or studies with the smallest sample size depending on the presence or absence of significant heterogeneity.
Publication bias was evaluated using Begg’s funnel plot and Egger’s linear regression test. All P values were 2-sided, and P < 0.05 was considered statistically significant. All statistical analyses were performed using STATA 15.0 (STATA, College Station, Texas).
Results
Description of included studies
Initially, 326 eligible publications were retrieved. After further evaluation and screening, 10 papers covering 7 studies were included in the meta-analysis,18 -27 totaling 1366 patients. Of these, 815 patients received anthracycline-based neoadjuvant regimens and 551 patients received anthracycline-free regimens. The workflow of the literature search and review is shown in Figure 1.

Flow diagram of identifying eligible studies.
The baseline characteristics are summarized with the extracted data in Table 1. Four of the 7 studies were prospective and 3 were retrospective. The ORs for pCR rates were directly available or known by calculation in all studies. Five studies reported the relationship between different treatment regimens and prognosis, but the observed metrics were inconsistent, including event-free survival (EFS), DFS, progress-free survival (PFS), relapse-free survival (RFS), and OS in at least one article.
Characteristics of the studies included.

Defined as LVEF declines of ⩾10% from baseline to <50%.
Anthracylines arm.
The characteristics were derived from the overall population of the study, including neoadjuvant and adjuvant therapies.
Abbreviations: C, cyclophosphamide; Cb, carboplatin; DFS, disease-free survival; E, epirubicin; EFS, event-free survival; ER, estrogen receptor; F, 5-fluoruoracil; H, Trastuzumab; LVEF, left ventricular ejection fraction; NR, not reported; OS, overall survival; P, paclitaxel; pCR, pathological complete response; PFS, progression-free survival; PR, progesterone receptor; Ptz, pertuzumab; RFS, relapse-free survival; T, docetaxel.
The quality of the included studies was evaluated using NOS, as shown in Figure 2. All included studies were of moderate or high quality and were therefore dependable. The evaluation using Begg’s test with Pr >|z| = 0.230 and Egger’s tests with P >|t| = 0.116 showed no significant publication bias, as seen in Figure 3.

Risk of bias of the studies included (Newcastle-Ottawa Scale).

Begg’s funnel plot (A) and Egger’s linear regression test (B) of OR of pathologic complete response in anthracycline-based therapy arms vs non-anthracycline-based therapy arms.
The pCR rate of anthracycline-based compared with nAB NAC
All 7 studies, including 1328 HER2-positive breast cancer patients that were available to assess the pCR rate of neoadjuvant therapy, used a regimen of chemotherapy with or without anthracyclines combined with anti-HER2 therapeutic agents. The range of pCR rates in the 7 studies ranged from 13.70% to 67.96%, and the pooled absolute rate of pCR for the total population after pooled analysis was 55.20%. The pCR rate of 53.32% for those receiving AB neoadjuvant regimens was not significantly different from the 58.00% pCR rate in the nAB neoadjuvant control group, with an OR of 0.73 with a 95% CI of 0.43 to 1.24 (P = .246) as seen in Figure 4A. Significant between-study heterogeneity was found, and the random-effects model was used.

Forest plot of odds ratios of the pathologic complete response for the comparison of the AB therapy vs nAB therapy neoadjuvant chemotherapy for HER2-positive breast cancer. (A) All studies. (B): Prospective study subgroup including 4 studies. (C) Tumor stage 0-2 ⩾ 50% subgroup including 5 studies.
A total of 4 studies were prospective randomized controlled studies. A meta-analysis of these studies showed that combining an AB chemotherapy regimen with anti-HER2 therapy also failed to yield positive findings, with a pCR rate of 57.26% in the experimental group and 61.71% in the control group without anthracyclines, with an OR of 0.84% and 95% CI of 0.55 to 1.30 (P = .440) as seen in Figure 4B.
The original tumor size of the included patients was described in all studies and 5 included more than 50% of patients with tumor diameters less than or equal to 5 cm, classified clinical T 0 to 2. The other 2 studies included patients with relatively larger tumor loads. A meta-analysis of patients in these 5 studies was performed as a low tumor load (T0-2) subgroup and showed that this patient population did not benefit from neoadjuvant regimens that included anthracyclines, with an OR of 0.73% and 95% CI of 0.37 to 1.44 (P = .357) summarized in Figure 4C.
