Abstract
The goal of therapy for patients with metastatic breast cancer (MBC) is prolonging life and palliation of symptoms. Thus the preferred approach remains to use, at least initially, non-cytotoxic drugs. In hormone receptor-positive breast cancer the sequential use of single anti-estrogen drugs, e.g. tamoxifen, aromatase inhibitors, and many others is standard, but eventually drug resistance will lead to failure of these compounds and a switch to chemotherapy will be necessary. Reversing resistance to anti-estrogen therapy in MBC is one of the strategies to avoid and delay the use of cytotoxic compounds. The mammalian target of rapamycin (mTOR) has been recently associated with in vitro reversal of drug resistance, including tamoxifen resistance. A number of early clinical studies have confirmed the concept and, more recently, everolimus was successfully tested in a randomized controlled trial in postmenopausal patients who progressed on previous anti-estrogen therapy for MBC. This manuscript will review the biology, preclinical and clinical data including the randomized controlled trial that lead to the approval of everolimus by the US FDA.
Introduction
Although metastatic breast cancer (MBC) continues to be an incurable disease in most cases, meaningful improvements in survival as well as in quality of life have been seen with the use of cytotoxic, endocrine, biological, and, more recently, mTOR inhibitors therapy. Hormonal therapies, such as tamoxifen (TAM) and aromatase inhibitors (AI), are considered the cornerstone treatment for postmenopausal women with hormone receptor-positive MBC (1). In the USA and western world, approximately 75% of patients with MBC have hormone receptor-positive tumors, and endocrine therapy is indeed an effective treatment strategy (1, 2).
Endocrine therapy should be used in all patients with metastatic disease and positive hormone receptors; systemic chemotherapy is the preferred treatment modality for patients with hormone receptor-positive MBC with an aggressive disease after hormonal failure, and those with visceral metastases. A meta-analysis compared the response rates of chemotherapy alone versus endocrine therapy alone, including 8 small randomized clinical trials (3). The overall conclusion favored chemotherapy, with a higher pooled estimated response rate than endocrine therapy (relative risk 1.25; 95% CI, 1.01-1.54). However, the 2 largest trials included in this work had conflicting findings, and no significant difference was seen in the overall survival (OS) (hazard ratio [HR] 0.94; 95% CI, 0.79-1.12) (4, 5). In addition, the subset analysis did not show significant differences suggesting an effect of age, menopausal status, or types of metastatic disease on the efficacy of either therapy. Moreover the hormone receptor status of many patients included in these studies was unknown, and little data comparing quality of life and toxicities between the 2 therapies was provided. Nevertheless, the evidence still points out to some benefit to chemotherapy alone in patients with aggressive disease, so this approach is still current to date. For these patients with advanced disease, as well as for those without estrogen receptor (ER)/human epidermal growth factor receptor 2 (HER2) positivity, cytotoxic chemotherapy is essential, given it provides faster responses with or without targeted agents. There are still certain ER-positive breast tumors that would require chemotherapy based on basal-like signature and established aggressive disease. Nonetheless, evidence suggests that the role of cytotoxic agents may be minimized in certain patients with ER and/or HER2 sensitivity and good end organ function (6).
The question that followed these trials was whether or not the combination of chemotherapy and endocrine therapy would have a positive impact on aggressive MBC outcomes. Two systematic reviews addressed this issue and no survival benefit was seen when chemotherapy and endocrine therapy were combined (7, 8). Even though endocrine therapy is effective for treatment of early and MBC, the increasing use of TAM and AI in the adjuvant setting impacts treatment choices when the disease relapses or becomes metastatic. In addition, around 30% of patients do not respond to AI, and all those who initially respond will eventually progress (9–11). The 5-year OS rates for patients with MBC ranged from 21% to 40% (12, 13). Long-term survival is very rare with only 2% of patients surviving more than 20 years after the initial diagnosis of MBC (12). To our knowledge the longest OS in a patient with MBC reported in the literature referred to a patient diagnosed with stage I (T2N0M0) breast cancer at 45 years of age, who developed metastasis 22 years after the initial diagnosis, and survived 18 additional years after metastasis presentation (14).
