Abstract
Introduction
The demographical/clinical characteristics of being Asian, having an adenocarcinoma, being female, and being a “never-smoker” are regarded as favorable predictors for epidermal growth factor receptor (EGFR) tyrosine kinase inhibitor (TKI) efficacy in non-small cell lung cancer (NSCLC) with unknown EGFR gene status. In this study, we examined the effects of the supposedly unfavorable clinical variables in EGFR-mutant patients.
Method
In total, 159 EGFR-mutant NSCLC patients' clinical features were correlated with progression-free survival (PFS), response rate (RR), and overall survival (OS). Multivariate analysis of clinical characteristics was performed using the Cox and logistic regression methods.
Result
There were 90 females (56.6%), 112 never-smokers (70.4%), and 153 patients with adenocarcinomas (96.2%). All patients were treated with EGFR-TKI, and 52.8% received TKI in a first-line setting. The median PFS of patients receiving first-line TKI was similar, regardless of gender (males vs females: 9.1 vs 9.7 months, p=0.793), smoking status (never-smokers vs smokers: 9.9 vs 9.1 months, p=0.570), or histology (adenocarcinoma vs non-adenocarcinoma: 9.7 vs 9.2 months, p=0.644). OS curves of first-line TKI-treated patients were also not associated with gender (p=0.722), smoking status (p=0.579), or histology (p=0.480). Similar results of PFS and OS were obtained for patients who received TKI beyond first-line. Multivariate analysis indicated that none of these clinical factors was an independent predictor of survival.
Conclusions
The supposedly ‘favorable’ clinical factors of female gender, non-smoking status, and adenocarcinoma were not independent predictive factors for PFS or OS in this population of EGFR-mutant NSCLC patients.
Introduction
The frequency of epidermal growth factor receptor (EGFR) gene mutations is approximately 30% in East Asia. Mutations are more common in females, non-smokers, and subjects with adenocarcinomas (1). Gefitinib and erlotinib, the first-generation EGFR tyrosine kinase inhibitors (TKIs), were initially studied in unselected populations of advanced non-small cell lung cancer (NSCLC) patients, and demonstrated no superiority to chemotherapy in terms of efficacy in clinical trials (2–4).
Further subgroup analysis showed that TKIs were more effective in patients of Asian ethnicity, female gender, with never/light smoking history, and adenocarcinomas (2, 3, 5, 6). Based on these data, patients with these clinical features were considered ‘favorable’ for EGFR-TKI efficacy in clinical practice. However, in subsequent studies there was no agreement on the predictive value of these clinical conditions (7–11).
The IPASS (Iressa Pan-Asia Study) trial (1) demonstrated that EGFR mutations were a strong predictor of favorable outcome with gefitinib in a clinically selected population. Since then, 5 more clinical trials (12–16) showed that EGFR mutations were a favorable predictive molecular biomarker. The clinical issue remained as to whether the clinical factors had predictive significance in EGFR-mutant patients. Rosell et al (16) reported that female gender was a positive prognostic factor in a Caucasian EGFR-positive cohort. However, there was still no sense of whether the clinical characteristics mentioned above affected the efficacy of EGFR-TKIs in Asian EGFR-mutant NSCLC patients.
Thus, we conducted this retrospective study of EGFR-sensitive mutant patients previously treated with EGFR-TKIs, and sought to determine the predictive values of the traditional supposedly unfavorable clinical characteristics, which included being male, having a smoking history and having an non-adenocarcinoma, by analyzing progression-free survival (PFS), response rate (RR), and overall survival (OS).
Materials and Methods
Patients
Consecutive EGFR-mutant NSCLC patients who received EGFR-TKIs (gefitinib or erlotinib) from January 2005 to December 2011 at the Guangdong Lung Cancer Institute (GLCI) were enrolled. The inclusion criteria were: (i) patients with EGFR-sensitive mutations, such as exon 19 deletion and the L858R mutation, (ii) pathologically confirmed stage III-IV NSCLC and treatment with EGFR-TKIs for longer than 3 months, and (iii) an assessable primary tumor for which the response was evaluated regularly. Exclusion criteria were: (i) other concomitant malignant disease, (ii) inadequate organ function, and (iii) lack of detailed information regarding subsequent treatment after EGFR-TKI failure.