Survival benefits of anthracycline-based vs nAB NAC
In the 5 studies containing survival data, the median follow-up time ranged from 26.8 to 61.1 months. The types of metrics used to describe prognostic relevance included EFS, DFS, PFS, RFS, and OS, but the types of metrics collected were not uniform across all studies, so only a pooled analysis for OS was performed. Total OS data from 3 studies were combined, and the pooled absolute rate of OS in the experimental group of 595 patients containing anthracyclines was 95.63%, which was not statistically different from 95.54% for the control group of 448, with an HR of 0.64% and 95% CI of 0.35 to 1.19 (P = .157), as seen in Figure 5. No significant heterogeneity was found in these studies, so a fixed-effects model was used.
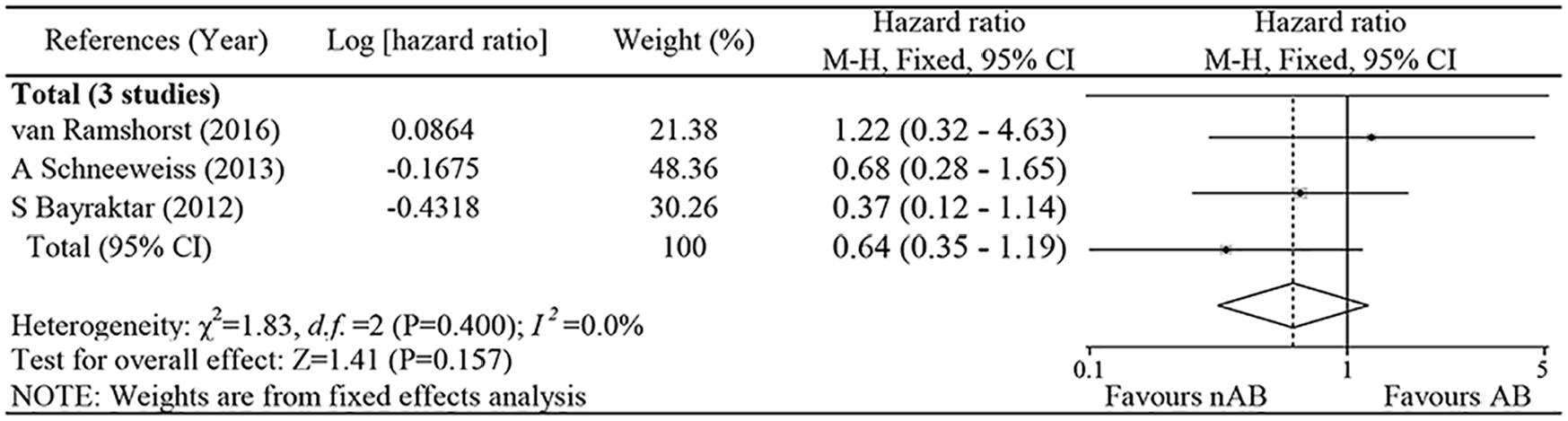
Forest plot of hazard ratios of the overall survival for the comparison of the AB therapy vs nAB therapy neoadjuvant chemotherapy for HER2-positive breast cancer.
Cardiac safety of anthracycline-based vs nAB NAC
A total of 4 studies reported cardiac safety data during treatment and follow-up and a total of 877 patients consisting of 472 in the experimental group and 405 in the control group, received at least 1 complete cycle of treatment to be included in the cardiac safety analysis. A total of 163 (18.59%) adverse cardiac events were reported in the experimental or control groups of the 4 studies. The left ventricular ejection fraction (LVEF) decreased according to the commonly reported non-CTCAE definition of LVEF decline of 10% or more, and LVEF below 50% was observed more frequently in the anthracycline-based 90 patients (19.07%) than in the non-anthracycline group of 54 patients (13.33%) with an HR of 1.62% and 95% CI of 1.11 to 2.36 (P = .013), as seen in Figure 6. No significant heterogeneity was found in these studies, so a fixed-effects model was used.