Endocrine resistance is, therefore, an important area of interest in the study of MBC. Several mechanisms have now been described. Inhibition of the mammalian target of rapamycin (mTOR) pathway has not only been proposed in preclinical models as an effective way to overcome this phenomenon, but recent clinical data demonstrated that mTOR inhibitors might reverse hormone resistance in MBC.
The PI3K/Akt/mTOR Pathway
The PI3K (Phosphatidylinositol 3-Kinase)/Akt (Protein Kinase B)/mTOR pathway (Fig. 1) is a well-characterized intracellular pathway that regulates several normal cellular functions including cellular proliferation, growth, survival, and mobility (15). This pathway has been linked to the pathology of a number of diseases when it is aberrantly activated. When dysregulation happens, apoptosis is downregulated and proliferation occurs causing a variety of tumors including breast cancer (15).
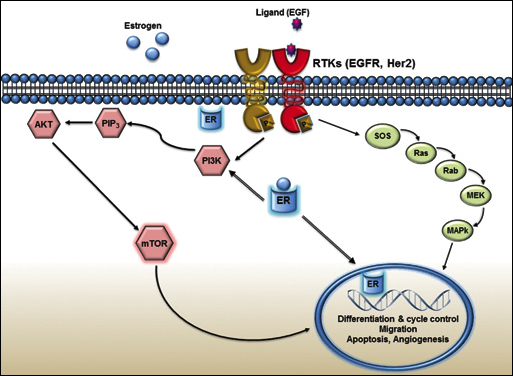
In the absence of hormone, estrogen receptors are largely located in the cytosol. Estrogen binding to the estrogen receptor triggers the migration of the receptor from the cytosol into the nucleus, dimerization of the receptor, and binding of the receptor dimer to DNA, causing transcription of downstream DNA into mRNA. Estrogen receptors also occur within the cell nucleus, and both estrogen receptor subtypes have a DNA-binding domain and can function as transcription factors to regulate the production of proteins. Estrogen receptors are also associated with cell membranes and can form complexes called receptor tyrosine kinases (RTK) (EGFR, HER2 and IGF-1). Through this, RTK's signals are sent to the nucleus through the mitogen-activated protein kinase (MAPK/ERK) pathway and phosphoinositide 3-kinase (Pl3K/Akt) pathway.
Activation
The pathway is activated through complex mechanisms of intracellular signaling and propagation, which begin with PI3K activation by receptor tyrosine kinase in response to growth factor receptors such as EGFR (epidermal growth factor receptor), HER2, IGF-1R (insulin-like growth factor-1 receptor), and oncogenes such as RAS (Rat sarcoma) (16). Upon activation PI3K (Class I) phosphorylates PIP2 (phosphatidylinositol-4, 5-biphosphate) into PIP3 (phosphatidylinositol-3,4,5-triphosphate), which then activates Akt at the cell membrane (15, 17). Activated Akt, in turn, phosphorylates several downstream targets that are involved in the control of apoptosis and cell proliferation. The most studied target of Akt is the serine/threonine kinase mTOR protein, which plays a critical role in regulation of protein translation (15, 18). Akt directly activates, as well as indirectly activates mTOR through inactivating TSC2 (tuberous sclerosis complex 2, also called tuberin) (16).
Inhibition
Strict control of the PI3K/Akt/mTOR pathway through regulatory feedbacks is essential for normal cellular function. PI3K signaling is terminated by dephosphorylation of PIP3. The tumor suppressor PTEN (phosphatase and tensin homolog) protein acts as a negative regulator of the PI3K pathway, antagonizing the activation of Akt. Another inhibitor of the PI3K pathway in human epithelial cells is the tumor suppressor INPP4B (inositol polyphosphate 4-phosphatase type II) (18). The downstream mTOR (mTOR Complex 2 [mTORC2]) probably provides some level of positive feedback to Akt as well. In addition, S6K1 (S6 kinase-1), an mTOR downstream effector, also inhibits Akt by preventing activation of PI3K (15–17). TSC2 normally inhibits mTOR-mediated downstream signaling (16).