Patient clinical and therapeutical information were collected from electronic medical records at GLCI. All tissues used for this study were from the GLCI tissue bank.
Informed consent was obtained from each person for subsequent molecular analyses before any biopsy was performed. This study was approved by the institutional review board of the Guangdong General Hospital.
According to the each of the supposed favorable clinical characteristics previously mentioned, we classified the 159 EGFR-mutant patients into 2 groups: males and females, never-smokers and ever-smokers, and histologically typed adenocarcinomas and non-adenocarcinomas. A never-smoker was defined as a patient who had smoked 100 or fewer cigarettes ever; the rest were classified as ever-smokers (14).
EGFR mutations
EGFR mutations were detected routinely in all patients who took EGFR-TKIs at GLCI from 2005. All tissues were assessed by 2 independent pathologists for tumor histology to ensure the tumor content was at least 50% before EGFR analysis. DNA was extracted from freshly frozen tissue, which had been frozen in liquid nitrogen for 30 minutes, and stored at −80°C. The procedure and conditions for EGFR analysis have been described previously (17).
Response evaluation and statistical analysis
The response evaluation of EGFR-TKIs was performed according to the Response Evaluation Criteria in Solid Tumours (RECIST) guidelines (ver. 1.1). PFS was calculated from the first day of treatment to the first radiological evidence of disease progression or death for any reason. OS was defined as the interval from the start of TKIs to the last visit or death; data from patients who were alive at the last follow-up visit were censored.
The chi-squared or Fisher's exact test were used to detect differences in demographic and clinicopathological characteristics and RR. The independent influence of clinical variables on RR was analyzed by logistic regression. Survival curves were constructed using the Kaplan-Meier method and the log-rank test was used to assess differences between curves. The hazard ratio (HR) and the 95% confidence interval (CI) were calculated using the Cox regression model. All analyses were conducted using the SPSS software (ver. 13.0). The p values were two-tailed, and p values <0.05 were considered to indicate statistical significance.
Results
Clinical characteristics
In total, 169 patients were screened. Two patients with exon 18 mutations, 2 with the T790M mutation, and 6 patients harboring coexisting mutations were excluded to avoid potential bias. Thus, a total of 159 EGFR-mutant patients were included; all patients were ethnically Chinese. In total, 97 patients (61.0%) died by the time of their last visit. The median PFS after EGFR-TKIs treatment was 9.7 months (95% CI, 8.97-10.43) and the median OS of the whole population was 20.5 months (95% CI, 17.00-24.00).
Patient clinicopathological characteristics are shown in Table I. Clinical factors were balanced between the groups, except smoking history; there were significantly more males with a history of smoking than females (63.8% vs 3.3%; p<0.001). First-line EGFR-TKI was used in 52.8% of the subjects in the study cohort; there were more males in the erlotinib treatment group than in the gefitinib group (61.5% vs 34.6%; p=0.001). No significant difference was observed between the patients' subsequent treatment distribution in the groups.