Forest plot of hazard ratios of the left ventricular ejection fraction decreased for the comparison of the AB therapy vs nAB therapy neoadjuvant chemotherapy for HER2-positive breast cancer.
Discussion
The main aim of neoadjuvant therapy is to reduce tumor load or to improve pCR for a better chance of surgery.28,29 The pCR rate after neoadjuvant chemotherapy (NAC) has been reported to be one of the valuable predictors of long-term survival prognosis and a higher pCR rate means better long-term survival. 30 In patients with locally advanced HER2-positive breast cancer, the combination of trastuzumab drugs had better efficacy compared with chemotherapy alone.31,32 The advent of dual-targeted treatment modalities had especially further increased the benefit of anti-HER2 therapy24,33 and avoided serious cardiotoxicity problems. This has led to a significant improvement in the prognosis of the subtype of HER2 overexpression. Whether anthracyclines can be exempted from an intensifying anti-HER2 therapy has always been the focus of discussion, as previous studies have shown conflicting results.34,35 While comparing therapeutic efficacy, the level of cardiotoxicity increase is a key factor in deciding whether anthracyclines should be used. This meta-analysis evaluated the efficacy and safety of concomitant use of anthracyclines based on anti-HER2 therapeutic agents for neoadjuvant treatment of HER2-positive breast cancer. Compared with previous studies and reviews, this study targeted the inclusion of all accessible controlled studies and provided more convincing evidence and conclusions.
This study showed that the use of anthracyclines in chemotherapy regimens did not further improve the pCR rate with neoadjuvant therapy in the AB group (53.32%) compared with the nAB group (58.00%) and the same conclusion was shown by pooled analysis of a small number of prospective studies. The results of the subgroup analysis also suggested that anthracyclines appear to be non-essential in studies with relatively small tumor loads. Cancer is a dynamic disease, and this feature becomes particularly prominent during tumor progression, with the same tumor exhibiting different sensitivities to cytotoxic drugs, and therefore heterogeneity is also an important cause of tumor resistance, forcing the development of therapies for locally advanced tumors. 36 Due to the limitation of eligible data information, this study was not able to perform further subgroup analysis on subgroups of the population where tumor diameter exceeded 5 cm, or when positive lymph node metastasis was present, so concerns exist about whether anthracyclines can be safely dispensed in HER2-positive patients with a locally advanced, large tumor load.
Unlike chemotherapy that targets rapidly growing and dividing cells, monoclonal antibodies can block the interaction and function of unregulated proteins within the extracellular compartment, making HER2 an ideal protein for targeted cancer therapy. 37 Early HERA studies and the BCIRG-006 trial11,38 have demonstrated the importance of anti-trastuzumab for HER2 overexpression in breast cancer. Adequate blockade of the HER2 signaling pathway by dual-targeted agents in both neoadjuvant and adjuvant stages can further improve prognosis33,39 and studies like PAMELA 40 explored the possibility of “de-chemothering” Her2-positive patients in neoadjuvant therapy. That study found that the combination of lapatinib and trastuzumab resulted in a much higher subtype pCR rate of 67% in patients with HER2 overexpression subtypes, but there was still a lack of strong evidence to support whether the high pCR rate of neoadjuvant dual targeting ultimately translates into improved long-term survival prognosis. This study showed no statistical difference in 3-year OS rates comparing the AB combination and nAB groups, suggesting that the additional use of anthracyclines does not seem to be meaningful in improving both neoadjuvant pCR rates and prognosis when using adequate anti-HER2 therapy.