Deregulation
The PI3K/Akt/mTOR pathway is intrinsically susceptible to deregulation because of the various components and complex transduction involved. Overexpression or activation of growth factor receptors, mutations in PI3K and mutations/amplifications of Akt are contributing factors. Downregulation of the tumor suppressor PTEN protein through mutations, deletion, and epigenetic silencing is also indicated as a contributing factor. Collectively, the general consensus of the common mechanism for upregulation of the PI3K/Akt/mTOR pathway is the loss of PTEN, which is also a poor prognostic factor in human cancers (16, 19–21).
Implication in the resistance mechanism of antiestrogen therapy in MBC
The resistance to anti-estrogen therapy in ER-positive MBC involves multiple factors and mechanisms. Preclinical models have shown intricate and complex interactions between estrogen-ER complexes and growth factors pathways. Estrogen hypersensitivity and increased growth factor signaling are partly responsible for the upregulation of the cancer cell pathway (22). The mTOR pathway has been found to have paramount importance in the genesis and development of drug resistance (23, 24). The crosstalk between these pathways was seen when long-term estrogen-deprived breast cancer cells were able to upregulate Erk1/2, and ErB2, and increase phosphorylation of Akt ultimately increasing the activation of the mTOR pathway (25–27). This observation led to the assumption that upregulation of growth factor pathways are extremely important for cells cultured in low-estrogen media, which are capable of overcoming this hormonal disadvantage, and continue to grow in the absence of hormonal stimuli. A clinical study demonstrated that patients with ER-positive tumors treated with TAM showed a higher ErB2 and p38 mitogen-activated protein kinase expressions at relapse (27). This observation suggests that targeting cross-talk pathways can be a sound idea to decrease endocrine therapy failure, as well as de novo resistance.
In this setting the mTOR pathway is a promising target, and the interest in this area has grown over the past decade. The PI3K/Akt pathway upregulation is fundamental for several cellular processes including proliferation, growth, apoptosis and cytoskeletal rearrangement. Consequently its deregulation can determine a more aggressive phenotype, which will translate in worse prognostic predictors for unwanted outcomes (28, 29). In addition, the same pathway has been identified to be associated with resistance to anti-estrogen therapy (30).
The rapamycin-derived compounds, such as everolimus, temsirolimus and sirolimus are capable of inhibiting the mTOR pathway. These drugs act inhibiting the enzyme complex mTORC1, in a downstream reaction of the PI3K pathway. More specifically, mTOR inhibitors bind to the protein FKBP12 that is present on the mTORC1 enzyme complex and, by doing so, the enzyme complex activity is inhibited (31). Of note, these drugs do not inhibit mTORC2 enzyme complex, hence the sole inhibition on mTORC1 enzyme complex leads to a feedback loop promoting mTORC2 overactivation, upregulating the AKT kinase and MAP kinase pathways, which could serve as an explanation to incomplete response or resistance to rapamycins (32).
Clinical Data of mTOR Inhibitors in Metastatic Breast Cancer
Everolimus
The safety and pharmacokinetic (PK) profile of everolimus in combination with aromatase inhibitors was initially evaluated in an open-label phase Ib clinical trial, where the association of letrozole and everolimus was evaluated in separate cohorts defined by fixed doses of letrozole 2.5 mg/day, and escalating doses of everolimus up to 10 mg/day (previously defined as the optimal biological active dose) (33). This study concluded that the combination of these medications did not produce significant PK interactions, and the overall tolerability was considered satisfactory. Of note, 22.2% of patients presented adverse events leading to medication discontinuation. The most relevant side effects were thrombocytopenia and neutropenia within 2 weeks of treatment, which led the authors to suggest a blood cell count follow-up at 15 days of therapy. Other common but less severe side effects were anorexia (5.5%), fatigue (11.1%), diarrhea (5.5%), and peripheral edema (5.5%).