Clinical Characteristics of Male and Female Patients
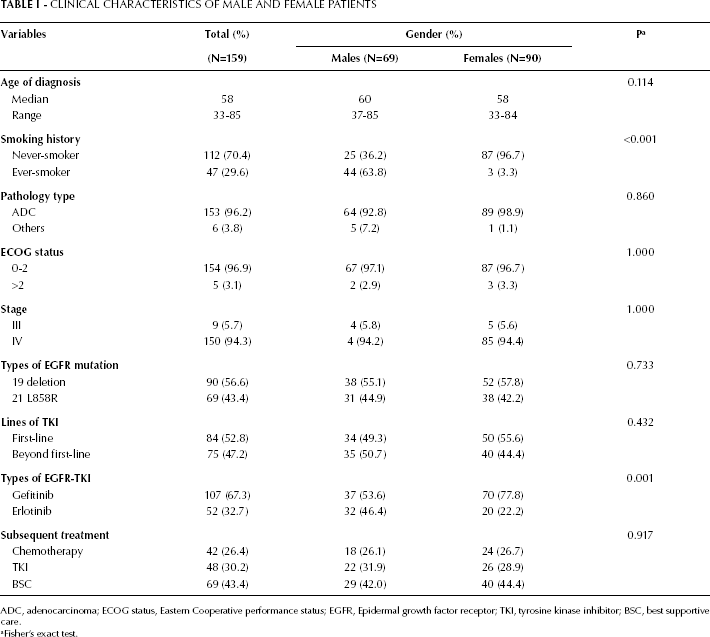
ADC, adenocarcinoma; ECOG status, Eastern Cooperative performance status; EGFR, Epidermal growth factor receptor; TKI, tyrosine kinase inhibitor; BSC, best supportive care.
Fisher's exact test.
Influence of clinical characteristics on EGFR-TKI efficacy
Gender
We first divided the study population into 2 groups according to gender. All 159 patients were available for response evaluation. The RR to EGFR-TKI was not influenced by gender. The RRs in the groups are listed in Table II. In males, the RR was 87.0%, compared with 82.2% in females, for any-line EGFR-TKI (p=0.416). Among the 159 patients, 84 were treated with EGFR-TKIs in the first-line setting (Tab. III); the first-line RR was not statistically significantly different between males and females (91.2% vs 84.0%; p=0.339).
Univariate and Multivariate Analysis of Response Rate (N=159)
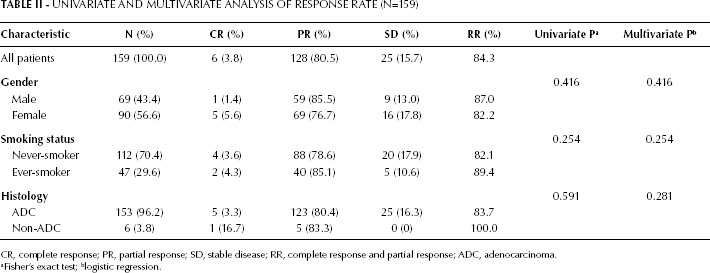
CR, complete response; PR, partial response; SD, stable disease; RR, complete response and partial response; ADC, adenocarcinoma.
Fisher's exact test;
logistic regression.
Relationship Between Clinical Variables and Anti-Tumor Efficacy in Patients Treated with First-Line TKI
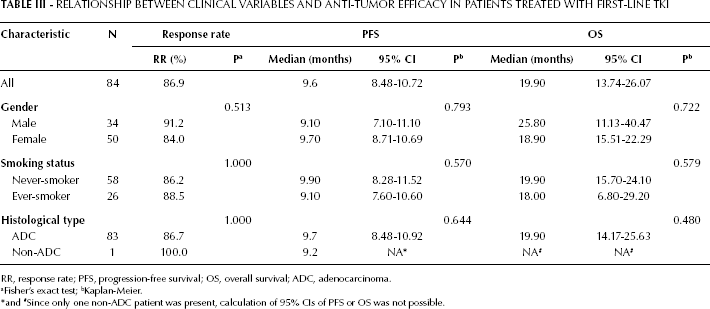
RR, response rate; PFS, progression-free survival; OS, overall survival; ADC, adenocarcinoma.
Fisher's exact test;
Kaplan-Meier.
*and #Since only one non-ADC patient was present, calculation of 95% CIs of PFS or OS was not possible.