Drug-related side effects are important factors for consideration in addition to the effect of neoadjuvant therapy. Cardiotoxicity induced by anticancer drugs can be broadly classified into 2 categories. The first category is primarily caused by targeted therapies, where trastuzumab increased the risk of cardiotoxicity because of the effect of heterodimer formation between ErbB2 and ErbB4 on the proliferation and contraction of cardiomyocytes, while inhibiting HER2-induced intracellular signaling. 41 This effect is mainly attributed to increased reactive oxygen species (ROS), reduced nitric oxide (NO) production, impaired vasodilation, and decreased myocardial blood flow, resulting in decreased LVEF, cardiomyocyte apoptosis, and increased cardiac workload. This type of cardiotoxicity is notably independent of drug dosage and does not directly cause myocardial damage and appears to be reversible and transient on drug discontinuation. The second category of cardiotoxicity is associated with anthracycline drugs and is characterized by irreversible and cumulative effects. Anthracyclines exert tumor suppressive effects by interacting with DNA rotase and topoisomerase II (TOP2A), causing chromosomal double-stranded DNA breaks 42 and generating ROS, which induce DNA damage and lipid peroxidation, which is the main mechanism of anthracycline-induced cardiotoxicity, 12 so whether anthracyclines can be safely used in combination with targeted drugs becomes a very important issue.
The occurrence of a cardiac adverse event can have different clinical manifestations, and in studies with dual-targeted drugs, these included symptomatic left ventricular systolic dysfunction, myocardial infarction myocardial infarction, and infarct stroke events. 21 A meta-analysis of 26 clinical studies that included patients with breast cancer and other tumors treated with lapatinib as monotherapy revealed an overall incidence of cardiovascular events of 3.0% with a 95% CI of 1.50% to 6.10%, including hypertension as the most common manifestation, 1.7% left ventricular dysfunction and insufficiency, and 1.8% LVEF decline (1.8%). 43 Prolonged QT interval has also been reported in several reports on patients taking sunitinib and has even been associated with sudden death, 44 illustrating its potential cardiotoxicity. Previous studies had found that, while severe cardiotoxicity is uncommon when anthracyclines are used in combination with targeted drugs, the decrease in LVEF was more variable and valuable to assess, making it the most important index for evaluating cardiac function. 9 This study found that the combination of anthracyclines significantly increased the event rate of LVEF decline from 19.07% in the AB group to 13.33% in the nAB group (P = .013), so this result may limit the use of anthracyclines in the HER2-positive breast cancer population, especially in cases where the cardiac underlying disease is already present. But breast cancer is complex including large tumors, lymph node metastases, and high Ki-67 expression, and a balance needs to be struck between oncologic therapy and cardiac safety; it is recommended that all patients undergo a baseline cardiac evaluation prior to treatment and consider monitoring strategies for high-risk individuals based on the oncologic treatment strategy and the patient’s past medical history.
An early analysis of the incidence of cardiac dysfunction due to HER2 inhibitors reported a 27% incidence of cardiac dysfunction when anthracyclines or cyclophosphamide were used in combination with trastuzumab. In sequential therapy, heart failure occurs in approximately 1% to 4.1% of patients, as well as a decrease in LVEF in 4.4% to 18.6% of patients. 45 Although the pathological mechanisms of cardiac impairment caused by HER2 inhibitors and anthracyclines are different, it is also more difficult to distinguish the source of these drug toxicities when patients do develop it. One characteristic of HER2 inhibitor-induced cardiotoxicity is that after discontinuation of HER2 inhibitors for LV decompensation, LV function usually improves for 2 to 4 months and the reintroduction of HER2 inhibitors can be considered after LVEF returns to normal. The authors therefore recommend that cardiac function needs to be monitored throughout the patient’s treatment including chemotherapy and targeted therapy and even after completion of treatment.