A phase II clinical trial (34) evaluated the efficacy and safety of everolimus in combination with TAMin postmenopausal women with MBC, ER positive/HER-2 negative, who had undergone previous treatment with AI in the adjuvant metastatic setting. In this multi-center open-label trial, patients were randomly assigned in a 1:1 ratio to receive TAM 20 mg daily, or the combination of TAM 20 mg daily plus everolimus 10 mg daily. The 6-month clinical benefit rate (stable disease plus response rate) was 61% (95% CI, 47-74) with TAM plus everolimus, compared to 42% (95% CI, 29-56) with TAM alone. Time to progression (TTP) increased from 4.5 months to 8.6 with the combination therapy, corresponding to a 46% reduction in risk of progression (HR, 0.54; 95% CI, 0.36-0.81). Mortality was reduced by 55% with the combination therapy (HR, 0.45%; 95% CI, 0.24-0.81). Most common adverse effects reported with the combination therapy were fatigue (72% vs 53% with TAM alone), stomatitis (56% vs 7%), rash (44% vs 7%), rash (44% vs 7%), anorexia (43% vs 18%), and diarrhea (39% vs 11%). In addition, this trial also showed that patients with secondary resistance experienced a greater benefit with the combined therapy compared to patients with primary resistance (CBR: 74% vs 48%). However, this study was not free from biases and limitations, such as the open label design, and the relatively small sample size (54 patients in the combination arm and 57 patients in the TAM alone group).
The randomized controlled phase III trial BOLERO-2, which was presented and published in 2011, provided striking evidence regarding the benefit of an mTOR inhibitor, everolimus, when added to endocrine therapy in postmenopausal women with ER-positive tumors (35). The study was an international randomized clinical trial that compared patients treated with everolimus and exemestane (a steroidal AI), versus exemestane and placebo, who have progressed or recurred while undergoing treatment with a non-steroidal AI. Stratification was applied according to the presence of visceral metastasis, and previous sensitivity to endocrine therapy. Progression-free survival (PFS) was considered the primary end point, with OS and overall response rate established as secondary endpoints. A total of 754 patients underwent 2:1 randomization, 485 on the experimental/combination group and 239 in the control group (35). No significant difference was observed in terms of baseline characteristics between the 2 groups. Of note, the median age was 62 years, 56% had visceral involvement, and 76% bone metastasis. All patients had ER-positive tumors, and 72% had progesterone receptor-positive disease. All patients had HER2-negative tumors, with the exception of 2 patients for whom HER2 analysis results were missing.
The primary end point was met with a median PFS by central assessment of 10.6 months in the combination group versus 4.1 months in the control group. Grade 3 and 4 adverse effects included stomatitis (8% vs 1% in the treatment vs control groups, respectively), anemia (6% vs 1%), dyspnea (4% vs 1%), hyperglycemia (4% vs <1%), fatigue (4% vs 1%), and pneumonitis (3% vs 0%). The conclusion from the results of the trial was a significant improvement in PFS, corresponding to a 57% reduction in HR. The results of this trial culminated in its FDA approval for treatment of MBC in July 2012, as well as in the inclusion of this drug in the 2012 NCCN guidelines for treatment of MBC in postmenopausal women with positive ER progressing on non-steroidal AI.