The median PFS for any-line EGFR-TKI treatment was 9.6 months in males (95% CI, 8.55-10.65) versus 9.7 months in females (95% CI, 8.77-10.63; p=0.494; (Fig. 1A). The median PFS for first-line EGFR-TKI-treated patients was 9.1 months (95% CI, 7.10-11.10) for males and 9.7 months (95% CI, 8.71-10.69; p=0.793) for females (Fig. 1B). Due to the imbalance in smoking history distribution between genders, we further subdivided the never-smokers (n=112) into 2 groups by gender. No significant difference was found between genders in the never-smokers subgroups in the any-line TKI treatment (p=0.653) or the first-line TKI treatment (p=0.589).
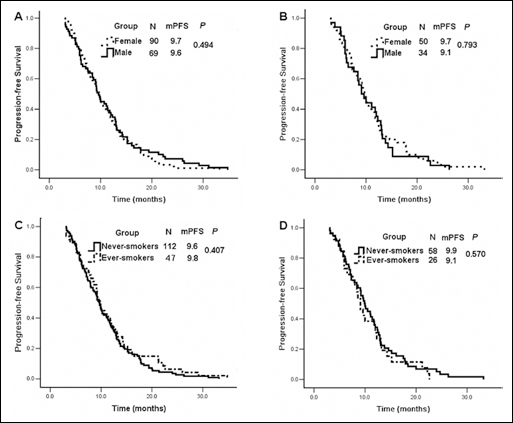
Progression-free survival (PFS) of EGFR-mutant patients treated with EGFR-TKIs. (A) TKIs given as any-line therapy according to gender. (B) TKIs given as first-line therapy according to gender. (C) TKIs given as any-line therapy according to smoking status. (D) TKIs given as first-line therapy according to smoking status.
The OS for first-line TKI therapy (medians: 23.8 vs 18.9 months, p=0.722) and beyond first-line TKI therapy (medians: 22.9 vs 20.5 months; p=0.571) were similar in males and females (Fig. 2A, 2B).
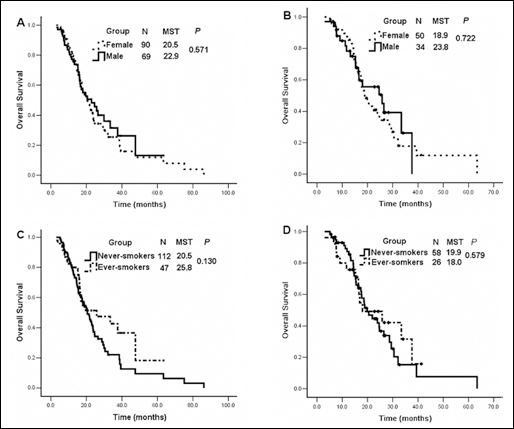
Median survival time (MST) of EGFR-mutant patients treated with EGFR-TKIs. (A) TKIs given as any-line therapy according to gender. (B) TKIs given as first-line therapy according to gender. (C) TKIs given as any-line therapy according to smoking status. (D) TKIs given as first-line therapy according to smoking status.
Smoking status
The whole cohort was divided into 2 categories according to smoking history. The RR of any-line TKI therapy was 82.1% in never-smokers and 89.4% in ever-smokers (p=0.254), and the RRs in the first-line setting were, respectively, 86.2% and 88.5% (p=1.000). PFS did not differ significantly between the never-smokers and ever-smokers in the any-line setting (respectively 9.6 vs 9.8 months; p=0.407) or the first-line setting (respectively 9.9 vs 9.1 months; p=0.570) (Fig. 1C, 1D).
Similarly, smoking history did not affect OS. The median OS of any-line TKI-treated patients was 20.5 (95% CI, 17.29-23.71) in never-smokers and 25.8 in ever-smokers (95% CI, 7.33-44.26; p=0.130). Never-smokers tended to live longer than ever-smokers in the first-line setting (p=0.579), although the difference was not significant (Fig. 2B).
Histology type
There were only 6 non-adenocarcinomas (3 adenosquamous carcinomas and 3 squamous carcinomas) harboring EGFR mutations (see Supplemental Tab. I for further information), giving a 3.8% incidence of EGFR mutation. Five of the 6 non-adenocarcinoma patients were males, and 4 had a smoking history. The RR in the 6 non-adenocarcinoma patients was 100.0%, compared with 83.7% (128/153) in the adenocarcinoma patients (p=0.591).