In recent years, with the emergence of anti-HER2 therapeutic agents and the optimization of (neo)adjuvant anti-HER2 treatment regimens for early-stage HER2-positive early-stage breast cancer, the combination of anti-HER2-targeted therapy with anthracycline-based chemotherapy has attracted much attention and has been intensified to avoid further myocardial damage from anti-HER2-targeted therapy. This meta-analysis suggests that neoadjuvant anti-HER2 therapy in combination with anthracycline-free therapy does not affect the long-term prognosis of patients treated with adjuvant therapy, but it can achieve similar efficacy to anthracycline-based chemotherapy regimens and avoid increased cardiotoxicity. At the same time, major guidelines have identified anti-HER2-targeted therapy in combination with deanthracycline chemotherapy as the first choice of (neo)adjuvant therapy for HER2-positive early-stage breast cancer, and the role of anthracyclines in early-stage HER2-positive breast cancer appears to be increasingly limited. Although there are many clues that anthracycline chemotherapy can be avoided in early-stage HER2-positive breast cancer, some physicians still believe that there is no independent phase III non-inferiority study that supports this conclusion, and therefore anthracycline chemotherapy should not be removed. The CompassHER2-pCR and CompassHER2-RD studies have attempted to optimize subsequent adjuvant regimens through the effects of neoadjuvant therapy, but prior to such clinical studies, adjuvant therapy for HER2-positive breast cancer has been explored in other directions.46,47 Before answering the question of if (neo)adjuvant therapy for early-stage HER2-positive breast cancer can avoid anthracycline-based chemotherapy, there are data to confirm that neoadjuvant platforms can be used to screen patients for the avoidance of chemotherapy or enhancement of chemotherapy. It is foreseeable in the future that patients with early-stage HER2-positive breast cancer will undoubtedly receive individualized adjuvant therapy that is more suitable for them.
This is the first meta-analysis of the efficacy and cardiac safety of concurrent anti-HER2 agents and anthracycline-based NAC for HER2-positive breast cancer by pooling 2-arm studies, avoiding the bias from single-arm studies. The meta-analysis provided evidence for the clinical practice of concurrent use of targeted agents in combination with anthracycline-based NAC for HER2-positive breast cancer but had some limitations. First, although all eligible studies were systematically searched, only 4 studies were prospective randomized trials. Second, among the included individual studies, although the treatment regimens in the control group all included anthracyclines, the treatment regimens and duration of treatment were not strictly the same between studies and when the number of included studies was less than 10, Egger’s and Begg’s tests had low efficacy and may not have been effective in detecting publication bias. Finally, heterogeneity among studies may also affect the findings because the raw data provided limited information, and subgroup analyses for populations with a higher risk of recurrence such as those with lymph node metastases were not able to be done, so more prospective evidence is needed to support whether anthracyclines can be safely exempted in this population. Heterogeneity among studies may also arise due to variations in NAC regimens and to mitigate the impact of these differences; a random-effects analysis was conducted to minimize confounding factors. Variations in the routine monitoring of cardiac toxicities during treatment across different studies may also have influenced the study outcomes and data integrity varied for other confounding factors, such as tumor size and lymph node metastasis status.
Conclusions
This study showed that the omission of anthracycline in neoadjuvant regimens for HER2-positive breast cancer is not inferior to anthracycline-based chemotherapy, especially in the population of patients with small tumor loads, and it did not significantly improve prognosis during long-term follow-up. Additional use of anthracyclines during and after completion of treatment significantly increased the probability of cardiotoxic events, so these findings suggested that neoadjuvant treatment without anthracyclines was feasible for some HER2-positive breast cancers.
Supplemental Material
sj-pdf-1-onc-10.1177_11795549231195293 – Supplemental material for The Value of Neoadjuvant Anthracycline-Based Regimens for HER2-Positive Breast Cancer: A Systematic Review and Meta-analysis Including 1366 Patients
Supplemental material, sj-pdf-1-onc-10.1177_11795549231195293 for The Value of Neoadjuvant Anthracycline-Based Regimens for HER2-Positive Breast Cancer: A Systematic Review and Meta-analysis Including 1366 Patients by Yuqin Ding, Kaijing Ding, Xiangming He, Wenju Mo, Chenlu Liang, Lijie Gong, Yuting Huang and Xiaowen Ding in Clinical Medicine Insights: Oncology
Footnotes
Author Contributions
Conceptualization and Writing—original draft preparation: Yuqin Ding; investigation: Kaijing Ding; visualization and formal analysis: Xiangming He; resources and data curation: Wenju Mo; software: Lijie Gong and Chenlu Liang; methodology: Yuting Huang; conceptualization and project administration: Xiaowen Ding.
Declaration of Conflicting Interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Natural Science Foundation of Zhejiang Province (LTGY23H160012 and LTGY23H160015) and the Health Science and Technology Program of Zhejiang Province (2021RC041, 2022RC014, 2021KY572, and 2021KY566).
Compliance with Ethical Standards
This article does not contain any human participants and/or animals.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