Temsirolimus
Temsirolimus, another mTOR inhibitor, was evaluated in combination with letrozole, a non-steroidal aromatase inhibitor in a 3-arm phase II clinical trial reported in 2005 (36). At that time little data was available on how these drugs' combination would affect clinical outcomes in patients with MBC. In this study patients were randomized in a 1:1:1 ratio into 1 of the 3 cohorts, and all groups received letrozole 2.5 mg daily. Each cycle lasted approximately 2 weeks, and a total of 7 cycles of treatment was performed. Patients in group 1 received temsirolimus 10 mg daily; those in group 2 received temsirolimus 30 mg intermittently (i.e. daily for 5 days every 2 weeks); finally, patients in group 3 received letrozole alone. A total of 92 patients were enrolled: 33 in group 1, 30 in group 2 and 29 in groups 3. The most common adverse events reported from patients in group 1 were peripheral edema (46%), asthenia (42%), and diarrhea (36%); in group 2 were asthenia (60%), diarrhea (43%), and mucositis (43%); in group 3 were asthenia (55%), arthralgia (31%), and nausea (31%). Grade 3-4 toxicities were hyperglycemia in both groups receiving temsirolimus. The trial reported a higher PFS in patients receiving temsirolimus 30 mg intermittently (group 2) than those in the other 2 cohorts (median PFS: group 1, 11.5; group 2, 13.2; group 3, 11.6 months). The combination of temsirolimus and letrozole was a tolerable and safe regimen, and intermittent doses of temsirolimus seemed to be more promising in prolonging PFS, compared to daily scheduled doses.
A phase III randomized trial compared temsirolimus plus letrozole and letrozole alone in postmenopausal women with locally advanced breast cancer or MBC who never received AI (37). The study groups and drug protocol were concordant with the previously reported phase II trial (36). Hence patients were randomly assigned to 2 groups in a 1:1 ratio. Group 1 received oral temsirolimus 30 mg daily intermittently for 5 days every 2 weeks, and group 2 received oral placebo on the same schedule. Both groups received letrozole 2.5 mg daily. A total of 493 patients were assigned to group 1 and 499 patients were assigned to group 2 (controls). In group 1 at least 1 grade 3 adverse event was reported in 37% of patients, and the most common events were: hyperglycemia (4%), dyspnea (3%), neutropenia (3%), and gamma glutamyltransferase (GGT) increase (3%). With regard to group 2, 22% of patients experienced at least 1 grade 3 adverse event, and dyspnea (3%), bone pain (2%), GGT increase (1%), and asthenia (1%) were the most commonly reported adverse reactions. Interestingly, both groups showed similar objective tumor response, clinical benefit, and median PFS after 382 events (52%). For this reason an independent data monitoring committee suggested that continuation of the trial would most likely not achieve the intended level of efficacy. As a consequence, the study was terminated and the final conclusion was the lack of PFS improvement seen in the study population. Of note, 56% of patients enrolled in this trial had no prior exposure to endocrine therapy and thus might not have yet developed endocrine resistance. Interestingly this fact could have greatly influenced the results towards the no-benefit direction as opposite to the BOLERO-2 trial, where 100% of the patients had prior exposure to letrozole, or anastrozole. This observation suggests that the absence of the activated pathway in untreated hormone-sensitive ER-positive tumors does not favor the addition of mTOR inhibitors at earlier treatment stages, where patients were not previously exposed to AI.
Sirolimus
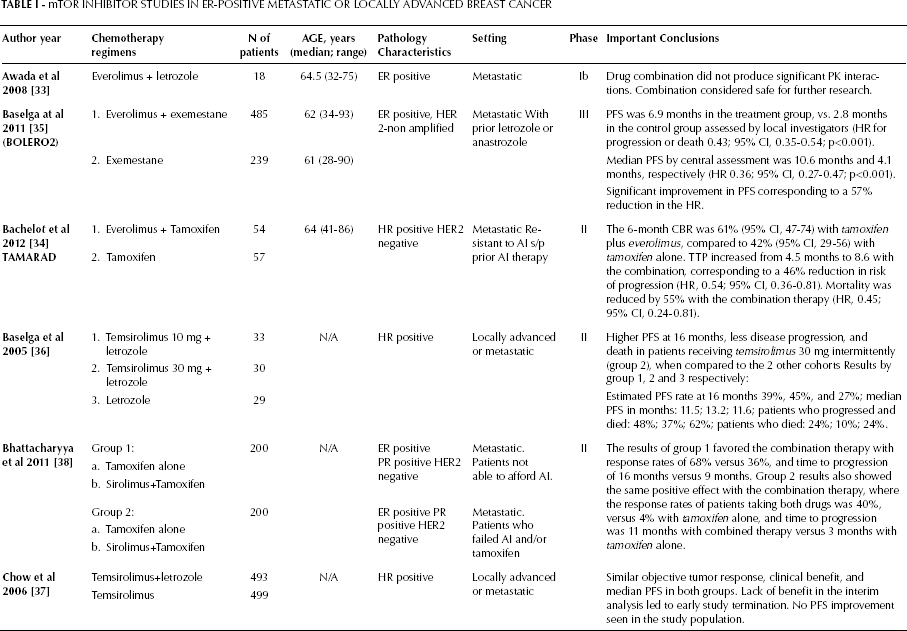