Clinical, Mutational, and Sampling Information of Patients with Non-Adenocarcinomas
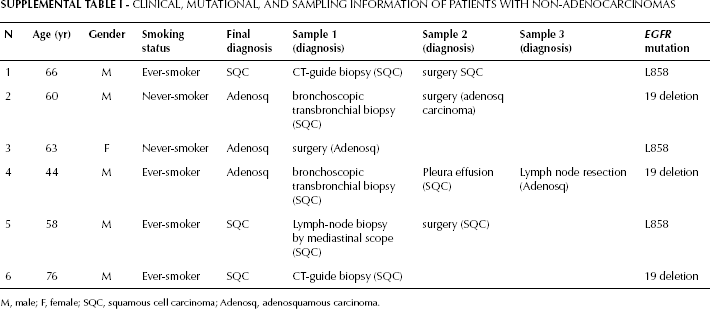
M, male; F, female; SQC, squamous cell carcinoma; Adenosq, adenosquamous carcinoma.
The median PFS was 9.7 months (95% CI, 8.93-10.47) in patients diagnosed with adenocarcinoma and 9.2 months (95% CI, 4.56-13.84) in non-adenocarcinoma patients (p=0.098; Fig. 3A). Four of the 6 non-adenocarcinoma patients received TKIs in a second-line setting, and the therapeutic efficacy was assessed. Among the 54 adenocarcinoma and the 4 non-adenocarcinoma patients who underwent EGFR-TKI treatment as second-line therapy, the median PFS was 9.9 months (95% CI, 8.22-11.58) in those with adenocarcinomas and 9.2 months (95% CI, 4.50-13.90) in those with non-adenocarcinomas (p=0.251).
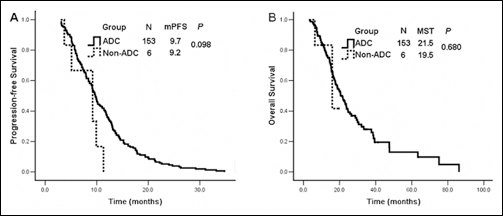
Survival curves of EGFR-mutant patients treated with EGFR-TKIs. (A) PFS for TKIs given as any-line therapy according to pathology. (B) MST on TKIs given as any-line therapy according to pathology.
The 6 non-adenocarcinoma patients were all alive at the time of last follow-up; their median OS was 15.9 months (95% CI, 2.16-29.64), compared with 21.5 months (95% CI, 17.98-25.02) in patients with adenocarcinomas (p=0.680) (Fig. 3B).
Multivariate analysis
In a logistic regression analysis identifying independent predictors of EGFR-TKI responses, no variable was statistically significant (Tab. III).
Age, gender, smoking history, tumor histology, ECOG status, type of EGFR-TKI, type of EGFR mutation, TNM stage, line of TKI, and subsequent treatment were all analyzed in a Cox regression model to evaluate the predictive and prognostic value of each factor for PFS and OS (Tab. IV). Multivariate analysis demonstrated that while gender, smoking status, and tumor histology were not independent prognostic factors, the subsequent treatment affected survival. Compared with patients who received systemic anti-cancer treatment, such as TKIs or chemotherapy, patients who received the best supportive care as post-TKI treatment had a higher risk of death.