Sirolimus, another mTOR inhibitor, was also evaluated in combination with TAM in patients with MBC hormone receptor-positive, HER2 negative. In this phase II single-center clinical trial 400 patients were enrolled and divided into 2 groups depending on their prior exposure to AI, or TAM (38) (Tab. I). Group 1 consisted of 200 patients that were randomized to TAM 20 mg daily plus sirolimus 2 mg daily, or TAM 20 mg daily alone. Group 2 also had 200 patients, but included patients that had failed prior therapy with TAM or AI. These patients were randomized in a similar fashion to group 1 to receive the combination of TAM plus sirolimus, or TAM alone at the same doses. The primary end points were response rate and time to progression. Secondary end points were toxicity, safety and pharmacoeconomic analysis. The results were analyzed within each of the groups, and no comparison between the 2 groups was performed, given patient allocation was based on prior treatment status, and was not randomized. The results of group 1 favored the combination therapy, with response rates of 68% versus 36%, and time to progression of 16 months versus 9 months. Results from group 2 also showed the same positive effect with the combination therapy, where the response rates of patients taking both drugs was 40%, versus 4% with TAM alone, and time to progression was 11 months with combined therapy versus 3 months with TAM alone. The authors concluded that the combination therapy was effective and safe. In addition, pharmacoeconomic analysis deemed the drug cost effective with a favorable toxicity profile.
mTOR Inhibitor Studies in Er-Positive Metastatic or Locally Advanced Breast Cancer
Biomarkers for mTOR Inhibition in MBC
Activating mutations in PIK3CA and/or functional loss/inactivation of PTEN have been proposed as the most common PI3K/Akt/mTOR pathway alterations found in breast cancers (35, 36). Several series have shown that 50% to more than 70% of breast cancers have at least one of these molecular alterations (39–41). Activating mutations in PIK3CA are more frequent in certain sites within the kinase and helical domains (exon 9 and 20, respectively) (38). Of note, mutations in exon 20 are seen more frequently in patients with breast cancer, when compared to mutations in exon 9 (38). PTEN inactivation is known to be secondary to several mechanisms and, given its high frequency, both PIK3CA and PTEN mutations are considered the front line of biomarkers investigation in MBC (43). Moreover, PIK3CA alterations are extremely sensitive to inhibitors of the PI3K/Akt/mTOR pathway, and preclinical studies showed increased sensitivity to everolimus, as well as to the allosteric mTORC 1/2 inhibitor PP242 in tumors possessing PIK3CA alterations (43–45). In contrast, no difference was seen in tumors presenting or not the PTEN loss.
This information was translated into clinical studies in order to evaluate if genetic alterations could predict sensitivity to PI3K/Akt/mTOR pathway inhibition. A single-center assessment enrolled patients with breast and gynecological cancers in phase I clinical trials to establish the relationship between PIK3CA mutations and PI3K/Akt/mTOR inhibitors response (42). Overall, 30% of patients with PIK3CA mutations (a total of 23 patients) showed complete or partial response, whereas only 10% of patients who lacked the aforementioned mutation presented a similar response (p=0.04). However, this last group of patients harboring PIK3CA wild-type could have had other pathway alterations in the course of their disease, such as the PTEN loss alteration. In addition, 6 out of 7 responding patients received temsirolimus plus doxorubicin, so it is unclear if the observed response was due to tumor sensitivity to the liposomal doxorubicin, or to the mTOR inhibitor (46). Another study evaluated the relationship between PTEN loss and/or PIK3CA kinase domain mutation, and everolimus response (47). In this study, 8 out of 12 patients with the abovementioned alteration presented partial response (1 patient), or stable disease (7 patients). In contrast, the remaining 4 patients experienced progressive disease (p=0.0128).