Univariate Analysis of Progression-Free Survival and Overall Survival (N=159)
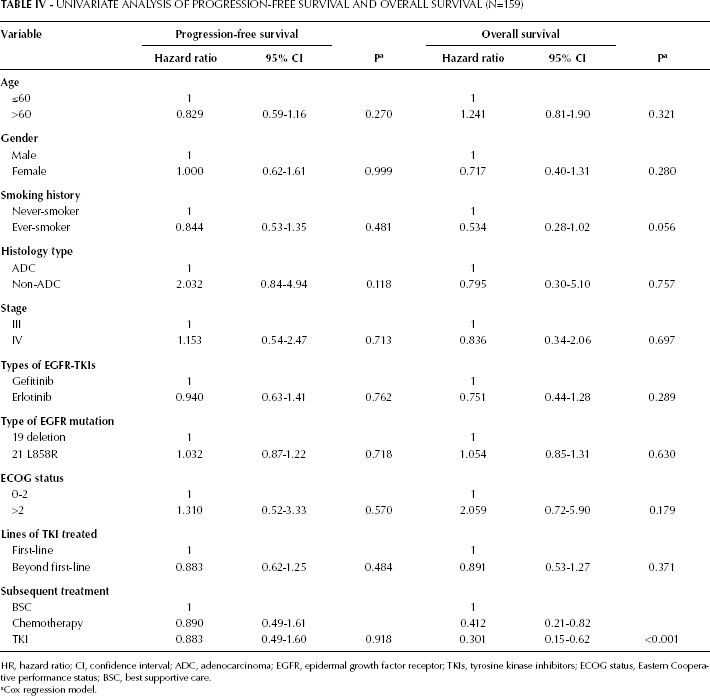
HR, hazard ratio; CI, confidence interval; ADC, adenocarcinoma; EGFR, epidermal growth factor receptor; TKIs, tyrosine kinase inhibitors; ECOG status, Eastern Cooperative performance status; BSC, best supportive care.
Cox regression model.
Discussion
Since the first-generation EGFR-TKIs entered the market, female gender, never-smokers, adenocarcinomas, and Asian ethnicity have been considered predictive factors for EGFR-TKI efficacy. Miller et al (10) reported that never-smokers and adenocarcinoma patients who harbored the bronchioloalveolar subtype of tumors responded to gefitinib. A large retrospective study in Taiwan (18), which included 428 East Asians, demonstrated that gender and smoking history were not independent predictors of EGFR-TKI efficacy, but adenocarcinoma histology was. Clark et al (11) reported that smoking history was an independent predictive factor for erlotinib efficacy. However, the clinical applicability of all of these studies is limited because they were conducted in patients for whom the EGFR-status was unknown. It is well known that EGFR mutations are more common in NSCLC patients of Asian ethnicity, females, never-smokers, and adenocarcinomas, and these factors always correlate with each other (1, 16). The predictive values of these factors need to be clarified in a population with known EGFR status.
All of the patients evaluated in the present study were EGFR mutation-positive. The L858R mutation in exon 21 or a deletion in exon 19 represent about 90% of all EGFR mutations, whereas other rare mutations have less effect on EGFR-TKIs (9). With this in mind, we enrolled only patients with the 2 major sensitive mutations to avoid potential bias. The demographical and clinical features of the EGFR-mutant patients in this study were consistent with those reported in previous clinical trials conducted in Asians (13, 14): females and never-smokers constituted high proportions of the population, the exon 19 deletion was present in 56.6% of the EGFR-mutant cohort, and the remaining 43.4% had the exon 21 point mutation.
To our knowledge, this is the largest study examining the influence of gender, smoking history, and histology on the efficacy of EGFR-TKI therapy and survival outcomes in EGFR-mutant NSCLC patients. Our work demonstrates that supposedly unfavorable clinical factors are not independent predictive and prognostic factors for treatment response or survival in EGFR-mutant patients, and confirm that activating EGFR mutations are the most important predictive factors for TKI efficacy. Our results are in agreement with the subgroup analyses of the OPTIMAL, EURTAC, and WJOTG 3405 trials, which suggested that the benefit to PFS was consistent across all clinical subgroups, including gender, smoking status, and tumor histology (12, 14, 15).
Morita et al (19) performed a combined survival analysis and identified 148 NSCLC patients with EGFR mutations, finding that female gender and never-smoker status were not associated with prolonged PFS or OS among EGFR-mutant patients. Satouchi et al (9) reported that smoking status did not influence OS in 28 EGFR-mutant patients, but no information on PFS for TKIs was provided in this study. Our study was more powerful not only because of a larger sample size of EGFR mutation-positive patients but also more detailed clinical and treatment information. Consequently, by analyzing a larger group of EGFR-mutant patients, subdivided in basis of the lines of TKI treatment, we showed that gender, smoking status, and histology may not be independent predictive factors for survival.
Few studies have focused on the efficacy of EGFR-TKIs in non-adenocarcinoma EGFR-mutant patients. The reported incidence of EGFR mutations in non-adenocarcinoma patients ranges from 3.4% to 13.3% (20–24). The incidence of EGFR mutations in our study was 3.8%, close to the lower limit of the reported range. There are several potential reasons for this. First, the methods used to analyze EGFR profiles differed in the studies. Cho et al (22) reported an 8.4% (24/250) EGFR mutation rate in non-adenocarcinomas using direct DNA sequencing. Tatematsu et al (24) detected a 4% (5/122) EGFR mutation rate using direct sequencing in patients with small-cell lung cancer. Recently, Hata et al (21) reported a 13.3% (33/249) incidence of EGFR mutations in patients with squamous lung cancer using the highly sensitive PNA-LNA PCR clamp method. At our institute, PCR amplification followed by direct sequencing has been the standard method for EGFR testing, and this method is moderately sensitive for EGFR mutation analysis (17). Thus, the rate of EGFR mutations in the present study is consistent with rates in other studies using direct sequencing, and lower than rates in studies using the PNA-LNA PCR clamp method. Second, only some non-adenocarcinoma EGFR-mutant patients received EGFR-TKIs in previous reports (21, 22, 24), but all patients underwent EGFR-TKI treatment in our study; some non-adenocarcinoma patients with EGFR mutations were not enrolled in the analysis.
In our study, 5 of the 6 non-adenocarcinoma patients achieved a partial response (PR), and the rest of them achieved a complete response (CR); this response rate of 100% was higher than the previously reported rates (21–23). Our results suggest that EGFR status analysis is vital for each patient, even for males, ever-smoking and non-adenocarcinoma patients who were regarded as having low EGFR mutation rate. We thus recommend EGFR testing for all NSCLC patients.
Like any retrospective study, also ours has some limitations. The first potential cause of bias could be in the smoking history: data on second-hand and environmental smoke exposure, which may be regarded as interference, were not recorded. Women who were never-smokers may be exposed to second-hand cigarette smoke and cooking fumes, which are also considered risk factors for lung cancer. Furthermore, due to the limited study population we could not subdivide the ever-smokers into light-smokers and heavy-smokers to evaluate any possible dose-related effect on EGFR-TKI efficacy. Second, although we evaluated the percentage of tumor samples before genetic analysis, most of the tissue samples were small biopsies and may not represent the whole tumor, according to the theory of tumor heterogeneity. However, for patients with stage III-IV lung cancer, it is difficult to obtain the whole tumor sample in clinical practice. Third, differences in terms of physician's preferences for TKIs may lead to potential bias. Finally, although our study population is the largest in this field to date, some clinical characteristics such as histology were unbalanced between groups. Furthermore, the interpretation of survival outcomes based on histology type might have been complicated by the small sample size of some types of tumors, despite the use of multivariate analysis.
In conclusion, our data demonstrate that gender, smoking history, and pathology did not predict the efficacy of EGFR-TKIs in EGFR-mutant NSCLC patients. Although patients with non-adenocarcinomas had a lower incidence of EGFR mutations, EGFR-TKIs were also effective in these patients. Thus, before EGFR-TKI treatment, EGFR status should be routinely tested in all patients, even those with non-adenocarcinoma histology, of male gender, or smokers. Further studies are needed to validate these findings in larger-scale study cohorts.
Footnotes
Acknowledgements
The authors thank Zhi-hong Chen, Jian-guang Chen, Shi-liang Chen, and Zhi Xie for careful laboratory data analysis, and Fei-yu Niu and Zhi-ming Huang for clinical information collection.