Despite the promising results demonstrated by these and many other similar trials, currently no significant conclusion regarding biomarkers application can be extrapolated to clinical practice. Biomarkers evaluation predicting drug response and PI3K/Akt/mTOR pathway inhibition are certainly promising research fields. Nevertheless the small sample sizes of the current trials warrant that further investigation by larger and well-powered prospective trials is needed for properly guiding clinical interventions. Meanwhile, personalized breast cancer therapy remains a challenge and the understanding of selected biomarkers is extremely important for further advances in this field.
Future Perspectives Combinations, HER2 Resistance, Cross Talk
Most of the abovementioned studies demonstrate a promising association between AI, and mTOR inhibitors in the treatment of MBC. However, there are still no clear guidelines recommending the ideal sequence of endocrine therapy, and its combination with other drug classes. The addition of mTOR inhibitors to the breast cancer drug arsenal makes this valid discussion even more interesting and challenging for specialists around the world. The paucity of data favors an environment where treatment choices are usually based on clinical experience, presence of co-morbidities, contra-indications, and treatment costs. Consequently, treatment choices are still being driven by generic and subjective parameters, which ultimately create a barrier for the institution of individualized and more effective targeted therapies. The ability to predict which patient subpopulations will adequately respond to a given therapy is fundamental in tailoring treatment strategies, which eventually will lead to better outcomes. A change in the cancer definition paradigm from classic anatomic and histological classifications to a molecular pathway-based approach, allied to the idea that one drug does not fit all, are fundamental steps in the era of personalized anti-cancer therapy (48, 49). Without a doubt this approach can delay the use of cytotoxic therapy and completely utilize endocrine intervention, thus reducing or delaying the side effects ofchemotherapy.
The pharmacologic differences between temsirolimus and everolimus do not seem to explain the improvement seen with one agent and the lack of improvement seen-with the other. Although everolimus is administered orally continuously and temsirolimus intermittently, both agents are specific inhibitors of mTORC and both bind to FK-binding proteins (FKBPs), and the complex binds then to mTORC1 (50). More importantly, both have a low activity in breast cancer as single agents with response rates of 0%-9%. However, the temsirolimus combination seems to have lower toxicity profile when compared with the everolimus combination, raising the question of relative drug effectiveness (50).
Certainly, BOLERO-2 showed a substantial decrease in the risk of disease progression overcoming endocrine resistance. However the question that follows is whether or not clinicians will correctly select the most appropriate patients for combined therapy. Some authors have proposed that BOLERO-2 findings should not be extrapolated to a new first line therapy for all ER-positive patients, regardless of prior exposure to AI, since the lack of benefit evidenced by the phase III data of temsirolimus in combination with letrozole points out that few patients might benefit from the combined therapy upfront (37, 51). As explained before, in the mentioned trial 56% of patients had no prior exposure to endocrine therapy, a fact that has been proposed as a possible explanation of the lack of benefit of the combination therapy in that study. Nonetheless, a small number of untreated patients harboring ER-positive tumors may already have an altered and activated pathway, with consequent ‘de novo’ resistance to endocrine therapy. These patients will not respond to endocrine therapy alone, but could present a very favorable response to the combined therapy. The combination of letrozole plus everolimus in ER-positive patients has also been evaluated in the neoadjuvant setting (52). A subgroup of patients carrying ER-positive tumors with activated PIK3CA mutations demonstrated a notably greater anti-proliferative effect with the combined therapy than those randomized to letrozole therapy alone. These results remain hypothesis-generating and will hopefully be analyzed in future trials.
